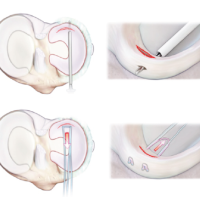
The use of intraoperative duplex ultrasound can reliably distinguish transient vascular compression or spasm from true popliteal artery injury during knee surgery, guiding timely and appropriate management.
Dr. Aidan A Jagasia, Department of Medical Education, Chicago Medical School at Rosalind Franklin University, BA, 3333 Green Bay Road, North Chicago, Illinois. E-mail: aidan.jagasia@med.usc.edu
Introduction: Vascular compromise during knee surgery is rare but can lead to serious limb-threatening consequences if not identified and managed promptly. Surgeons must rapidly determine whether loss of distal pulses represents reversible compromise, such as compression or spasm, or a true arterial injury requiring urgent repair. Duplex ultrasound provides real-time visualization of arterial anatomy and blood flow patterns, yet its use during orthopedic procedures has been described infrequently. This report presents two intraoperative vascular events during knee surgery that initially appeared similar but required very different management strategies. To our knowledge, this side-by-side comparison emphasizes an uncommon but critical diagnostic role for duplex ultrasound in orthopedic practice.
Case Report: The first patient was a 37-year-old man who developed a sudden loss of distal pulses during arthroscopic surgery for severe knee arthrofibrosis after prior ligament reconstruction. Duplex ultrasound showed preserved pulsatile flow in the popliteal artery despite absent signals distally. Further evaluation revealed an expanding hematoma near the posteromedial portal. After hematoma evacuation, distal pulses returned and the patient recovered without vascular intervention. The second patient was a 56-year-old man undergoing corrective osteotomy for longstanding knee instability. He experienced an abrupt loss of distal pulses accompanied by brisk bleeding during wound closure. Duplex ultrasound demonstrated the absence of flow in the popliteal artery. Surgical exploration revealed an intraluminal thrombus and a partial anterior wall laceration of the popliteal artery. Thrombectomy and primary repair restored distal perfusion, and the patient recovered uneventfully after vascular repair.
Conclusion: These two cases highlight the importance of distinguishing reversible vascular compromise from true arterial injury during knee surgery. Duplex ultrasound offered a rapid, non-invasive assessment of arterial patency and waveform patterns that helped guide decision-making when distal signals were lost. Incorporating duplex evaluation into the intraoperative workflow may reduce unnecessary exploration, ensure timely vascular consultation, and improve surgical safety. This comparison demonstrates an important and underreported diagnostic application of duplex ultrasound in orthopedic surgery and reinforces its potential value for surgeons managing complex posterior knee exposures or osteotomy procedures.
Keywords: Popliteal artery, duplex ultrasound, vascular injury.
Vascular injuries during total knee arthroplasty (TKA) occur at an incidence of 0.31% (31.42/10,000 TKAs) [1,2]. The reported incidence of vascular injury associated with osteotomies ranges from approximately 0.6–1.7%, with the largest clinical series and systematic reviews reporting rates near 1% [3,4,5,6,7,8,9,10]. Although rare, these complications can lead to limb-threatening consequences, emphasizing the need for timely diagnosis and intervention [11,12,13]. The popliteal artery is particularly vulnerable given its close proximity to the posterior capsule, especially during posterior capsular release or posterior instrumentation [14]. Mechanisms of injury include direct laceration, thrombosis from intimal disruption, kinking, extrinsic compression from hematoma, and transient vasospasm [15]. While true arterial injury requires urgent repair to prevent ischemia, transient compromise from spasm or compression often resolves without invasive intervention, making accurate intraoperative differentiation critical to avoid unnecessary morbidity [16]. Among vascular complications, transient vascular compromise from spasm or extrinsic compression may be confused with thrombosis or laceration. Early and accurate differentiation is essential, particularly in cases where intervention carries significant morbidity. Duplex ultrasound provides valuable information on both vessel patency and flow waveforms, aiding intraoperative assessment [17]. Here, we compare two distinct intraoperative vascular events: One due to presumed vascular compression or spasm successfully managed conservatively and one due to true arterial injury requiring repair. Intraoperative assessment of suspected vascular compromise often relies on palpation of distal pulses or use of a sterile handheld Doppler to detect flow signals [18]. However, these methods lack the ability to visualize vessel anatomy or assess waveform morphology, making it difficult to distinguish between reversible causes, such as vasospasm, and irreversible pathology, such as thrombosis or transection [16,19]. Duplex ultrasound offers both real-time vessel imaging and spectral waveform analysis, enabling the surgeon to assess patency, lumen diameter, and flow patterns (triphasic, biphasic, and monophasic) that correlate with severity and chronicity of arterial compromise [20]. Despite these advantages, reports describing intraoperative duplex use in orthopedic surgery remain scarce, and there is limited guidance on interpreting findings in the setting of acute vascular events [21]. This gap underscores the need for case-based descriptions to guide decision-making with duplex ultrasound intraoperatively.
Case 1: Transient Vascular Compression
A 37-year-old male with a history of right anterior cruciate ligament (ACL) reconstruction presented with severe loss of knee motion secondary to arthrofibrosis and a large cyclops lesion measuring over 3 cm anterior to the graft. Additional imaging findings included a full-thickness cartilage lesion of the medial femoral condyle and diffuse scar tissue in the suprapatellar pouch and posteromedial capsule. The patient underwent arthroscopic excision of the cyclops lesion, extensive anterior and posterior capsular releases, and a reverse notchplasty. A posteromedial portal was used to access the scarred posterior compartment. To help regain knee extension, the ACL graft was intentionally thinned to remove hypertrophic scar tissue (the cyclops lesion itself) off of its anterior surface, but the original native implanted graft was kept intact. Following capsular releases, a gentle manipulation under anesthesia achieved 140° of flexion and full extension. Throughout the case, the patient maintained intact palpable and Dopplerable distal pulses. However, near the end of the procedure, Dopplerable signals in both the dorsalis pedis and posterior tibial arteries became absent. Vascular surgery was consulted intraoperatively. Duplex ultrasound revealed a preserved pulsatile waveform in the popliteal artery despite absent signals distally (Fig. 1).
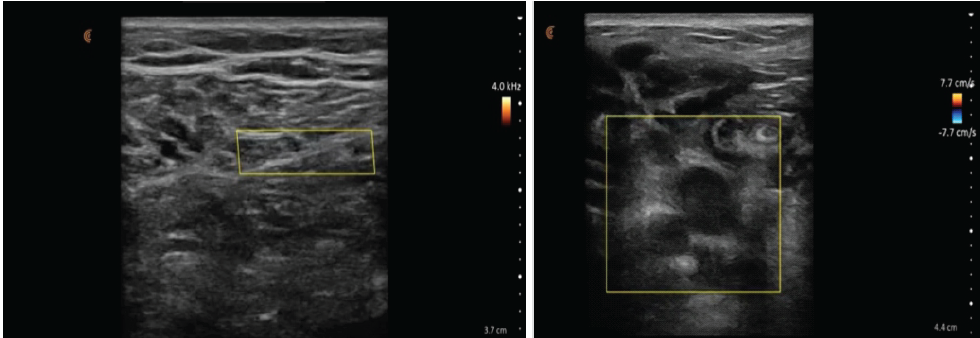
Figure 1: (a) Intraoperative duplex ultrasound demonstrating normal popliteal artery flow. Intraoperative duplex ultrasound image of the popliteal fossa obtained during routine vascular assessment. The popliteal artery is visualized centrally within the imaging field, with color Doppler demonstrating normal-velocity antegrade pulsatile flow. Spectral Doppler evaluation confirms preserved arterial waveform morphology without evidence of dampening or flow acceleration. No surrounding hypoechoic soft-tissue changes, extrinsic compression, or intraluminal echogenic material are identified, consistent with normal arterial patency and unobstructed distal perfusion. (b) Intraoperative duplex ultrasound showing abnormal popliteal artery flow. Intraoperative duplex ultrasound image of the popliteal fossa obtained during evaluation of suspected vascular compromise. The popliteal artery is visualized centrally within the imaging field, with the color Doppler scale demonstrating low-velocity antegrade flow. Despite the absence of palpable distal pulses during the procedure, the artery maintains preserved pulsatile luminal flow, indicating arterial patency. The surrounding hypoechoic region is consistent with local soft-tissue swelling and hematoma formation. This image was taken before hematoma evacuation in Case 1 and demonstrates that the diminished distal perfusion resulted from external compression rather than true arterial occlusion.
After further exploration, an expanding hematoma was observed near the posteromedial portal. Following evacuation of the hematoma and closure of the portal site, pulses returned and were confirmed both palpably and on Doppler (Fig. 2). The patient was admitted overnight for observation. Serial neurovascular examinations and duplex imaging showed no evidence of vascular injury or thrombosis. He was discharged the following day with an uneventful post-operative course.
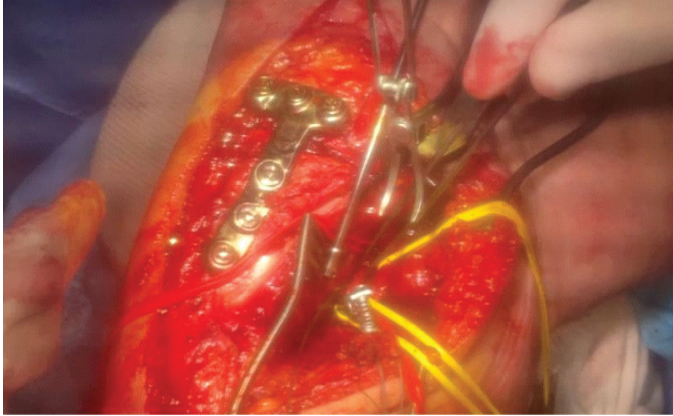
Figure 2: Intraoperative exposure during hematoma evacuation in Case 1. Intraoperative photograph of the posteromedial aspect of the knee during Case 1, taken immediately after surgical exposure for hematoma evacuation. Fixation hardware from the prior ligament reconstruction is visible along the medial femoral condyle. The organizing hematoma adjacent to the posteromedial portal exerted external compression on the popliteal artery, producing loss of distal Doppler signals before exploration. After evacuation of the hematoma, distal arterial pulses and Doppler signals were restored, confirming the absence of direct arterial injury. Vessel loops and retractors are shown maintaining exposure during decompression.
Although initially interpreted as arterial spasm, the clinical course suggests that external compression from the hematoma likely contributed to signal loss. The presence of preserved popliteal flow on duplex, followed by pulse restoration after hematoma evacuation, supports this interpretation (Fig. 2).
Case 2: Popliteal Artery Injury
A 56-year-old male with chronic right knee instability and a history of posterior cruciate ligament and posterolateral corner insufficiency presented with varus alignment and decreased posterior tibial slope. He underwent a staged reconstruction beginning with a high tibial osteotomy (HTO) using patient-specific instrumentation, along with a tibial tubercle osteotomy (TTO) to correct deformity and offload posterior and lateral structures to improve both sagittal and coronal knee joint stability.
The osteotomy was performed using a patient-specific instrumentation guide (Bodycad, Quebec City, Canada) with a planned posterior tibial slope increase and valgus-producing biplanar correction. Fixation was achieved with a customized HTO plate and compression screws. The TTO was repositioned and fixed using large-fragment screws. The tourniquet was released following fixation, and initially, there was no concern for bleeding. However, during closure, brisk dark bleeding was noted. Pulses were no longer palpable or Dopplerable distally. Duplex ultrasound demonstrated no flow in the popliteal artery, raising concern for thrombosis or laceration. Vascular surgery was emergently consulted. Exploration revealed an intra-arterial thrombus and partial laceration of the anterior wall of the popliteal artery, distal to the osteotomy site (Fig. 3). A thrombectomy and primary repair were performed by the vascular team (Fig. 4).
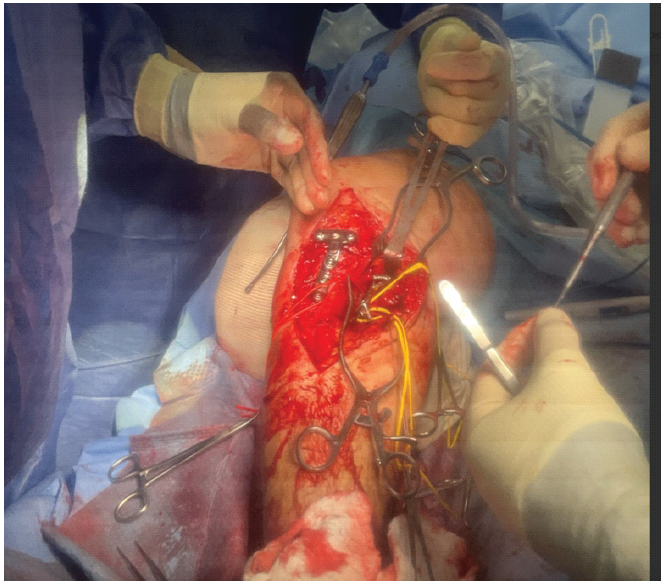
Figure 3: Intraoperative exposure during exploration for popliteal artery injury in Case 2. Intraoperative photograph from Case 2 showing the surgical field of the popliteal fossa during exploration for suspected arterial injury. Wide exposure was obtained to visualize the popliteal artery and adjacent neurovascular structures. The image demonstrates the acute operative setting before vascular repair, with retractors maintaining access to the deep posterior compartment. This step was performed following the loss of distal pulses and Doppler signals, raising concern for true arterial thrombosis rather than external compression. This figure illustrates the level of exposure required to assess the vessel fully.
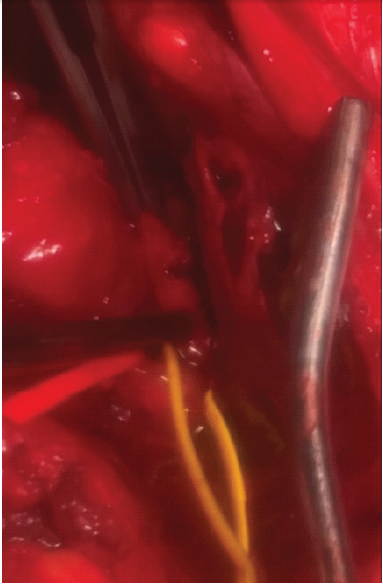
Figure 4: Intraoperative view following thrombectomy and primary repair in Case 2. Intraoperative close-up photograph demonstrating the repaired popliteal artery after thrombectomy and primary vascular repair performed in Case 2. The vessel is encircled with color-coded vessel loops to aid identification and maintain proximal and distal control. The lumen appears re-established, and arterial continuity is restored. The repair was performed after identification of a focal intraluminal thrombus causing complete loss of distal perfusion. This image was taken immediately following completion of the repair, before wound closure, confirming successful restoration of arterial patency.
Following repair, distal pulses returned and were confirmed on duplex imaging. The patient was placed on 325 mg aspirin for 2 weeks, followed by 81 mg for 10 additional weeks. He was monitored overnight with duplex ultrasound; the following morning, he remained stable and was discharged with intact neurovascular status. Notably, it was the presence of brisk bleeding during closure – rather than duplex findings alone – that prompted exploration. This case illustrates the importance of combining clinical signs with proper imaging in diagnosing arterial injury.
Vascular compromise during knee surgery, though rare, is a well-recognized source of morbidity and is associated with high rates of limb loss when diagnosis or intervention is delayed [22]. Mechanisms include direct arterial laceration, intimal injury with subsequent thrombosis, kinking or traction injury during manipulation, and extrinsic compression from retractors, implants, or hematoma [15]. Previous reports have emphasized that intimal injuries may initially appear deceptively benign yet progress to occlusion hours later, underscoring the importance of vigilant intraoperative monitoring [23]. Conversely, transient vasospasm – particularly in younger patients or after vessel manipulation – may resolve spontaneously or with conservative measures such as warming, topical vasodilators, or removal of external compression [24]. Differentiating these scenarios intraoperatively is therefore critical to prevent unnecessary exploration or, conversely, missed injuries that could lead to ischemia.
These two cases underscore the importance of distinguishing between transient vascular compression/spasm and true arterial injury during knee surgery. Both events presented as intraoperative loss of palpable and Dopplerable pulses, but management and outcomes diverged significantly. In the first case, duplex ultrasound revealed a preserved waveform in the popliteal artery along with absent distal pulses. In concordance with the duplex ultrasound readings and vascular surgery consultation, restoration of distal flow occurred after hematoma evacuation. This suggests that external compression was the likely cause of signal loss. In contrast, the second case demonstrated the absence of flow on duplex and clinical signs of arterial injury, necessitating emergent repair. The use of intraoperative duplex ultrasound in these cases provided a minimally invasive and immediate decisive advantage by allowing simultaneous visualization of the arterial lumen and assessment of spectral waveforms [21,25]. Triphasic or biphasic waveforms in the popliteal artery can help rule out complete occlusion, while monophasic or absent waveforms should raise suspicion for significant arterial injury [26]. However, accurate interpretation depends on operator expertise, familiarity with normal and pathological waveforms, and the ability to account for confounding factors such as hematoma-induced compression [27]. These cases illustrate how incorporating duplex assessment into the intraoperative algorithm can help stratify patients for immediate repair versus observation. Broader adoption may require targeted training for orthopedic and vascular teams, standardized protocols, and ready access to appropriate equipment in operating rooms where knee surgeries are performed. Establishing clear intraoperative algorithms that incorporate duplex findings into decision-making could help surgeons respond more decisively to vascular signal loss. By reinforcing the role of duplex ultrasound as both a diagnostic and triage tool, these measures have the potential to improve patient safety, reduce unnecessary interventions, and preserve limb function in high-stakes scenarios.
Intraoperative loss of vascular signal during knee surgery may represent transient vascular compromise due to compression or spasm or true arterial injury. These cases demonstrate how duplex ultrasound, when interpreted alongside clinical findings, can aid intraoperative decision-making. The intraoperative duplex ultrasound allows the surgical team access to immediate and non-invasively obtained critical information regarding vascular patency while avoiding the morbidity of unnecessary exploration. Accurate waveform characterization is ideal, and in ambiguous cases, early vascular consultation remains essential to optimize outcomes. Beyond preventing unwarranted vascular intervention, timely differentiation between reversible and irreversible causes of signal loss can shorten operative time, minimize patient morbidity, and preserve limb function. These cases underscore the importance of integrating duplex ultrasound into the intraoperative workflow for high-risk procedures, especially those involving posterior knee exposure or osteotomy fixation. Wider adoption of this technique, supported by training in vascular imaging interpretation, may enhance surgical safety and provide orthopedic teams with a reliable, real-time method for guiding management when vascular compromise is suspected.
When distal pulses are lost during knee surgery, differentiating transient vascular compromise from true arterial injury is critical. These cases demonstrate that intraoperative duplex ultrasound can provide immediate clarity on arterial patency and flow patterns, helping orthopedic surgeons avoid unnecessary exploration while ensuring timely vascular repair when needed.
References
- 1. Vaquero-Picado A, Rodríguez-Merchán EC. Arterial Injuries in Total Knee Arthroplasty. Cham: Springer; 2020. p. 131-4. [Google Scholar] [PubMed]
- 2. Butt U, Samuel R, Sahu A, Butt IS, Johnson DS, Turner PG. Arterial injury in total knee arthroplasty. J Arthroplasty 2010;25:1311-8. [Google Scholar] [PubMed]
- 3. Ferner F, Lutter C, Schubert I, Schenke M, Strecker W, Dickschas J. Perioperative complications in osteotomies around the knee: A study in 858 cases. Arch Orthop Trauma Surg 2022;142:769-75. [Google Scholar] [PubMed]
- 4. Schenke M, Dickschas J, Simon M, Strecker W. Corrective osteotomies of the lower limb show a low intra- and perioperative complication rate-an analysis of 1003 patients. Knee Surg Sports Traumatol Arthrosc 2018;26:1867-72. [Google Scholar] [PubMed]
- 5. Miltenberg B, Puzzitiello RN, Ruelos VC, Masood R, Pagani NR, Moverman MA, et al. Incidence of complications and revision surgery after high tibial osteotomy: A systematic review. Am J Sports Med 2024;52:258-68. [Google Scholar] [PubMed]
- 6. Woodacre T, Ricketts M, Evans JT, Pavlou G, Schranz P, Hockings M, et al. Complications associated with opening wedge high tibial osteotomy–a review of the literature and of 15 years of experience. Knee 2016;23:276-82. [Google Scholar] [PubMed]
- 7. Bisicchia S, Rosso F, Pizzimenti MA, Rungprai C, Goetz JE, Amendola A. Injury risk to extraosseous knee vasculature during osteotomies: A cadaveric study with CT and dissection analysis. Clin Orthop Relat Res 2015;473:1030-9. [Google Scholar] [PubMed]
- 8. Darnis A, Villa V, Debette C, Lustig S, Servien E, Neyret P. Vascular injuries during closing-wedge high tibial osteotomy: A cadaveric angiographic study. Orthop Traumatol Surg Res 2014;100:891-4. [Google Scholar] [PubMed]
- 9. Kim J, Allaire R, Harner CD. Vascular safety during high tibial osteotomy: A cadaveric angiographic study. Am J Sports Med 2010;38:810-5. [Google Scholar] [PubMed]
- 10. Armstrong PA, Powell A, Bandyk DF. Intraoperative Ultrasound Assessment of Carotid Endarterectomy and Stent-Angioplasty. London: Springer; 2013. p. 211-9. [Google Scholar] [PubMed]
- 11. Agarwala S, Menon A, Gupta M, Kulkarni A, Kapadia F, Padate B, et al. Multidimensional management of a vascular injury following total knee arthroplasty: A rare case report. J Clin Orthop Trauma 2019;10:991-4. [Google Scholar] [PubMed]
- 12. Bohlen HL, Gargiulo DJ, Thon SG. Occlusion outside the norm: A unique case of the pulseless, well-perfused hand following closed humeral shaft fracture. Orthopedics 2019;42:e279-81. [Google Scholar] [PubMed]
- 13. Hodgson H, Saghir N, Saghir R, Coughlin P, Scott D, Howard A. Arterial complications following total knee arthroplasty (TKA): A systematic review and proposal for improved monitoring. Malays Orthop J 2023;17:80-9. [Google Scholar] [PubMed]
- 14. Ninomiya JT, Dean JC, Goldberg VM. Injury to the popliteal artery and its anatomic location in total knee arthroplasty. J Arthroplasty 1999;14:803-9. [Google Scholar] [PubMed]
- 15. Imerci A, Ozaksar K, Gürbüz Y, Sügün TS, Canbek U, Savran A. Popliteal artery injury associated with blunt trauma to the knee without fracture or dislocation. West J Emerg Med 2014;15:145-8. [Google Scholar] [PubMed]
- 16. MacLean LD. The diagnosis and treatment of arterial injuries. Can Med Assoc J 1963;88:1091-101. [Google Scholar] [PubMed]
- 17. Tensho K, Kumaki D, Iwaasa T, Koyama S, Shimodaira H, Ikegami S, et al. Anatomical implications of the subvastus approach on major vascular injury during a distal femoral osteotomy: A computed tomographic venography study. Knee Surg Sports Traumatol Arthrosc 2023;31:1556-62. [Google Scholar] [PubMed]
- 18. Zierler RE. Doppler Techniques for Intraoperative Arterial Assessment. Dordrecht; Springer; 1989. p. 463-71. [Google Scholar] [PubMed]
- 19. Behrouz R. The rise and fall of transcranial doppler ultrasonography for the diagnosis of vasospasm in aneurysmal subarachnoid hemorrhage. J Neurosurg Anesthesiol 2019;31:79-80. [Google Scholar] [PubMed]
- 20. Armstrong PA, Carroll MI, Bandyk DF. Duplex Ultrasound Assessment of Lower Extremity Arterial Disease. Cham: Springer; 2017. p. 349-61. [Google Scholar] [PubMed]
- 21. Sommerset J, Condron M, Sea S, Teso D, Karmy-Jones R. Arterial duplex as an adjunct to assist decision making in the management of arterial trauma. J Vasc Ultrasound 2019;43:25-9. [Google Scholar] [PubMed]
- 22. McAuley CE, Steed DL, Webster MW. Arterial complications of total knee replacement. Arch Surg 1984;119:960-2. [Google Scholar] [PubMed]
- 23. Gaheer RS, Rysavy M, Al Khayarin MM, Kumar K. Femoral artery intimal injury following open reduction of an acetabular fracture. Orthopedics 2009;32:212. [Google Scholar] [PubMed]
- 24. Wöpking S, Kastrup A, Lentschig M, Brunner F. Recurrent strokes due to transient vasospasms of the extracranial internal carotid artery. Case Rep Neurol 2013;5:143-8. [Google Scholar] [PubMed]
- 25. Rodriguez-Luna D, Molina CA. Vascular imaging: Ultrasound. Handb Clin Neurol 2016;136:1055-64. [Google Scholar] [PubMed]
- 26. Tai N, Raj JP, Walsh M. Vascular trauma. Surgery (Oxford) 2004;22:288-93. [Google Scholar] [PubMed]
- 27. Morales H. Pitfalls in the imaging interpretation of intracranial hemorrhage. Semin Ultrasound CT MR 2018;39:457-68. [Google Scholar] [PubMed]