[box type=”bio”] Learning Point of the Article: [/box]
The rarity of Phosphaturic tumor / oncogenic osteomalacia occurrence, the pattern of diagnosis using serum calcium, phosphorus, alkaline phosphatase and gallium dotanoc scan and management.
Case Report | Volume 9 | Issue 1 | JOCR January – February 2019 | Page 49-52 | T.Shenbaghavalli, J. K. Giriraj Harshavardhan, P. Gopinath Menon. DOI: 10.13107/jocr.2250-0685.1304
Authors: T.Shenbaghavalli[1], J. K. Giriraj Harshavardhan[1], P. Gopinath Menon[2]
[1]Department of Orthopaedic Surgery, Sri Ramachandra Medical College and Research Institute, Sri Ramachandra Institute of Higher Education and Research, Chennai, Tamil Nadu, India,
[2]Department of Orthopaedics, Sri Ramachandra Medical College and Research Institute, Sri Ramachandra Institute of Higher Education and Research, Chennai, Tamil Nadu. India.
Address of Correspondence:
Dr. T Shenbaghavalli,
Department of Orthopaedic Surgery, Sri Ramachandra Medical College and Research Institute, Sri Ramachandra Institute of Higher Education and Research, Porur, Chennai -600116, Tamil Nadu. India.
E-mail: shenscorp@gmail.com
Abstract
Introduction: Oncogenic osteomalacia is a rare paraneoplastic syndrome of acquired hypophosphatemic osteomalacia, resulting from a deficit in renal tubular phosphate reabsorption, in which fibroblast growth factor 23 seems to be implicated.
Case Report: A 42-year-old male presented with complaints of low back pain for the past 4 years and inability to walk for the past 3 years. Blood investigations showed hypophosphatemia and elevated serum alkaline phosphatase. Radiographs and magnetic resonance imaging revealed features of osteomalacia. DOTONAC PET-computed tomography (CT) showed a lesion in the greater trochanter suggestive of a phosphaturic mesenchymal tumor. CT-guided biopsy of the lesion was done which confirmed the diagnosis. The tumor was resected by intralesional methods (burring and cementation) with correction of hypophosphatemia by oral phosphorus supplementation. Patient clinically improved and was able to walk with support at the time of discharge.
Conclusion: Phosphaturic tumor was localized to be in the posterior aspect of the right greater trochanter. To find the tumor was a major diagnostic challenge involving various diagnostic modalities. The occurrence of such a condition is rare and it may take even years to correctly diagnose the disease.
Keywords: Phosphaturic mesenchymal tumor, proximal femur, intralesional extended curettage
Introduction
Phosphaturic mesenchymal tumors (PMTs) are very rare tumors which are frequently associated with tumor-induced osteomalacia (TIO), a paraneoplastic syndrome that manifests as renal phosphate wasting [1]. It is caused due to abnormal tumor production of fibroblast growth factor 23 (FGF23) [2]. It may cause bone fractures, bone and muscular pains, and sometimes loss of height and even weight. Weight loss is unusual, but sometimes observed, and could be explained by general debilitated state of the patient with consequent poor nutrient intake and loss of muscle mass [3]. The diagnosis and locating the responsible tumors can be challenging and the tumor is often missed or misdiagnosed [2]. We present a case of oncogenic osteomalacia associated with PMTs, which presented mimicking paraperesis.
Case Report
A 42-year-old male presented with complaints of low back pain for the past 4years and inability to walk for the past 3 years. He was apparently normal 4years back when he developed pain over the lower back which was insidious in onset, gradually progressive, intermittent in nature, radiating to bilateral lower limbs, aggravated on any movement of both lower limbs or turning sideways, decreased on re stand medications. History of rest pain was present but no history of night cries or diurnal variations. He was mobilizing with walker during the first few years of pain but he developed complete inability to walk even with support for the past 1 year. There was no history of fever or trauma, loss of weight, or appetite. There was no history of loss of sensations in the upper or lower limbs and bowel or bladder incontinence. He also gave a history of intermittent diffuse pain in the chest and left elbow for the past 3years which was not progressive. Treatment history included medications for pain for the past 4years. There was no significant family history. General examination and systemic examination of the cardiovascular, respiratory, and abdomen showed no significant findings. There was diffuse tenderness over the dorsolumbar spine from D6-L4. Movements of both hips were severely painful with tenderness over the proximal femur. Neurological examination of the upper limbs was normal. Motor power of muscles acting on the hip and knee could not be assessed due to severe pain, but muscles acting on the ankle and foot were normal. There was no sensory blunting and perianal sensations were normal with normal anal sphincter contraction. Babinski response was normal. The findings suggested that there was no cord or cauda equina compression but the back and bilateral hips pain was causing a pain inhibition of the muscles acting on the hip and knee. On examination of the left elbow, swelling and tenderness were present over the proximal third ulna. Movements of the wrist and forearm were full but associated with mild pain. All routine blood investigations were done. X-rays of the spine (Fig. 1) showed scalloping of the end plates, X-ray of the pelvis showed bilateral Looser’s zone in the proximal femurs (Fig. 2),andX-ray of the left elbow showed an old proximal 1/3 ulna fracture nonunion (Fig. 3).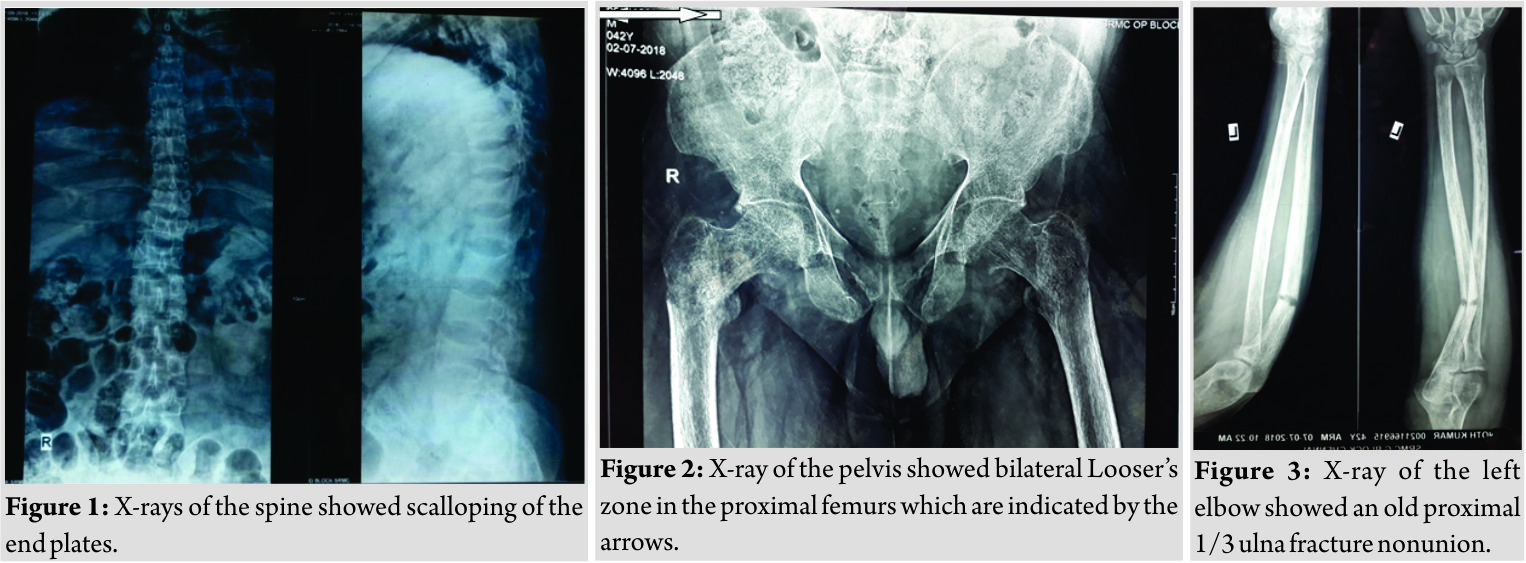 He was found to have elevated levels of alkaline phosphatase (575 U/L) and low levels of phosphorus (1.1mg/dl) and Vitamin D 25OH (16.3ng/ml). 24 h urine phosphorus excretion was also found to be low (351mg/day). Hypophosphatemia was further evaluated by screening for FGF23 which was positive (>300RU/ml). Magnetic resonance imaging (MRI) of the spine showed compressed (biconcave) lumbar and dorsal vertebral bodies with multiple loose zones scattered along the ribs and spinous processes of dorsal vertebrae. Screening of hips and pelvis showed looser zones in the bilateral proximal femur, both iliac wings, sacroiliac joints, and pubic rami. Gallium DOTANOC scan done, as advised by the endocrinologist, suggested a well-defined sclerotic lesion with focal abnormal increased 68Ga uptake in the greater trochanteric region of the right proximal femur posteriorly – suggesting a possibility of oncogenic osteomalacia (Fig. 4).
He was found to have elevated levels of alkaline phosphatase (575 U/L) and low levels of phosphorus (1.1mg/dl) and Vitamin D 25OH (16.3ng/ml). 24 h urine phosphorus excretion was also found to be low (351mg/day). Hypophosphatemia was further evaluated by screening for FGF23 which was positive (>300RU/ml). Magnetic resonance imaging (MRI) of the spine showed compressed (biconcave) lumbar and dorsal vertebral bodies with multiple loose zones scattered along the ribs and spinous processes of dorsal vertebrae. Screening of hips and pelvis showed looser zones in the bilateral proximal femur, both iliac wings, sacroiliac joints, and pubic rami. Gallium DOTANOC scan done, as advised by the endocrinologist, suggested a well-defined sclerotic lesion with focal abnormal increased 68Ga uptake in the greater trochanteric region of the right proximal femur posteriorly – suggesting a possibility of oncogenic osteomalacia (Fig. 4).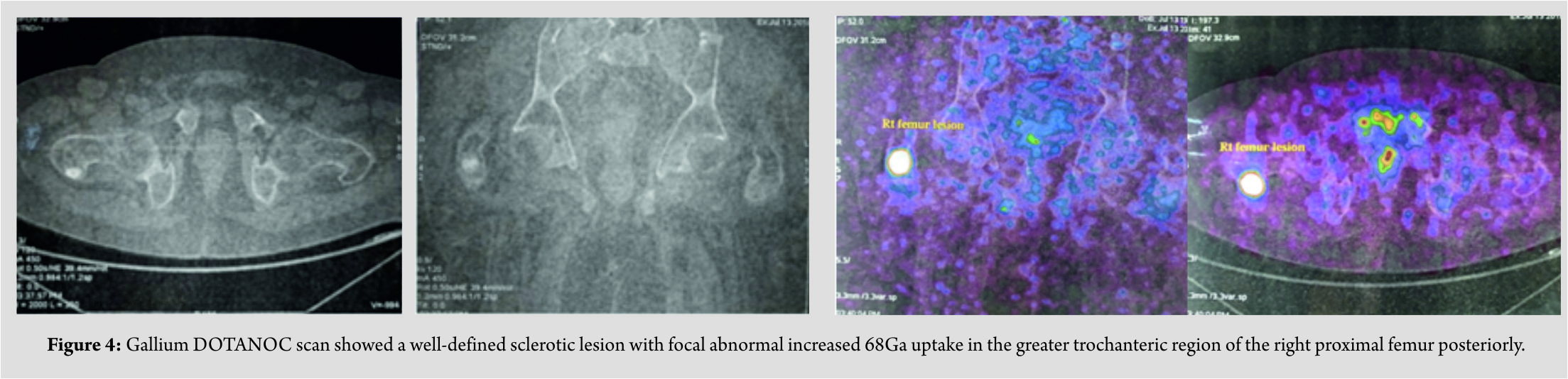 Endocrinologist opinion was sought, and the patient was started on oral phosphorus supplements. Serial serum phosphorus levels showed improvement (1.1–2.0–3.1 mg/dl),but the patient’s symptoms did not improve. A computed tomography (CT)-guided biopsy of the right proximal femur sclerotic lesion was done and was reported as spindle cell neoplasm consistent with PMT. Phosphaturic tumor was localized to be in the posterior aspect of the right greater trochanter. The intraoperatively patient was positioned in the left lateral position to facilitate access to the posterior aspect of the greater trochanter. An incision was made over the greater trochanter. Hip was initially rotated to facilitate the approach. A 2 cm × 1 cm oval cortical window was made in this region sparing the tip. The tumor was identified as a hard mass in the subcortical surface. The hard mass was burred and the cavity was packed with cement (Fig. 5). Resected intraoperative specimen sent for histopathology showed features consistent with a PMT. Following surgery, back pain increased probably due to the positioning of the patient for spinal and epidural anesthesia. The patient was given thoracolumbosacral orthoses, and oral phosphate supplements were continued. Pain over the back gradually improved over a period of 2–3 weeks. Patient was mobilized with spinal orthoses and walker support 3 weeks following surgery – the 1sttime the patient had walked after 3 years! He is now walking up to a 100 m with walker support. Oral phosphorous supplementation was gradually reduced with improving and sustained levels of serum phosphorus.
Endocrinologist opinion was sought, and the patient was started on oral phosphorus supplements. Serial serum phosphorus levels showed improvement (1.1–2.0–3.1 mg/dl),but the patient’s symptoms did not improve. A computed tomography (CT)-guided biopsy of the right proximal femur sclerotic lesion was done and was reported as spindle cell neoplasm consistent with PMT. Phosphaturic tumor was localized to be in the posterior aspect of the right greater trochanter. The intraoperatively patient was positioned in the left lateral position to facilitate access to the posterior aspect of the greater trochanter. An incision was made over the greater trochanter. Hip was initially rotated to facilitate the approach. A 2 cm × 1 cm oval cortical window was made in this region sparing the tip. The tumor was identified as a hard mass in the subcortical surface. The hard mass was burred and the cavity was packed with cement (Fig. 5). Resected intraoperative specimen sent for histopathology showed features consistent with a PMT. Following surgery, back pain increased probably due to the positioning of the patient for spinal and epidural anesthesia. The patient was given thoracolumbosacral orthoses, and oral phosphate supplements were continued. Pain over the back gradually improved over a period of 2–3 weeks. Patient was mobilized with spinal orthoses and walker support 3 weeks following surgery – the 1sttime the patient had walked after 3 years! He is now walking up to a 100 m with walker support. Oral phosphorous supplementation was gradually reduced with improving and sustained levels of serum phosphorus.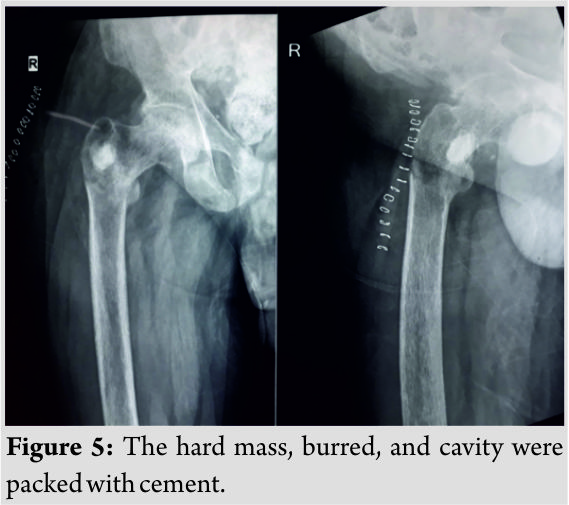
Discussion
In 1987, Weidner first described mesenchymal tumors causing osteomalacia histological morphology and divided it into four histological types: PMTs connective tissue subtype; osteoblastoma-like subtype; nonossifying fibroma-like subtype; and ossifying fibroma-like subtype [4]. Weidner and Santa Cruz[4] emphasized that these tumors are frequently very small and occur in peculiar locations making discovery difficult (e.g., a 1-cm tumor of the big toe). It may occur anywhere in the body including soft tissue and bone, thigh, foot, hand, waist, hips, head and neck, and back [5]. The most frequent are bone tumors (55% of cases) occur in the long bones of the lower limbs, such as the femur and tibia [6]. Our tumor presented in greater trochanter. The main biochemical manifestations of this disorder include hypophosphatemia, inappropriately low or normal tubular reabsorption of phosphate, low serum calcitriol concentrations, high serum alkaline phosphatase, and high or normal serum FGF-23 concentrations [7]. The clinical manifestations include fatigue, muscle weakness, bone pain, and spontaneous fractures [2]. In our case report, the main clinical findings were the inability to walk, bone pain, and spontaneous fracture. Hypophosphatemia with positive FGF-23 concentrations was the diagnostic finding. The results of Stewart et al. [8] support the concept that FGF23 is an important regulator of phosphate metabolism. Administration of synthetic FGF23 and genetic over expression of FGF23 in mice produce hypophosphatemia and renal phosphate loss. Genetic inactivity of FGF23 (in both mice and humans) causes hyperphosphatemia. Moreover, FGF23 production can be regulated by the phosphate both in vitro and in vivo [8]. FGF23 has been reported as a common diagnostic factor for oncogenic osteomalacia [5, 6]. Finding the tumors can be a major diagnostic challenge as these tumors can occur anywhere and may involve total body MRI, CT, scintigraphy using radio labelled somatostatin analog, and positron emission tomography with CT and selective venous sampling for FGF23 [3, 9]. CT is useful in localizing TIO lesions missed by conventional imaging [10]. Galium – 68 labeled somatostatin analogs are short peptide analogs of somatostatin which are linked to the positron-emitter Ga-68 by a chelate, namely, 1, 4, 7, 10-tetraazacyclododecane- 1, 4, 7, 10-tetraacetic acid (DOTA). They bind to the somatostatin receptors which are over-expressed on neuroendocrine tumor cells. Three major peptides currently available are Ga-68 DOTA-TOC, DOTA-NOC, and DOTA-TATE and all have good success rates. The choice of which peptide to be used is based on cost, availability, and logistical factors [11]. X-ray, MRI, gallium DOTANOC scan, and CT-guided biopsy of the right proximal femur lesion confirmed the tumor in this case report. Following surgical removal, the patient has improved significantly. The prognosis of patients with osteogenic osteomalacia is excellent in most cases, reflecting the generally benign nature of the disease. Reversal of bone changes gradually occurs following resection of the tumor. However, vigilant post-operative follow-up is necessary due to its rare metastatic nature [2].
Conclusion
The diagnosis of phosphaturictumoris challenging and often misdiagnosed. Phosphaturic tumor/oncogenic osteomalacia must be included in the differential diagnosis in patients with diffuse bone pain and difficulty to walk. Due to the nonspecific nature of the presenting symptoms, the condition is often detected late sometimes after years. Elevated levels of alkaline phosphatase and low levels of phosphorus and gallium DOTANOC scan led us to the diagnosis of oncogenic osteomalacia.
Clinical Message
The diagnosis of phosphaturic tumor/oncogenic osteomalacia requires a high degree of suspicion. Patients with this condition present with severe bone pain generally. Altered levels of serum alkaline phosphatase, calcium, and phosphorous will help to suspect this condition which can be confirmed by a Gallium DOTANOC scan. Ablation of the tumor leads to early recovery and resolution of symptoms.
References
1. Ghorbani-Aghbolaghi A, Darrow MA, Wang T. Phosphaturic mesenchymal tumor (PMT): Exceptionally rare disease, yet crucial not to miss. Autops Case Rep 2017;7:32-7.
2. Zuo QY, Wang H, Li W, Niu XH, Huang YH, Chen J, et al. Treatment and outcomes of tumor-induced osteomalacia associated with phosphaturic mesenchymal tumors: Retrospective review of 12 patients. BMC Musculoskelet Disord 2017;18:403.
3. Dadoniene J, Miglinas M, Miltiniene D, Vajauskas D, Seinin D, Butenas P, et al. Tumour-induced osteomalacia: A literature review and a case report. World J Surg Oncol 2016;14:4.
4. Weidner N, Santa Cruz D. Phosphaturic mesenchymal tumors. A polymorphous group causing osteomalacia or rickets. Cancer 1987;59:1442-54.
5. Qiu S, Cao LL, Qiu Y, Yan P, Li ZX, Du J, et al. Malignant phosphaturic mesenchymal tumor with pulmonary metastasis: A case report. Medicine (Baltimore) 2017;96:e6750.
6. Seijas R, Ares O, Sierra J, Pérez-Dominguez M. Oncogenic osteomalacia: Two case reports with surprisingly different outcomes. Arch Orthop Trauma Surg 2009;129:533-9.
7. Alonso G, Varsavsky M. Tumour-induced osteomalacia: An emergent paraneoplastic syndrome. Endocrinol Nutr 2016;63:181-6.
8. Stewart I, Roddie C, Gill A, Clarkson A, Mirams M, Coyle L, et al. Elevated serum FGF23 concentrations in plasma cell dyscrasias. Bone 2006;39:369-76.
9. Chong WH, Yavuz S, Patel SM, Chen CC, Collins MT. The importance of whole body imaging in tumor-induced osteomalacia. J Clin Endocrinol Metab 2011;96:3599-600.
10. Dutta D, Pandey RK, Gogoi R, Solanki N, Madan R, Mondal A, et al. Occult phosphaturic mesenchymal tumour of femur cortex causing oncogenic osteomalacia-diagnostic challenges and clinical outcomes. Endokrynol Pol 2018;69:205-10.
11. Pruthi A, Pankaj P, Verma R, Jain A, Belho ES, Mahajan H, et al. Ga-68 DOTANOC PET/CT imaging in detection of primary site in patients with metastatic neuroendocrine tumours of unknown origin and its impact on clinical decision making: Experience from a tertiary care centre in India. J Gastrointest Oncol 2016;7:449-61.
 |
 |
 |
| Dr. T. Shenbaghavalli | Dr. J. K. Giriraj Harshavardhan | Dr. P. Gopinath Menon |
| How to Cite This Article: Shenbaghavalli T, Harshavardhan JKG, Menon PG.A Rare Case of Phosphaturic Tumor/Oncogenic Osteomalacia – Diagnostic Challenges and Management Algorithm. Journal of Orthopaedic Case Reports 2019 Jan-Feb; 9(1): 49-52. |
[Full Text HTML] [Full Text PDF] [XML]
[rate_this_page]
Dear Reader, We are very excited about New Features in JOCR. Please do let us know what you think by Clicking on the Sliding “Feedback Form” button on the <<< left of the page or sending a mail to us at editor.jocr@gmail.com




