Spinal epidural cavernous hemangioma usually has insidious presentation in 3rd to 6th decade but hemorrhagic variant can present with sudden onset neurological deficit in as early as 2nd decade individual hence must be considered in differentials.
Dr. Siddharth Manik Katkade, Department of Spine, Sancheti Institute of Orthopaedics and Rehabilitation, Shivajinagar, Pune - 411 005, Maharashtra, India. E-mail: siddharthkatkade@gmail.com
Introduction: Commonly spinal cavernous hemangioma (CH) is found in vertebral body at thoracic level. Purely extraosseous epidural CH contributes only 4% of all CH. In addition, these patients usually present in their 3rd–6th decade of life. Here, we report emergently treated rare case of hemorrhagic extraosseous epidural thoracic CH in a 26-year-old male
Case Report: A 26-year-old male presented with inability to walk without support with acute onset paraparesis and hypoesthesia below D7 with positive myelopathy signs. Magnetic resonance imaging showed posterior epidural well-defined homogenous mass from D6-8 region. PET scan ruled out other primary lesions in body. Intraoperative after D6-8 laminectomy extradural vascular mass was found which was bleeding on touch. With adequate hemostasis complete mass was excised and sent for sampling which revealed CH. The patient showed gradual clinical recovery with complete neurological recovery after 3 months with no signs of clinical and radiological recurrence on 2 years of follow-up.
Conclusion: Extraosseous epidural CH is very rare presentation; hence, the differentials of schwannoma, lymphoma, tubercular/pyogenic epidural abscess, metastasis, and others must always be ruled out. They can present a decade earlier in a case of hemorrhagic CH with rapid progressive neurological deficit which warrants early surgical decompression like in our case and the foraminal or paravertebral extension needs foraminotomy or thoracotomy for complete excision.
Keywords: Spinal cavernous hemangioma, extra-osseous, epidural, hemorrhagic, emergency
Cavernous hemangioma (CH) is not treated as vascular neoplasms rather they are neuroaxial hamartomas or vascular malformations [1]. Majority times they are seen intracranially with supratentotrial region being most common site. Vertebral location is rare but the most common among 5–12% of spinal lesion. Epidural hemangiomas usually are the extension from a vertebral hemangioma and just 4% of these are purely extraosseous epidural. Here, we report a successfully treated rare case of hemorrhagic extraosseous epidural thoracic cavernous hemangioma. Consent mentioning submission of clinic- radiological data related to the case was granted from the patient.
A 26-old-year male student presented to outpatient department with mid back pain since a year and sudden onset weakness in both lower limbs and inability to walk independently over a period of a week. On examination, the patient had ataxia and broad-based gait with bilateral lower limb hypertonia with power MRC grade 2–3, hypoasthesia below D7 dermatome with hyper-reflexic knee, ankle jerks, and ankle clonus present. Babinski sign was positive with intact perianal sensations and intact bowel-bladder functions.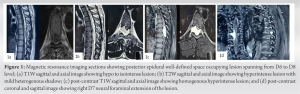
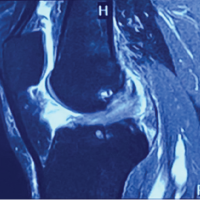
On investigation, hemoglobin level was 17.5 g/dl that rest routine blood tests were normal. Radiographs showed straightening of dorsolumbar spine with no other abnormality. Magnetic resonance imaging (MRI) was suggestive of well-defined homogenous enhancing mass in posterior epidural space spanning D6-8 levels with the right D7 foraminal extension, hyperintense on T2, post-contrast images, and iso to hypointense on T1-weighted images (Fig. 1a-d) and reported as lymphoproliferative disease/extradural neoplasm/metastasis. To rule out other foci of lesion, PET scan was done which was suggestive of low grade intraspinal lesion at D6-8 level with the low-grade FDG uptake and ruling out metastasis or other primary foci (Fig. 2).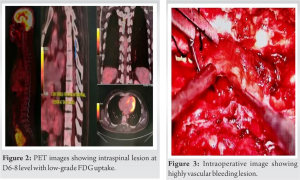
With posterior midline approach D5-8 lamina were exposed and laminectomy was performed. Highly vascular mass (Fig. 3) was observed over posterior aspect of dura which was cauterized adequately with bipolar cautery followed by mass was completely resected and sent for microbiological and histopathological evaluation which showed CH (Fig. 4). Post-operative immediately patient showed signs of neurological improvement. The patient was put on aggressive neurorehabilitation and discharged with advised follow-up on 1st, 3rd and 6th month and yearly thereafter. On 3rd month follow-up, the patient came walking independently with complete neurological recovery. The latest follow-up after 2 years showed no clinical signs of recurrence and MRI showing no signs of remnant tissue and completely decompressed cord (Fig. 5).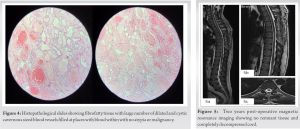
CHs are irregularly dilated vascular lesions having single endothelial layered small capillaries made of numerous lobules interlinked with connective tissue fibrous septae [2]. They are categorized into four types (capillary, cavernous, arteriovenous, or venous) on basis of microscopic examination-based classification, in which cavernous type is major type [3]. As they grow with hypertrophy and not with mitotic activity they are not considered and vascular neoplasms [4]. The most common site is supratentorial area and in spine commonly seen in vertebral body with following frequency thoracic (54–60%), cervical (30%), and lumbar (10%) vertebral regions; they are rarely found in spinal canal and only 4% of all cavernous hemangiomas are purely epidural CHs; our case is one of them. They are more common in thoracic region probably because posterior part of the thoracic spinal canal having less resistance and wider epidural space [5]. However, they create clinical signs with mass effects, cysts or caverns formation, fluid movement within vascular structure, hemorrhage, and thrombosis. CHs are most common seen in age group of 40 (30–60) and in female more than male. In our case, the patient presented at early age with acute onset indicating hemorrhagic CH [2,6,7]. Most commonly, they present with slow progressive paraparesis and radiculopathy [8], but extradural hemorrhage or thrombotic occlusion can accelerate neurological deficits and demands acute presentation [7,9] which was seen in our case. MRI is the investigation of choice for CH which shows well-defined lobular homogenous lesion which is isointense on T1W and hyperintense on T2W images and intensely homogeneous with contrast-enhanced T1-weighted images. However, the possibilities of schwannoma, meningioma, metastasis, lymphoma, multiple myeloma, extraosseous Ewing’s sarcoma, disk fragments, or epidural angiolipoma cannot be ignored [2,9]. The CHs demand early surgical excision in view of tendency to bleed and grow causing neurological deterioration. Total excision of the mass is ideally indicated however concerns like intramedullary extension of the tumor and intraoperative bleeding can limit the total excision and adjuvant radiotherapy can be considered in such situations [2,10]. Massive bleeding can be seen in epidural CHs with less risk of bleeding in intramedullary variant. In our case, we managed to control the bleeding with complete excision of the mass and the adjuvant radiotherapy was not indicated.
Extraosseous epidural CH is very rare presentation; hence, the differentials of schwannoma, lymphoma, tubercular/pyogenic epidural abscess, metastasis, and others must always be ruled out. They can present a decade earlier that is case of hemorrhagic CH with rapid progressive neurodeficit which warrants early surgical decompression like in our case and the foraminal or paravertebral extension needs foraminotomy or thoracotomy for complete excision.
Epidural CH is known to grow slowly causing gradual onset paraparesis, myelopathy in around 4th decade but in hemorrhagic event or thrombotic occlusion patient will present earlier with sudden onset of neurological deficit and it must be included as one of the differentials.
References
- 1.Lee JP, Wang AD, Wai YY, Ho YS. Spinal extradural cavernous haemangioma. Surg Neurol 1990;34:345-51. [Google Scholar | PubMed]
- 2.Khalatbari MR, Abbassioun K, Amirjmshidi A. Solitary spinal epidural cavernous angioma: Report of nine surgically treated cases and review of the literature. Eur Spine J 2013;22:542-7. [Google Scholar | PubMed]
- 3.Zhang L, Yang W, Jia W, Kong D, Yang J, Wang G, et al. Comparison of outcome between surgical and conservative management of symptomatic spinal cord cavernous malformations. Neurosurgery 2016;78:552-61. [Google Scholar | PubMed]
- 4.Dobyns WB, Michels VV, Groover RV, Mokri B, Trautmann JC, Forbes GS, et al. Familial cavernous malformations of the central nervous system and retina. Ann Neurol 1987;21:578-83 [Google Scholar | PubMed]
- 5.Feng J, Xu YK, Li L, Yang RM, Ye XH, Zhang N, et al. MRI diagnosis and preoperative evaluation for pure epidural cavernous hemangiomas. Neuroradiology 2009;51:741-7. [Google Scholar | PubMed]
- 6.Zhong W, Huang S, Chen H, Sun H, Cai B, Liu Y, et al. Pure spinal epidural cavernous hemangioma. Acta Neurochir (Wien) 2012;154:739-45. [Google Scholar | PubMed]
- 7.Hemalatha AL, Ravikumar T, Chamarthy NP, Puri K. A pure epidural spinal cavernous hemangioma-with an innocuous face but a perilous behaviour!! J Clin Diagn Res 2013;7:1434-5. [Google Scholar | PubMed]
- 8.Hegde A, Mohan S, Tan KK, Lim CC. Spinal cavernous malformations: Magnetic resonance imaging and associated findings. Singapore Med J 2012;53:582-6. [Google Scholar | PubMed]
- 9.Shin JH, Lee HK, Rhim SC, Park SH, Choi CG, Suh DC. Spinal epidural cavernous hemangioma: MR findings. J Comput Assist Tomogr 2001;25:257-61. [Google Scholar | PubMed]
- 10.Sharma MS, Borkar SA, Kumar A, Sharma MC, Sharma BS, Mahapatra AK. Thoracic extra-osseous, epidural, cavernous hemangioma: Case report and review of literature. J Neurosci Rural Pract 2013;4:309-12. [Google Scholar | PubMed]