Primary intra-osseous malignant nerve sheath tumors are rare and aggressive tumors, the knowledge and early detection of which can ensure prompt treatment and optimal patient outcomes.
Dr. Purusharth Yagnik, Department of Orthopaedics, Central Institute of Orthopaedics, VMMC and Safdarjung Hospital, Delhi, 110024, India. E-mail: puruyagnik@hotmail.com
Introduction: Malignant peripheral nerve sheath tumors (MPNST) are rare, aggressive soft-tissue sarcomas arising from peripheral nerves, usually associated with neurofibromatosis Type-I. Intraosseous MPNSTs are exceedingly uncommon and are typically located in the maxillofacial region. There are only 45 documented cases of intraosseous MPNSTs outside the head and neck region, of which only two are of humeral origin and only one reported as pathological fracture.
Case Report: We present a rare case of a de novo MPNST of the humerus that presented with pathological fracture and radial nerve palsy. The patient was initially managed with intercalary wide resection, but developed recurrence within 3 months, which was subsequently managed by shoulder disarticulation. The patient remained asymptomatic till 12-month follow-up, when he was found to have lung metastasis and eventually succumbed to the disease.
Conclusion: This report adds to the handful of cases of primary MPNST of bony origin, the understanding of which would reduce the possibilities of delayed/missed diagnosis. These tumors are highly aggressive and require prompt detection and multidisciplinary treatment for improved patient outcomes.
Keywords: Malignant peripheral nerve sheath tumor, neurofibromatosis, neurofibrosarcoma, malignant schwannoma.
Malignant peripheral nerve sheath tumors (MPNSTs) are high-grade lesions arising from peripheral nerve sheaths, accounting for around 5% of all soft-tissue sarcomas [1]. They commonly affect the age group of 30–60 years old and present with localized pain, paresthesia, or loss of sensory/motor function over the distribution of the nerve [2,3]. Almost 50% of all cases are associated with Neurofibromatosis type-I [1]. Intraosseous occurrence of these tumors is uncommon and usually affects the bones of the head and neck (mandible, maxilla). Primary osseous MPNSTs outside the maxillofacial region are uncommon, with only 45 documented cases in the literature (with only two being of humeral origin and only one having a pathological fracture) [4]. We present a challenging case of pathological fracture of the humerus, with radial nerve palsy, which was diagnosed as primary MPNST of bone on further evaluation.
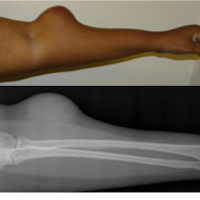
A 52-year-old male presented to the outpatient clinic with a history of pain and swelling in his right arm for 4 months, which increased following a history of a trivial trauma (fall from standing height 2 weeks before presentation). The pain was localized at the mid-portion of his right arm. It was insidious in onset, gradually progressive, dull aching in nature, and associated with night cries. Over the course of 4 months, the patient also noticed a diffuse swelling over his right mid-arm, which gradually increased in size along with the pain. He also noticed an increasing weakness in his ability to extend his fingers and wrist, loss of grip strength, and numbness over the dorsal aspect of his thumb, index, and middle fingers. There was no history of fever, loss of weight or appetite, skin changes over the swelling, or similar complaints elsewhere in the body. There were no systemic complaints, relevant history/co-morbidities, or history of substance use. There was an acute exacerbation of both pain and swelling two weeks before his presentation, following a ground-level fall. Prior to this, he was managed with painkillers from a local clinic (with increasing requirement of analgesic dose). However, they failed to provide relief following the trauma. The clinical examination revealed a single swelling of the middle third of his right arm, with overlying tenderness and ill-defined borders of approximately 10 × 6 cm size. The swelling had an irregular surface, variegated consistency, and was fixed to the underlying bone. The overlying skin was free without any secondary changes (such as dilated/engorged veins or ulcers). No evidence of regional lymphadenopathy was found. The examination also revealed motor weakness and sensory loss in the distribution of the radial nerve (loss of wrist and finger extension, loss of radial abduction of the thumb, and sensory loss over the radial two-thirds of the dorsum of the hand), suggestive of radial nerve palsy. No features suggestive of neurofibromatosis were found. An initial frontal radiograph of the right arm (Fig. 1) demonstrated a permeative lytic lesion of the mid-diaphysis of the humerus with a wide zone of transition, cortical breach, and a pathological fracture of the mid-shaft humerus, suggesting an aggressive bone lesion.

Figure 1: Frontal radiograph of the right arm demonstrates a permeative lytic lesion in mid-shaft humerus (arrow) with pathological fracture and cortical destruction.
The patient was subsequently evaluated with a magnetic resonance imaging (MRI) which showed a heterogeneous mass lesion of the mid-shaft humerus, hypointense on T1, hyperintense on T2 and short tau inversion recovery, of 11 × 6.2 × 5.6 cm, with involvement of both the anterior and posterior compartment soft tissue, and likely encasement of both the radial and ulnar nerves (Fig. 2).

Figure 2: T2-Sagittal (a), Short tau inversion recovery (STIR)- coronal (b), T1 axial (c) and STIR-axial (d) MR images of the right arm demonstrate a destruction of mid shaft humerus by a soft-tissue mass lesion (asterisk) isointense to muscle on T1 (c), hyperintense on T2 and STIR (a, b, d). The mass lesion has involved anterior and posterior compartments (c and d). Brachial artery is free from the mass (curved arrow).
Based on the clinical and radiological findings, the cause was thought to be a malignant lesion of the humerus, with differentials being a metastatic lesion, multiple myeloma, lymphoma, or a primary malignancy. Workup for the same included serum electrophoresis to identify an M-band (which was negative) and a complete blood workup (including serum alkaline phosphatase and serum lactate dehydrogenase) with a peripheral smear – all of which were normal. Fluorodeoxyglucose positron emission tomography (FDG-PET) computed tomography (CT) (Fig. 3) was done for systemic staging, which showed an increased FDG uptake in the middle 1/3rd of the humerus. There were no other lesions.

Figure 3: Fluorodeoxyglucose positron emission tomography computed tomography showing uptake in the middle 1/3rd of humerus.
After the staging workup, ultrasound-guided core needle biopsy was done, which was suggestive of an undifferentiated pleomorphic sarcoma. After informed consent, the patient was planned for wide intercalary resection and reconstruction with autologous non-vascularized fibula (Fig. 4). The patient had an uneventful post-operative period.

Figure 4: Immediate post-operative X-ray.
The histology of the resected specimen (Fig. 5) was suggestive of MPNST of bone. The tumor cells were positive for Vimentin and S-100 (focal) and negative for Pan CK, SMA, CD 10, Desmin, SOX 10, Myo D1, EMA, CD68, CD34, CD 117. The proximal, distal, and lateral margins were free from tumor; however, the medial margin was positive. The patient was advised adjuvant radiotherapy for the same, but was reluctant for further treatment.

Figure 5: Histopathology of resected specimen showing (a) Spindled tumor cells in fascicles and herringbone pattern under 10× magnification with H & E stains. (b) Atypical spindle cells showing moderate pleomorphism, irregular hyperchromatic nucleus and inconspicuous nucleoli under 20× magnification. (c) 30% tumor cells showing positivity for S100. (d) Tumor cells showing 70–90% positivity for Vimentin.
The patient returned at 3 months with recurrence of pain and swelling over the operated area of his right arm. The symptoms started insidiously 10 days before presentation, with no preceding trauma, and gradually increased in severity. Radiographs (Fig. 6) and MRI were both suggestive of tumor recurrence. An FDG-PET scan was done, which was also indicative of recurrence and revealed a sub-centimetric fibronodular lesion of the left lung. The case was discussed in tumor board, and the lung nodule was thought to be reactive.

Figure 6: X-Ray at the time of recurrence
The patient was given an option for a limb salvage procedure or a radical amputation of the right arm. The patient chose the latter as he did not want any possibility of local recurrence. After informed consent, right shoulder disarticulation was done. The resected specimen concurred with the previous histopathology report, and the post-operative period was uneventful. The patient remained asymptomatic and disease-free for 12 months, after which he developed episodes of cough with blood-tinged expectoration. The evaluation with contrast-enhanced CT revealed heterogenous enhancing lesions in the upper lobes of both lungs, as well as in the bronchi, suggestive of lung metastasis. The patient ultimately succumbed to the disease at two years after the index surgery.
MPNSTs (also known as neurofibrosarcoma or malignant schwannoma) account for around 5% of all soft-tissue sarcomas. In almost 50% of cases, they are associated with neurofibromatosis [1]. Sporadic MPNSTs are uncommon in themselves, and the primary intra-osseous origin of these tumors is even rarer [4,5]. Intra-osseous MPNSTs usually affect the bones of the head and neck (with the mandible being the most common.) Only 45 cases have been reported to occur in the spine or appendicular skeleton, with only two being reported in the humerus [4].


Table 1: Primary intra-osseous MPNST excluding facial bones reported in the literature [4]
These tumors are usually high grade, with no clear sex predilection, and occur between 30 and 60 years of age [1,5]. Our review of literature reveals a mean age at presentation to be 38 years (range 6–76 years), with no specific gender predisposition (21 males, 20 females). MPNSTs present with localized pain or swelling, or features of nerve involvement (paresthesia, motor weakness, sensory loss) [2]. Our patient presented with similar complaints that developed within a short period of four months and had radial nerve involvement. He also suffered a pathological fracture – which is a very rare presentation of these tumors and suggests a delay in the diagnosis in these cases. MRI has classically been the investigation of choice for soft-tissue sarcomas. Yu et al. described the MRI findings of MPNSTs as characteristically hypo-intense on T1-weighted and hyper-intense on T2-weighted images. The imaging also helps clear any suspicions of whether the tumor is of a primary bony origin or has come to involve bone secondarily, and also delineates local infiltration (nerve/vessel encasement) [7]. PET scans are used to rule out systemic metastasis [8]. Our case had similar MRI findings, but X-rays additionally served as an important imaging tool in view of the primary bony origin of the tumor and the associated pathological fracture. Final diagnosis can be made on the basis of immunohistochemistry, with MPNST being positive for S-100 and SOX – 10, with a loss of H3K27me3 expression [9,10]. In our case, tumor cells were positive for Vimentin and S-100 (Focal) and negative for Pan CK, SMA, CD 10, Desmin, SOX 10, Myo D1, EMA, CD68, CD34, and CD 117. The molecular tests were not available at our institute, and the patient was unable to afford it from a private laboratory. Hence, the case was discussed in a tumor board, which agreed to the diagnosis of MPNST of humerus. A wide surgical resection has been the primary line of management, with adjuvant radiotherapy playing a role in preventing relapses [11,12]. Recent literature also emphasizes the importance of chemotherapy, mainly a doxorubicin – ifosfamide based regime, although there is an ongoing debate with respect to its efficacy [13,14]. Le Guellec et al. found that lack of radiotherapy is an independent risk factor for recurrence [15]. This is also seen in our case, where adjuvant radiotherapy following wide resection was denied by the patient, with a subsequent recurrence at three months. Our review of literature also showed that MPNST is a high-grade sarcoma with increased risk of recurrence and metastasis. Eight patients (17.39%) had a local recurrence, of which five were observed to have metastatic lesions at the time of recurrence. Seventeen patients (36.95%) were diagnosed to have metastatic lesions, with 11 cases having a pulmonary metastasis, making lungs the most common site. 14 patients (30.43%) were confirmed to have died of disease. Our case also developed lung metastasis 12 months after the shoulder disarticulation and finally succumbed to the disease at two years of the index surgery.
The primary intraosseous malignant nerve sheath tumor of the humerus is an extremely rare entity. Any aggressive bony lesion with early clinical involvement of the surrounding nerve should raise clinical suspicion. Systematic workup, including IHC and molecular panel, will help in clinching the diagnosis. These high-grade, aggressive tumors are classically treated with wide resection and adjuvant radiotherapy, though recent literature has also shed light on the possible role of chemotherapy in their management. Regular follow-up is mandated in all cases due to the high risk of local recurrence and systemic metastasis.
A high index of suspicion is required for any tumor with rapid local involvement despite any classical systemic features of malignancy. These should undergo a thorough histo-radiological workup and be discussed in the tumor board to decide an appropriate line of management, which is usually multidisciplinary in nature.
References
- 1. Amirian ES, Goodman JC, New P, Scheurer ME. Pediatric and adult malignant peripheral nerve sheath tumors: An analysis of data from the surveillance, epidemiology, and end results program. J Neurooncol 2014;116:609-16. [Google Scholar] [PubMed]
- 2. Widemann BC. Current status of sporadic and neurofibromatosis type 1-associated malignant peripheral nerve sheath tumors. Curr Oncol Rep 2009;11:322-8. [Google Scholar] [PubMed]
- 3. Ferrari A, Bisogno G, Carli M. Management of childhood malignant peripheral nerve sheath tumor. Paediatr Drugs 2007;9:239-48. [Google Scholar] [PubMed]
- 4. Gambarotti M, Righi A, Sbaraglia M, Cocchi S, Benini S, Magagnoli G, et al. Primary malignant peripheral nerve sheath tumors of bone: A clinicopathologic reappraisal of 8 cases. Hum Pathol 2022;122:92-102. [Google Scholar] [PubMed]
- 5. Knight SW, Knight TE, Santiago T, Murphy AJ, Abdelhafeez AH. Malignant peripheral nerve sheath tumors-a comprehensive review of pathophysiology, diagnosis, and multidisciplinary management. Children (Basel) 2022;9:38. [Google Scholar] [PubMed]
- 6. Ortiz WJ, Salazar MS, Eager JJ, Sajid S, Cervantes M. Primary intraosseous malignant peripheral nerve sheath tumor of the humerus: Report of a rare case. Cureus 2022;14:e33178. [Google Scholar] [PubMed]
- 7. Yu YH, Wu JT, Ye J, Chen MX. Radiological findings of malignant peripheral nerve sheath tumor: Reports of six cases and review of literature. World J Surg Oncol 2016;14:142. [Google Scholar] [PubMed]
- 8. Geitenbeek RT, Martin E, Graven LH, Broen MP, Anten MH, van der Pol JA, et al. Diagnostic value of (18)F-FDG PET-CT in detecting malignant peripheral nerve sheath tumors among adult and pediatric neurofibromatosis type 1 patients. J Neurooncol 2022;156:559-67. [Google Scholar] [PubMed]
- 9. Prieto-Granada CN, Wiesner T, Messina JL, Jungbluth AA, Chi P, Antonescu CR. Loss of H3K27me3 expression is a highly sensitive marker for sporadic and radiation-induced MPNST. Am J Surg Pathol 2016;40:479-89. [Google Scholar] [PubMed]
- 10. Rodriguez FJ, Folpe AL, Giannini C, Perry A. Pathology of peripheral nerve sheath tumors: Diagnostic overview and update on selected diagnostic problems. Acta Neuropathol 2012;123:295-319. [Google Scholar] [PubMed]
- 11. Dunn GP, Spiliopoulos K, Plotkin SR, Hornicek FJ, Harmon DC, Delaney TF, et al. Role of resection of malignant peripheral nerve sheath tumors in patients with neurofibromatosis type 1. J Neurosurg 2013;118:142-8. [Google Scholar] [PubMed]
- 12. Kahn J, Gillespie A, Tsokos M, Ondos J, Dombi E, Camphausen K, et al. Radiation therapy in management of sporadic and neurofibromatosis type 1-associated malignant peripheral nerve sheath tumors. Front Oncol 2014;4:324. [Google Scholar] [PubMed]
- 13. Zehou O, Fabre E, Zelek L, Sbidian E, Ortonne N, Banu E, et al. Chemotherapy for the treatment of malignant peripheral nerve sheath tumors in neurofibromatosis 1: A 10-year institutional review. Orphanet J Rare Dis 2013;8:127. [Google Scholar] [PubMed]
- 14. Kolberg M, Høland M, Agesen TH, Brekke HR, Liestøl K, Hall KS, et al. Survival meta-analyses for >1800 malignant peripheral nerve sheath tumor patients with and without neurofibromatosis type 1. Neuro Oncol 2013;15:135-47. [Google Scholar] [PubMed]
- 15. Le Guellec S, Decouvelaere AV, Filleron T, Valo I, Charon-Barra C, Robin YM, et al. Malignant peripheral nerve sheath tumor is a challenging diagnosis: A systematic pathology review, immunohistochemistry, and molecular analysis in 160 patients from the french sarcoma group database. Am J Surg Pathol 2016;40:896-908. [Google Scholar] [PubMed]