Although exceedingly rare, forefoot Charcot neuroarthropathy should be considered in the differential diagnosis of an unexpectedly inflamed foot following forefoot surgery. This may occur even in non-diabetic patients, particularly if they have risk factors such as myelopathy.
Dr. Karan Malhotra, Foot & Ankle Unit, Royal National Orthopaedic Hospital, Stanmore, United Kingdom; E-mail: k.malhotra@ucl.ac.uk
Introduction: Charcot neuroarthropathy (CN) commonly affects the foot in diabetic patients but can also occur in patients with other conditions, such as myelopathy. It is important to recognize as it can occur and progress rapidly and is associated with significant long-term morbidity. CN can affect the forefoot although this is much rarer, and it can occur after surgery. The combination of CN affecting the forefoot following forefoot surgery in a non-diabetic patient has never been described before. We present a case report of a patient to highlight the importance of including CN in the differential diagnosis of a post-operative inflamed foot in patients with myelopathy.
Case Report: A 51-year-old non-diabetic man sustained an intra-articular fracture to his second toe. It did not heal despite appropriate non-operative management over 3 months. He was discovered to have myelopathy arising from his cervical and thoracic spine. He underwent reconstructive forefoot surgery to address an arthritic bunion and the fracture but had a persistently inflamed foot post-operatively. Initially, this was suspected to be an infection, but the wounds healed well, and inflammatory markers were normal. The swelling was noted to improve on elevation and there was a significant side-to-side temperature variation. Radiographs revealed early failure of fixation and following subsequent magnetic resonance imaging scan, he was diagnosed with forefoot CN. He was treated in a total contact cast and rapidly improved. At 6 weeks, he was transitioned to a boot and underwent staged spinal surgery to address the myelopathy. By 5 months post-operatively, his foot had recovered and there was no longer a side-to-side temperature difference. By 10-month post-operatively, he was able to return to light jogging with a pain-free, well-corrected foot.
Conclusion: CN affecting the forefoot is rare, particularly in the setting of myelopathy in the non-diabetic patient. It can, however, be triggered by forefoot surgery. It is important to be aware of this potential diagnosis as if it is diagnosed and treated early, rapid resolution can occur, but if missed, it could potentially result in long-term complications.
Keywords: Charcot neuroarthropathy, myelopathy, forefoot.
Charcot neuroarthropathy (CN) can result in significant morbidity for patients, and it is important to recognize and treat it early. One of the earliest descriptions was in 1868 by Jean-Martin Charcot, in patients with myelopathy secondary to syphilis [1]. In 1936, it was described in patients with diabetes mellitus, and since then, most of the literature has focused on its classification and management in diabetic patients [2]. The incidence of CN in diabetic patients has been estimated at about 0.5%; however, in patients with diabetic neuropathy, the incidence may be as high as 35% [3]. Although CN can affect any part of the foot, the rarest location is the forefoot, with an estimated rate of about 2–8% of CN foot cases in diabetic patients [4,5]. Myelopathy and other abnormalities affecting the spinal cord such as syringomyelia and spina bifida have also been associated with CN affecting the foot and ankle [6,7,8]. However, the rate of forefoot CN in patients with neurological conditions such as myelopathy is not well described, and it is a very rare occurrence. The causes for CN are not always clear, but foot surgery has been described as a potential trigger for CN; however, the location in the foot is poorly reported, with most occurring in the midfoot [5,7,9,10]. We describe the case of a non-diabetic patient with myelopathy who developed forefoot CN following forefoot surgery. This presentation has never been described before in the literature and initially, it was assumed to be infection. This case highlights the diagnostic dilemmas, evolving clinical findings, rapid onset, and final relatively rapid resolution of symptoms once the CN was treated appropriately.
A 51-year-old male patient sustained a comminuted, intra-articular fracture of the proximal phalanx of his left second toe in March 2024. He sustained this while practicing martial arts. The mechanism was presumed axial loading, and he did not initially realize he had a fracture. He consequently continued running and martial arts for a further 3 weeks before seeking medical advice. His previous history was notable for cardiac arrhythmias, ulcerative colitis, and gout. He was a non-smoker with occasional alcohol intake. Before injury, he practiced martial arts twice a week and ran 2–3 times a week (up to 5 km). The fracture was initially managed non-operatively but failed to unite over 3 months. It was noted on clinical examination that he had a dorsally subluxed, shortened second toe, and a non-tender bunion which was under-riding the second toe. The hallux had a restricted range of movement (rigidus), and the second toe was not reducible. He was additionally noted to have significantly less pain than expected and reduced sensation to touch, vibration, temperature, and proprioception in his lower legs. He also exhibited Lhermitte’s sign. He was assessed by a neurologist and nerve conduction studies were performed. These revealed chronic denervation of the L5/S1 myotomes, reduced peroneal motor responses, and preserved sensory responses. The findings were in keeping with a radiculopathy or myelopathy rather than a peripheral neuropathy.
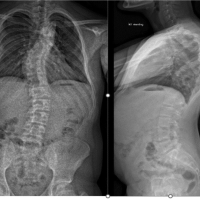
He had a computed tomography (CT) scan and magnetic resonance imaging (MRI) scan of his foot to characterize the fracture (Fig. 1) – this confirmed a comminuted fracture with joint effusion and reactive edema at the base of the second toe, and arthritis in his hallux metatarsophalangeal joint (MTPJ).

Figure 1: Pre-operative radiographs taken at 3 weeks post injury (a) Oblique, (b) Anterior-posterior view, and pre-operative computed tomography taken at 3-month post-injury (c) Axial view, (d) Sagittal view. The images demonstrate periosteal reaction without healing and comminuted, intra-articular fracture with depression of the articular surface of the proximal phalanx of the second toe.
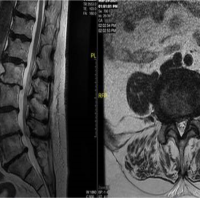
An MRI of his cervical and thoraco-lumbar spine demonstrated a T10/11-disc bulge with associated cord change suggesting myelopathy and severe multi-level cervical spine stenosis at C3/4, C4/5, and C5/6 with cord signal change suggestive of myelopathy (Fig. 2).

Figure 2: T2-weighted sagittal magnetic resonance imaging (MRI) of cervical spine (a) showing multi-level disc disease and subtle hyperintensity of the cord suggestive of myelopathy. T2-weighted sagittal MRI of the thoracic spine (b) showing significant impingement of the cord at T10/T11 with changes of myelopathy.
After a multidisciplinary discussion, the decision was to proceed with forefoot surgery followed by staged spinal surgery to address the cervical and thoracic myelopathy. In July 2024, he underwent a left hallux shortening chevron osteotomy (correcting the hallux valgus and decompressing the arthritic hallux MTPJ), an akin osteotomy, a second toe proximal interphalangeal joint (PIPJ) fusion, excision of bony fragments from the base of proximal phalanx and flexor tenodesis, a shortening Weil’s osteotomy of the second metatarsal (to decompress the joint), and a balancing percutaneous distal metaphyseal metatarsal osteotomy of the third metatarsal (Fig. 3).

Figure 3: (a) The pre-operative appearance of the foot. Anterior-posterior (b) and Lateral (c) intra-operative fluoroscopy images of the operative procedure.
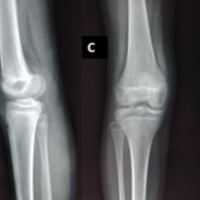
He had antibiotic prophylaxis pre-operatively and the procedure was carried out as planned. He was discharged in a bulky dressing the same day and allowed to mobilize fully weight-bearing in a flat post-operative shoe, as per our standard protocol for forefoot surgery. He was instructed to elevate for 23 h a day for the first 2 weeks. He was seen 2 weeks post-operatively for the reduction of dressings and wound inspection. The wounds were healing well, and swelling was as expected. He returned a week later for review with blistering and significantly increased swelling and erythema. No pus was noted; and the wounds were healing, however, he was commenced on flucloxacillin 500 mg 4 times a day as the initial suspicion was of a post-operative superficial infection. The wounds were re-dressed, with instructions for further elevation. At 4 weeks post-operatively, the wounds had completely healed but the swelling and erythema were yet to settle. The second toe was noted to be drifting into valgus. At 5 weeks, the redness and swelling were noted to be worse, with exacerbation on walking and improvement with elevation. The wire was removed from the second toe PIPJ in case there was a reaction to the metal. He was commenced on co-amoxiclav 500 mg 3 times a day as he was due for spinal surgery in a few weeks. He was noted to have developed an ulcer on his other foot under the third metatarsal head. Inflammatory markers and glucose were normal (white cell count 6.6 × 109/L, erythrocyte sedimentation rate 10 mm/h, C-reactive protein 1 mg/L, HbA1c 38 mmol/mol hemoglobin), but he was noted to have a mildly raised alkaline phosphatase (188 IU/L). At 6 weeks post-operatively, radiographs revealed a fracture of the dorsal first metatarsal shaft (at the site of osteotomy) with significant periosteal reaction and hypertrophy around all osteotomy sites, but with no evidence of bony healing. An MRI was then performed which demonstrated widespread edema and multiple joint effusions around the forefoot (extending beyond the surgical zone) with significant soft-tissue swelling, but no evidence of collection or infection (Fig. 4). There were therefore no targets for aspiration or sampling.

Figure 4: Imaging 6 weeks post-operatively. Radiographs (a) demonstrating failure of fixation of the hallux and excess bone formation and periosteal reaction without evidence of healing. T1-weighted axial (b), T1-weighted sagittal (c) and T2-weighted coronal (d) magnetic resonance imaging slices demonstrating significant and diffuse soft tissue swelling and joint effusions, without collection and edema of the second metatarsal shaft and loss of position. (e) demonstrates the clinical appearance and side-to-side difference at this stage.
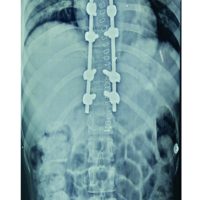
He was noted to have a side-to-side foot temperature difference of over 3°. Given the normal inflammatory markers, lack of any pus from the wounds, the improvements noted with elevation, and disorganization of joints in the absence of a collection, a diagnosis of post-operative forefoot CN was made, secondary to myelopathy and the trauma of surgery. Antibiotics were discontinued, his right-sided ulcer was dressed, and he was given a rocker shoe. He was placed into a total contact cast for his left foot. The toes of the left foot were strapped into the desired position using wound closure strips. He was commenced on low-molecular-weight heparin for thromboprophylaxis and kept non-weight-bearing. He was seen 5 days later, and the swelling, erythema, and temperature were all significantly improved. He was reviewed weekly for cast changes with continued steady and rapid improvement in symptoms. After 6 weeks in the total contact cast, the side-to-side temperature difference of his feet was less than half a degree, the alignment had significantly improved, and swelling had settled. A CT scan showed exuberant callus with bridging bone indicating healing. He was progressed into a VACOcast boot (Oped UK Ltd, Melksham, UK) and allowed to bear weight. The ulcer on the right foot had healed (September 2024). In October 2024, he had a C3-C6 anterior cervical discectomy/decompression and fusion. By November 2024, he was able to wear trainers (rocker shoes). By December 2024, his foot was pain-free and deformity-free with only a slightly discoloration on walking 3 km. Radiographs showed satisfactory consolidation and stable appearances (Fig. 5) and it was felt that he had entered the consolidation phase of the disease process. In January 2025, he had a posterior-instrumented decompression and fusion of T10–11.

Figure 5: Post-operative radiographs (a) anterior-posterior, (b) lateral and clinical photograph (c) at 6 months post-operatively after the episode of Charcot had entered the consolidation phase.
By May 2025 (10 months post-operatively), he was comfortably walking longer distances with no residual pain or deformity, his myelopathy was improving, and he was able to return to light (non-contact) martial arts training and light jogging over 100 m. By December 2025 (18 months post-operatively), he had returned comfortably to his pre-operative activities with no evidence of reactivation or further deformity.
In this case, the patient was eventually diagnosed with non-diabetic CN. The suspected pathology was loss of proprioception due to myelopathy, compounded by the trauma of a fracture and subsequent delayed ‘second hit’ of surgery. Although this is an accepted mechanism for the development of CN [11,12], forefoot surgery as a trigger in a non-diabetic patients has not been previously reported in the literature and there was a delay in diagnosis of about 3 weeks. The pathogenesis of CN is multi-factorial. The prevailing theories are the “neuro-vascular” and “neuro-traumatic” [2]. The neuro-vascular theory presupposes that the altered blood flow to the foot results in bone resorption and weakening. The neuro-traumatic theory suggests that loss of protective mechanisms results in repetitive micro-trauma which accumulates over time, resulting in disorganization of the joints and inflammation. Either way, in susceptible individuals (usually with neuropathy/myelopathy), an upregulated inflammatory response may be triggered which results in continual production of proinflammatory cytokines, which may further activate RANKL and osteoclasts, resulting in bony resorption [10]. The stages of CN are well described with the Eichenholtz classification being most commonly used (Table 1). The location in the foot that CN affects is also well described, with the midfoot being most commonly affected, and the forefoot being least commonly affected [4,5]. Trigger factors are commonly noted, with trauma being an established factor, and surgery also being a recognized trigger (although rarer at about 15% of cases), in keeping with the neuro-traumatic theory of CN [7,9,10].

Table 1: A table describing the Einchenholtz stages of Charcot neuroarthropathy, along with clinical signs, radiographic and MRI findings and suggested treatments for each stage
Our patient’s initial presentation of CN began at 3 weeks, once the elevation was reduced, and the patient increased their walking. Although this is a fairly typical time point for CN to develop post-injury [13], initially, this was not recognized as CN and a superficial infection was suspected and treated with antibiotics. This is often the case with unexpected CN cases [14], partly because post-surgical factors that can cause CN can also predispose to infection [15]. However, as time progressed, it was noted that the wounds continued to heal, the swelling was localized and responsive to elevation, inflammatory markers were normal, and there was abnormal failure of fixation. In the setting of infection, higher inflammatory markers, poor wound healing, pus and/or a collection on MRI would have been expected. A diagnosis of CN was therefore made. Diagnosis of the acute CN is often challenging and requires a high index of suspicion. Common differentials include infection and trauma [16]. Clinical signs include a warm, erythematous, swollen (inflamed) foot. However, pain may not be a significant feature due to altered sensation. There may also be a change of shape, which may be acute [17]. The inflammation may be fairly localized. There is often a side-to-side temperature difference between feet with the affected side being at least 2°C warmer [18]. A key clinical differentiator is the Charcot leg elevation test, wherein the inflammation may settle upon elevation of the affected leg for 10 min [19]. Imaging findings may guide diagnosis and staging as per Table 1 [20]. Bloods for inflammatory markers are useful to exclude infection, but infection may co-exist and be a trigger, so raised inflammatory markers do not exclude CN. Other specific cytokine markers (such as tumor necrosis factor -alpha) can also be used to assess for acute stages of CN, but these are not specific to CN and may also be raised with infection [21]. MRI scans are often employed in diagnosis, looking for collections and patterns of bone edema which can help differentiate infection and CN. However, in some cases, additional sequences such as diffusion weighted imaging and post-contrast enhanced scans may be required [22]. Ultimately, diagnosis is challenging and a combination of clinical, biochemical, and radiological investigations may be required for diagnosis. Unfortunately, the mean time to diagnosis CN after can be up to 3 months, particularly when other potential diagnoses co-exist [23]. It is therefore important that investigations be carried out expediently. Once diagnosed, treatment is aimed at reducing trauma and halting the immune cascade. It is important to institute treatment expediently as deformity can progress rapidly, causing significant bony destruction and increasing the risk of long-term complications/ulceration [23]. This is done by offloading in a total contact cast and restricting weight-bearing. Resolution in inflammation, and in particular swelling often occurs relatively quickly, which aids in diagnosis [24]. Immobilization should continue until the acute phase resolves, which can be identified when the side-to-side temperature difference becomes <0.5°C. However, there is no definitive consensus on the best ways in which to monitor resolution [2]. Once the coalescence stage occurs, more weight is permitted. This can be identified with repeat imaging which was a CT scan in this patient and demonstrated new callus and bone formation. In a forefoot case, a stiff-soled shoe or rocker shoe may be used to support through to the consolidation stage [25]. Fortunately, because our patient was picked up relatively early, once appropriate treatment was instituted, there was a rapid resolution of symptoms and no significant deformity arose. Subsequent spinal surgery treated the myelopathy and improved his sensation further. This may mitigate the risk of further recurrences, although there is no evidence to advise of the risk of recurrence in this situation, and it is not clear what role the spinal surgery played in the resolution of his CN after the initial acute phase. It is also unclear from the literature whether non-diabetics and forefoot CN cases respond faster than other cases, but in this case, the relatively rapid resolution of symptoms meant that after 6 months of symptom onset, the patient had completed his consolidation and could begin to return to normal activities. This is an isolated case report for a rare condition. Therefore, generalizability to varied patient populations is limited and as this is the first case report of this occurrence, it is not possible to make any comparison with the literature. As with all cases where fixation has failed, it is difficult to solely attribute the symptoms and presentation to a Charcot process, as there is some inevitable overlap with post-operative changes and primary failure of fixation. However, in this case, the pattern of fracture was highly unusual, and the edema and inflammation of joints extended beyond the area of fracture and beyond the surgical zone, reinforcing this as a Charcot process. Although our patient improved, the follow-up is still of relatively short duration, and we do not have validated outcome measures to report. This does make it difficult to quantify improvement over time. Ultimately, this case demonstrates an unusual manifestation and trigger for a rare but potentially devastating foot and ankle condition. The case and the challenges in diagnosis have been discussed to highlight the importance of recognition and vigilance. We have further demonstrated that by intervening early, the overall outcome of the patient was unaffected by the CN.
Forefoot CN in non-diabetic patients with myelopathy can be triggered by forefoot surgery. Although exceedingly rare, it is important to be aware of this potential diagnosis as having a high index of suspicion when faced with an inflamed post-operative foot will allow earlier diagnosis and appropriate treatment.
Forefoot CN should be suspected in patients with myelopathy following forefoot surgery in the presence of a persistently inflamed foot. Important differentials include infection and post-operative fracture, and a combination of history, clinical signs, and imaging may be required to make the diagnosis.
References
- 1. Charcot JM. [On some arthropathies that appear to depend on a lesion of the brain or spinal cord]. Arch Des Phys Norm et Pathol 1868;1:161. [Google Scholar] [PubMed]
- 2. Dardari D. An overview of Charcot’s neuroarthropathy. J Clin Transl Endocrinol 2020;22:100239. [Google Scholar] [PubMed]
- 3. Sinha S, Munichoodappa CS, Kozak GP. Neuro-arthropathy (Charcot joints) in diabetes mellitus (clinical study of 101 cases). Medicine (Baltimore) 1972;51:191-210. [Google Scholar] [PubMed]
- 4. Frygberg RG. The High Risk Foot in Diabetes Mellitus. London, England: Churchill Livingstone; 1991. [Google Scholar] [PubMed]
- 5. Schon LC, Easley ME, Weinfeld SB. Charcot neuroarthropathy of the foot and ankle. Clin Orthop Relat Res 1998;349:116-31. [Google Scholar] [PubMed]
- 6. Paliwal VK, Kumar A, Rahi SK, Pradhan S. Charcot foot in post-tubercular spinal arachnoiditis may indicate emerging dorsal cord syringomyelia. Neurol India 2011;59:299-301. [Google Scholar] [PubMed]
- 7. Shi X, Li P, Wang L, Zhang J. Multi-joint Charcot arthropathy caused by cervical spondylotic myelopathy and adult degenerative scoliosis with syringomyelia: A case report. Br J Neurosurg 2023;37:1843-9. [Google Scholar] [PubMed]
- 8. Nagarkatti DG, Banta JV, Thomson JD. Charcot arthropathy in spina bifida. J Pediatr Orthop 2000;20:82-7. [Google Scholar] [PubMed]
- 9. Aragon-Sanchez J, Lazaro-Martinez JL, Hernandez-Herrero MJ. Triggering mechanisms of neuroarthropathy following conservative surgery for osteomyelitis. Diabet Med 2010;27:844-7. [Google Scholar] [PubMed]
- 10. Kucera T, Shaikh HH, Sponer P. Charcot neuropathic arthropathy of the foot: A literature review and single-center experience. J Diabetes Res 2016;2016:3207043. [Google Scholar] [PubMed]
- 11. Kaynak G, Birsel O, Güven MF, Oğüt T. An overview of the Charcot foot pathophysiology. Diabet Foot Ankle 2013;4:10.3402/dfa.v4i0.21117. [Google Scholar] [PubMed] [CrossRef]
- 12. Papanas N, Maltezos E. Etiology, pathophysiology and classifications of the diabetic Charcot foot. Diabet Foot Ankle 2013;4:20872. [Google Scholar] [PubMed]
- 13. Harris A, Violand M. Charcot neuropathic osteoarthropathy. In: StatPearls. Treasure Island, FL: StatPearls Publishing; 2025. [Google Scholar] [PubMed]
- 14. Galeazzi Rech AL, Stüve Y, Toepfer A, Schimke KE. Missing the boat: Fatal ending to a missed case of Charcot arthropathy. Endocrinol Diabetes Metab Case Rep 2020;2020:20-0013. [Google Scholar] [PubMed]
- 15. Wukich DK, Crim BE, Frykberg RG, Rosario BL. Neuropathy and poorly controlled diabetes increase the rate of surgical site infection after foot and ankle surgery. J Bone Joint Surg Am 2014;96:832-9. [Google Scholar] [PubMed]
- 16. Carro GV, Ricci A, Torterola I, Saurral R, Portillo V, Amato PS, et al. Differential diagnosis of charcot neuroarthropathy in subacute and chronic phases: Unusual diseases. Int J Low Extrem Wounds 2024;23:181-90. [Google Scholar] [PubMed]
- 17. Vopat ML, Nentwig MJ, Chong AC, Agan JL, Shields NN, Yang SY. Initial diagnosis and management for acute Charcot neuroarthropathy. Kans J Med 2018;11:114-9. [Google Scholar] [PubMed]
- 18. Bramham R, Wraight P, May K. Management of Charcot neuroarthropathy. Diabet Foot J 2011;14:163-70. [Google Scholar] [PubMed]
- 19. Brodsky JW. Outpatient diagnosis and care of the diabetic foot. Instr Course Lect 1993;42:121-39. [Google Scholar] [PubMed]
- 20. Madan SS, Pai DR. Charcot neuroarthropathy of the foot and ankle. Orthop Surg 2013;5:86-93. [Google Scholar] [PubMed]
- 21. Bobircă A, Musetescu AE, Bordianu A, Pantea Stoian A, Salmen T, Marinescu DC, et al. Novel biomarkers predictive of diabetic Charcot foot-an overview of the literature. Life (Basel) 2022;12:1944. [Google Scholar] [PubMed]
- 22. Martín Noguerol T, Luna Alcalá A, Beltrán LS, Gómez Cabrera M, Broncano Cabrero J, Vilanova JC. Advanced MR imaging techniques for differentiation of neuropathic arthropathy and osteomyelitis in the diabetic foot. Radiographics 2017;37:1161-80. [Google Scholar] [PubMed]
- 23. Shazadeh Safavi K, Janney C, Shazadeh Safavi P, Kunzler D, Jupiter D, Panchbhavi V. Inappropriate antibiotic administration in the setting of Charcot arthropathy: A case series and literature review. Prim Care Diabetes 2022;16:202-6. [Google Scholar] [PubMed]
- 24. Armstrong DG, Todd WF, Lavery LA, Harkless LB, Bushman TR. The natural history of acute Charcot’s arthropathy in a diabetic foot specialty clinic. J Am Podiatr Med Assoc 1997;87:272-8. [Google Scholar] [PubMed]
- 25. Moonot P, Pawar P. Approach to Charcot neuroarthropathy of the great toe: A case report. Cureus 2024;16:e71368. [Google Scholar] [PubMed]