Neurological deficit though rare in Paget’s disease of spine can be the presenting complaint of the patient.
Dr. Pramod Bhilare, 11/12 Thube Park, 16, Shivaji Nagar, Pune. Maharashtra - 4110 005. India. Email: pramod.bhilare@gmail.com
Introduction: Paget’s disease of bone (PDB) is a metabolic bone disease presenting as polyostotic or monostotic lesions of the spine. Although common in the Anglo-Saxon population, it is rare on the Indian subcontinent. Neurological complications though infrequent can be severe in pagetic spine.
Case Report: We report a case of a polyostotic variant of PDB involving lumbar spine (L2 vertebrae), iliac bones, and femur presenting as chronic low back pain and neurological deficit, i.e., cauda equina syndrome. On initial workup, a diagnosis of PDB was made and given cauda equina compression with neurological deficit, posterior spinal decompression, and biopsy was performed. The histopathological evaluation confirmed the diagnosis and the patient was treated with bisphosphonates for 6 months, along with serial monitoring of alkaline phosphatase levels.
Conclusion: Through this case report, we hope to emphasize that PDB should be considered as a possible cause of neurological symptoms at presentation, especially in elderly patients. Also furthermore, early surgical intervention followed by bisphosphonates therapy can lead to favorable outcomes in such patients.
Keywords: Polyostotic, Paget’s disease, cauda equine syndrome, lumbar spine.
Neurological deficit is an uncommon sequela of the Paget’s disease of bone (PDB). Bisphosphonate therapy remains the main treatment in the management of PDB and its sequelae [1,2,3]. Surgery is rarely required in the management of PDB and is reserved for cases refractory to medical treatment and/or surgical emergencies [4,5,6]. Here, we report a case of polyostotic Paget’s disease involving lumbar spine presenting as a Cauda equina syndrome treated with early surgical decompression followed by bisphosphonate therapy which showed excellent outcome.
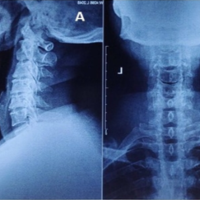
A 75-year-old woman presented to the outpatient clinic with complaints of low back pain (visual analog scale [VAS] 8/10) and progressive weakness in both lower limbs – left more than right since 3 weeks. She also had urinary complaints of difficulty initiating micturition and incontinence. On clinical examination, she had axial tenderness in the lumbar spine with paraspinal muscle spasm. On neurological evaluation, the patient had bilateral tibialis anterior power ⅗ and extensor hallucis longus ⅗ with hypoesthesia in the saddle and perineal area. Anteroposterior (AP) and lateral (Lat) roentgenogram (X-rays) of the lumbar spine and pelvis with hips (pelvis and both hips [PBH]) X-ray revealed a sacralized L5 along with loss of height, expansion, and sclerosis of L2 vertebral body with increased interpedicular distance. PBH AP view with femur showed multiple lucencies and sclerosis with cortical thickening involving the right hemipelvis and the left proximal femur (Fig. 1a-c). Magnetic resonance imaging (MRI) showed hypointense signal on T1-weighted and hyperintense signal in the T2 and short tau inversion recovery weighted sagittal films with an enlarged L2 vertebral body and posterior elements. Axial cuts showed enlargement of the L2 body with hypointense signal in T1 and hyperintense signal in T2 in L2 body and posterior elements with severe cauda equina compression. The preservation of intraosseous fat reduced the likelihood of a neoplastic lesion (Fig. 2a-e). Laboratory investigations showed a tenfold increase in the Serum Alkaline Phosphatase (ALP) (1094 IU/L, Normal range: 44 to 147 IU/L) while the rest of the blood parameters including other liver enzymes and tumor markers were within normal limits. These findings were suggestive of a bony abnormality either metabolic or neoplastic for which positron emission tomography computed tomography (PET-CT) was done which revealed cortical thickening and sclerosis in almost entire right iliac bone, left proximal femur, and L2 vertebra with increased FDG activity (Fig. 3a-c).
Laboratory investigations showed a tenfold increase in the Serum Alkaline Phosphatase (ALP) (1094 IU/L, Normal range: 44 to 147 IU/L) while the rest of the blood parameters including other liver enzymes and tumor markers were within normal limits. These findings were suggestive of a bony abnormality either metabolic or neoplastic for which positron emission tomography computed tomography (PET-CT) was done which revealed cortical thickening and sclerosis in almost entire right iliac bone, left proximal femur, and L2 vertebra with increased FDG activity (Fig. 3a-c).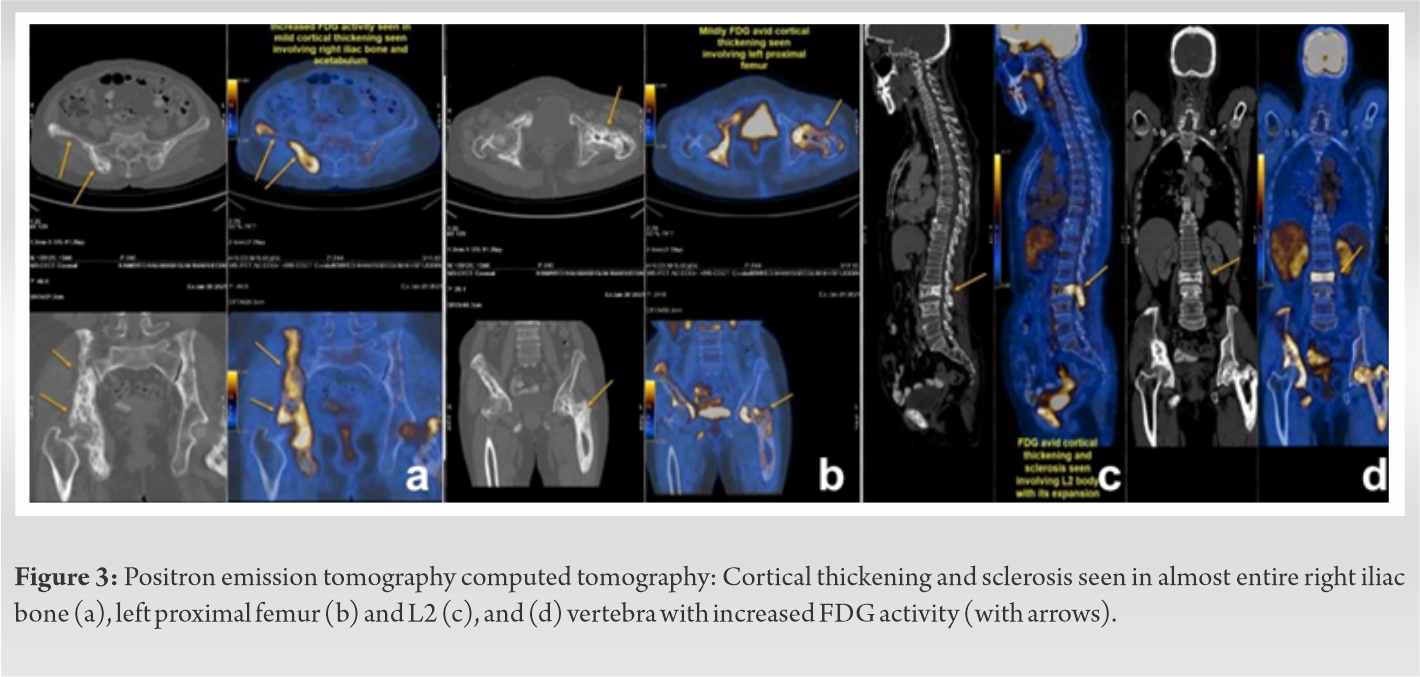 All these investigations led to a probable diagnosis of a polyostotic Paget’s disease of the bone with severe cauda compression causing the neurological deficit. Since the patient had radicular symptoms, neurodeficit in the form of weakness of ankle dorsiflexion, and bladder involvement surgical intervention was planned. Clinico-radiological diagnosis of polyostotic Paget’s disease was made for which the patient underwent L2 laminectomy, decompression, and transpedicular biopsy from the L2 vertebral body, which was sent for histopathological examination. Intraoperatively blood loss of approximately 300 ml was adequately replenished by crystalloids.
All these investigations led to a probable diagnosis of a polyostotic Paget’s disease of the bone with severe cauda compression causing the neurological deficit. Since the patient had radicular symptoms, neurodeficit in the form of weakness of ankle dorsiflexion, and bladder involvement surgical intervention was planned. Clinico-radiological diagnosis of polyostotic Paget’s disease was made for which the patient underwent L2 laminectomy, decompression, and transpedicular biopsy from the L2 vertebral body, which was sent for histopathological examination. Intraoperatively blood loss of approximately 300 ml was adequately replenished by crystalloids.
Postoperatively patient had symptomatic improvement with VAS 3/10. Histopathology report revealed woven bone with thick trabeculae, lamellar bone showing mosaic pattern, prominent irregular cement lines, and osteoclastic rimming. Stroma showed hypervascularity with congested blood vessels (Fig. 4a-e). These features confirmed the diagnosis of PDB.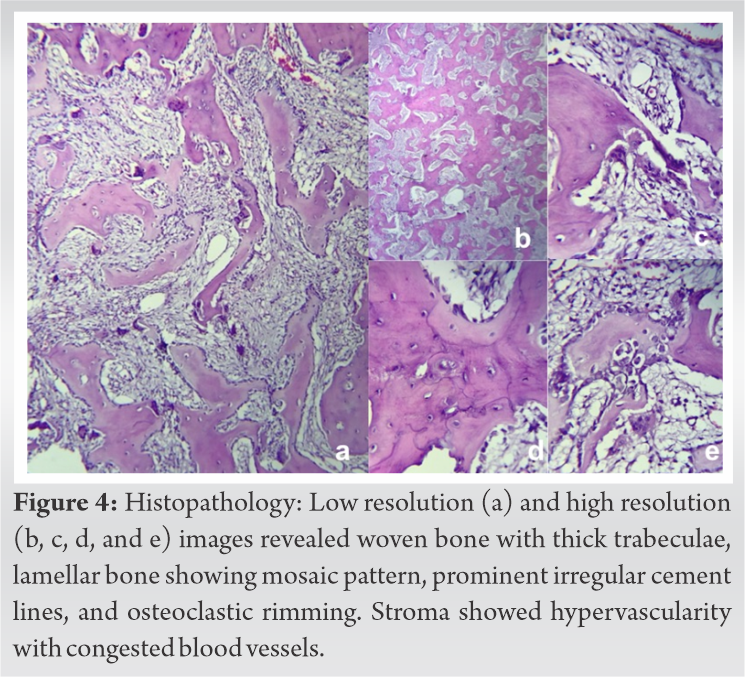 Postoperatively bisphosphonate therapy in the form of alendronate 80 mg daily was given for 6 months. The patient was followed up with serial monitoring of serum ALP levels which showed declining trends. At 1-year follow-up patient symptomatically and neurologically improved with normal power in both lower limbs and serum ALP within normal limits (Fig. 5a, b).
Postoperatively bisphosphonate therapy in the form of alendronate 80 mg daily was given for 6 months. The patient was followed up with serial monitoring of serum ALP levels which showed declining trends. At 1-year follow-up patient symptomatically and neurologically improved with normal power in both lower limbs and serum ALP within normal limits (Fig. 5a, b).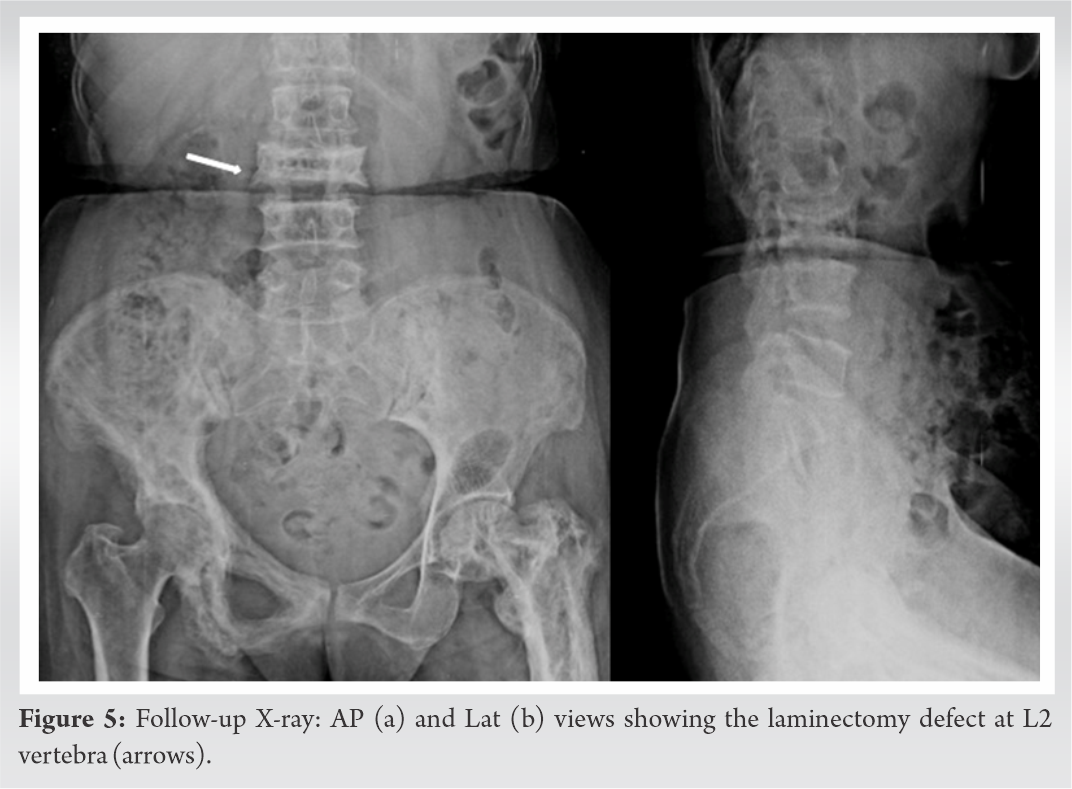
PDB is an osteo-metabolic disorder frequently encountered in Anglo-Saxon populations – particularly Europe, with the prevalence of 3.7% [1,2,3]. PDB is encountered infrequently in China, Japan, India, and the Middle East countries [3,7]. Men are more commonly affected than women and the typical patient is >40 years of age at presentation [2,3]. Although the etiology is not yet well understood, the disease itself is characterized by excessive bone resorption followed by abundant new bone formation due to an increase in osteoclastic and osteoblastic activity. Eventually, the bone marrow is replaced by fibrous and vascular tissue. These processes lead to “enlarged, unnaturally curved, and misshapen” bones as described by Sir James Paget in 1877 [3]. Usually, more than one bone is affected (polyostotic variant) and the commonly affected bones are the pelvis, spine, femur, and skull in the same order [1,3]. The spine is involved in half of all polyostotic affections, out of which PDB is most common in the lumbar region (58%), followed by the thoracic (45%) and cervical (14%) [8,9].
Vertebral PDB can have a varied clinical presentation from back pain, spinal stenosis, and neural dysfunction. While each of these clinical scenarios can be caused by a variety of pagetic complications, but majority time PDB and its complications are entirely asymptomatic [2,7,10]. In most symptomatic PDB, back pain is the most common clinical symptom which varies from 11 to 54%. Different causes for back pain in PDB have been described, that is, periosteal stretching, vascular engorgement, microfractures to overt fractures of vertebrae and sacrum, facet arthritis, intervertebral disc disease, spondylolysis/spondylolisthesis, and very rarely sarcomatous degeneration [1,2]. In 50% of cases, back pain is due to complicating factors including fracture, spinal stenosis, and facet arthropathy; less commonly due to spondylolysis with or without spondylolisthesis and discal involvement [10,11]. Spinal stenosis in PDB of the spine has a prevalence of 33% of cases. Spinal stenosis can present clinically as back pain and/or neurological dysfunction, as a consequence of cord or nerve root compression. Patients with stenosis in PDB can have a wide array of presentations with severe stenosis seen on imaging but present without clinical symptoms and patients with mild or moderate stenosis can present with back pain. The proportion of symptomatic patients with spinal stenosis ranges from 26 to 33% [2,9,12]. It is caused due to disturbance in the bone remodeling process which, in turn, leads to a subsequent decrease in the spinal canal size [1,10].
Neural dysfunction in PDB can be due to spinal stenosis (33%), more commonly with thoracic spine involvement and less commonly with cervical spine involvement [1,2]. The less frequent neural involvement in lumbar spine involvement is due to the relatively capacious spinal canal in the lumbar region [5,13]. Although the pagetic process itself, together with compressive effects from the expanding bone, can give rise to symptoms and signs, other complications may develop and contribute to the symptoms complex of neural dysfunction, that is, ligament ossification, spondylolisthesis, fracture retropulsion, extraosseous involvement, and very rarely due to “arterial steal syndrome” [5,14]. The “arterial steal phenomenon” is described as a deprivation of blood supply to the spinal cord due to the preferential blood flow of the pagetic vertebra [14]. Few cases of PDB have been reported in the literature who presented with cord and/or nerve root compression causing neurological deficit [5,15]. In recent years a decline in the prevalence of the disease has been reported in most, but not all, countries in which this was estimated and it has been suggested that the clinical severity of the disease has been attenuated [3,16]. Hence, the possibility of this disease in the differential diagnosis is rarely considered; nowadays, especially in the Indian subcontinent. In our case, the patient had symptoms due to acute on chronic severe canal stenosis, presenting as a “cauda equina syndrome.”
The majority of patients with vertebral PDB are asymptomatic, and the disease is detected through incidental findings of an elevated ALP level, radiologic features, and bone scintigraphy [1,2]. Elevated ALP is considered a useful biomarker especially in the diagnosis of new PDB patients. However, monostotic PDB may correlate with total ALP levels within the reference interval, introducing difficulties in both the diagnosis and follow-up management of such patients [1,17]. In our patient’s, total ALP level showed a tenfold increase in the values (1094 IU/L, normal range 90–250U/L).
In PDB the loss of homeostatic control leads to increased osteoblastic and osteoclastic activity and constitutes the background for the main three phases, that is, the initial lytic phase, intervening mixed phase, and the late osteoblastic phase [3,10]. In bones with a low trabecular/cortex ratio such as the skull, femur, and humerus, the early lytic phase is radiologically depicted by a clear leading edge at the interface with normal bone. The lytic phase is usually not detected in bones with a high trabecular/cortex ratio such as the vertebra, sacrum, and pelvis. Vertebral body involvement at radiological diagnosis is virtually always complete, and therefore the leading-edge present in the other affected bones is not seen in the vertebra. These phases can be evident in the same patient and at the same time in different bones including the vertebral column [1,2]. Radiographically, the vertebral body may show a typical “picture frame” appearance due to the combination of trabecular bone hypertrophy and thickening at the end-plate, with apposition/absorption on the periosteal/endosteal surfaces at the anterior and posterior vertebral borders in the later phase of the disease. However, early radiographic appearance shows thickening and hypertrophy of the trabecular bone, parallel to the endplate, which can appear similar to a thickened cortex [2,18]. Radionuclide helps in establishing the diagnosis and to determine the disease’s distribution in PDB patients [2,10,18]. PDB is primarily a disorder of the bone, not of the bone marrow; however, secondary bone marrow changes can occur in the latter stages of the disease on MRI. In osteolysis of pagetic vertebrae, a fat signal within the lesion is a useful clinical determinant for distinguishing between vertebral Paget’s disease and osteolytic metastasis [1,18]. However, in early PDB especially the monostotic vertebral lesion it is not always easily distinguishable from a metastatic tumor; hence, physicians can biopsy the lesion for confirmation. In our case, the patient had multiple bony involvements, (polyostotic variant) with elevated ALP levels, pathognomonic MRI, and PET CT findings which was then confirmed on biopsy.
Treatment of Paget disease mainly consists of a combination of bisphosphonates and calcitonin for 3–6 months duration. Indication for treatment includes pain in pagetic bone, neurological complication, osteolytic lesion with increased fracture risk, involvement of long bones, vertebrae, the base of the skull, asymptomatic joint involvement, and patients undergoing surgery on pagetic bones [1,3,10]. The majority of patients responds well to medical therapy and seldom requires any surgical intervention. The main indications for surgery are neurological deficit and pain not responding to medical management. Surgical interventions in the form of percutaneous vertebroplasty for resistant PDB to open surgery for neurological complications have been described in the literature with varied outcomes [1,13,15]. Our patient presented with cauda equina syndrome secondary to severe canal stenosis at L2 vertebral level; hence, early surgery was performed in the form of open decompression. The early surgical intervention followed by bisphosphonates therapy led to the complete neurological recovery of the patient.
The neurological complications of Paget’s disease is infrequent but can be severe. Paget’s disease should be considered as a possible cause of neurological symptoms at presentation, especially in elderly patients. Treatment should be individualized, which mainly comprises symptomatic treatment, calcitonin, oral, and intravenous bisphosphonates but early surgical intervention in emergencies such as cauda equina syndrome can have favorable outcomes.
The prevalence of pagetic spine is showing decreasing trend in the past two decades and so is its complications. Pagetic spine is seldom considered as a differential in patients presenting with Cauda equine syndrome, especially in Indian subcontinent. Through this case report, we hope to emphasize that PDB should be considered as a possible cause of neurological symptoms at presentation, especially in elderly patients.
References
- 1.Hadjipavlou AG, Gaitanis IN, Katonis PG, Lander P. Paget’s disease of the spine and its management. Eur Spine J 2001;10:370-84. [Google Scholar | PubMed]
- 2.Dell C, Cassar-Pullicino VN, Lalam RK, Tins BJ, Tyrrell PN. The spine in Paget’s disease. Skeletal Radiol 2007;36:609-26. [Google Scholar | PubMed]
- 3.Appelman-Dijkstra NM, Papapoulos SE. Paget’s disease of bone. Best Pract Res Clin Endocrinol Metab 2018;32:657-68. [Google Scholar | PubMed]
- 4.Jorge-Mora A, Amhaz-Escanlar S, Lois-Iglesias A, Leborans-Eiris S, Pino-Minguez J. Surgical treatment in spine Paget’s disease: A systematic review. Eur J Orthop Surg Traumatol 2016;26:27-30. [Google Scholar | PubMed]
- 5.Poncelet A. The neurologic complications of paget’s disease. J Bone Miner Res 1999;14 Suppl 2:88-91. [Google Scholar | PubMed]
- 6.Guyer PB, Shepherd DF. Paget’s disease of the lumbar spine. Br J Radiol 1980;53:286-8. [Google Scholar | PubMed]
- 7.Yates AJ. Paget’s disease of bone. Baillières Clin Endocrinol Metab 1988;2:267-85. [Google Scholar | PubMed]
- 8.Mirra JM, Brien EW, Tehranzadeh J. Paget’s disease of bone: Review with emphasis on radiologic features, Part I. Skeletal Radiol 1995;24:163-71. [Google Scholar | PubMed]
- 9.Meunier PJ, Salson C, Mathieu L, Chapuy MC, Delmas P, Alexandre C, et al. Skeletal distribution and biochemical parameters of Paget’s disease. Clin Orthop Relat Res 1987;217:37-44. [Google Scholar | PubMed]
- 10.Altman RD, Brown M, Gargano F. Low back pain in Paget’s disease of bone. Clin Orthop Relat Res 1987;217:152-61. [Google Scholar | PubMed]
- 11.11. Lander P, Hadjipavlou A. Intradiscal invasion of Paget’s disease of the spine. Spine (Phila Pa 1976) 1991;16:46-51. [Google Scholar | PubMed]
- 12.Cartlidge NE, McCollum JP, Ayyar RD. Spinal cord compression in Paget’s disease. J Neurol Neurosurg Psychiatry 1972;35:825-8. [Google Scholar | PubMed]
- 13.Tancioni F, Di Ieva AD, Levi D, Aimar E, Debernardi A, Colombo P, et al. Spinal decompression and vertebroplasty in Paget’s disease of the spine. Surg Neurol 2006;66:189-91; discussion 191. [Google Scholar | PubMed]
- 14.Herzberg L, Bayliss E. Spinal-cord syndrome due to non-compressive Paget’s disease of bone: A spinal-artery steal phenomenon reversible with calcitonin. Lancet 1980;2:13-5. [Google Scholar | PubMed]
- 15.Demir MK, Yapıcıer Ö, Toktaş ZO, Yılmaz B, Akakın A, Konya D. Lumbar Paget disease with spinal stenosis and conus medullaris compression. Spine J 2016;16:e51-2. [Google Scholar | PubMed]
- 16.Poór G, Donáth J, Fornet B, Cooper C. Epidemiology of Paget’s disease in Europe: The prevalence is decreasing. J Bone Miner Res 2006;21:1545-9. [Google Scholar | PubMed]
- 17.Magnusson P, Davie MW, Sharp CA. Circulating and tissue-derived isoforms of bone alkaline phosphatase in Paget’s disease of bone. Scand J Clin Lab Invest 2010;70:128-35. [Google Scholar | PubMed]
- 18.Wu LC, Tseng CH, Chiang YF, Tsuang YH. Monostotic vertebral Paget’s disease of the lumbar spine. J Chin Med Assoc 2009;72:52-5. [Google Scholar | PubMed]