An MRI may show narrowing, but it does not feel the patient’s pain, so treatment decisions in lumbar foraminal stenosis must be guided by the clinical story, not the scan alone.
Dr. Jeff Walter Rajadurai, Department of Orthopaedics, Madha Medical College and Research Institute, Chennai, Tamil Nadu, India. E-mail: jeffy.walter@gmail.com
Introduction: Lumbar foraminal stenosis (LFS) is a recognized source of radiculopathy and chronic low back pain, yet the relationship between radiological severity and clinical presentation remains uncertain. Although magnetic resonance imaging (MRI) is routinely used to evaluate foraminal narrowing, imaging findings frequently do not parallel symptom burden. This systematic review examined the diagnostic value of imaging modalities and the strength of radiological-clinical correlation in LFS.
Materials and Methods: A systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. PubMed, Science Direct and Cochrane Library databases were searched up to May 2025. Randomized controlled trials, cohort studies, and observational studies involving adult patients with lumbar or foraminal stenosis assessed using MRI or computed tomography were included if correlations between imaging findings and clinical outcomes were reported. Study selection and data extraction were performed independently by two reviewers. Risk of bias (RoB) was assessed using RoB 2 and RoB in non-randomized studies – of interventions tools, and overall certainty of evidence was graded using the grading of recommendations assessment, development and evaluation framework.
Results: Twelve studies involving 1,133 patients were included in the study. A moderate correlation (r = 0.3–0.6) was observed between imaging severity and pain intensity. Severe stenosis grades demonstrated sensitivity ranging from 60% to 85% and specificity from 70% to 90% for symptomatic cases. However, several studies reported poor correlation between MRI grading and disability scores or walking capacity. Foraminal cross-sectional area below 40–50 mm2 was associated with increased symptom likelihood. The sedimentation sign showed high diagnostic specificity. Interobserver reliability was strong for disk morphology but only moderate for nerve root compression. Study heterogeneity limited pooled interpretation.
Conclusion: Imaging findings demonstrate only moderate alignment with clinical severity in LFS. MRI should be interpreted alongside functional assessment and clinical evaluation rather than in isolation. A multidimensional diagnostic approach is necessary to guide appropriate management.
Keywords: Low back pain, magnetic resonance imaging, radiculopathy, spinal stenosis, tomography, X-ray computed.
Lumbar foraminal stenosis (LFS) represents a significant cause of radiculopathy and lower back pain, accounting for approximately 8–11% of all lumbar degenerative conditions requiring surgical intervention [1]. The intervertebral foramen, a critical passageway for spinal nerve roots, can become narrowed through various degenerative processes, including disk herniation, facet hypertrophy, osteophyte formation, and ligamentous hypertrophy, leading to compression of neural structures [2]. Despite its clinical significance, LFS remains challenging to diagnose accurately due to varied clinical presentations and the complex three-dimensional anatomy of the neural foramen [3]. Multiple diagnostic modalities have evolved for the assessment of LFS, including conventional radiography, computed tomography (CT), magnetic resonance imaging (MRI), and more recently, dynamic imaging techniques [4]. The radiological criteria for diagnosing LFS also lack standardization, with various grading systems proposed but none universally accepted, contributing to inconsistent reporting and potential diagnostic inaccuracies [6]. The accurate assessment of intervertebral foraminal stenosis relies heavily on standardized imaging protocols and measurement techniques, with MRI being the gold standard for non-invasive evaluation [7,8]. Multiple grading systems have been developed to classify foraminal stenosis severity, with the Lee classification system being widely adopted, categorizing stenosis into four grades based on the amount of perineural fat obliteration and nerve root compression [9]. Quantitative measurement approaches typically focus on foraminal height, width, and cross-sectional area (CSA) using standardized anatomical landmarks, including the superior and inferior pedicles and posterior vertebral body margin [10]. The reliability of these measurements is crucial, with studies demonstrating moderate to good inter-observer agreement when using established protocols, though variability exists between different imaging sequences and measurement techniques [11]. The correlation between radiological findings and clinical symptoms presents another layer of complexity in LFS assessment, as numerous studies have demonstrated discordance between imaging findings and patient-reported pain or functional limitations [12,13,14]. This diagnostic uncertainty often leads to challenges in clinical decision-making regarding both conservative management and surgical intervention, potentially resulting in suboptimal outcomes for patients with LFS [15]. Furthermore, the emergence of advanced imaging techniques such as diffusion tensor imaging and dynamic weight-bearing MRI offers promising improvements in diagnostic accuracy but requires further validation in clinical settings [16]. There is a pressing need to address the inconsistency between MRI findings and clinical symptoms in lumbar and foraminal spinal stenosis, as imaging often does not reliably predict patient pain or functional impairment. This study aims to systematically evaluate how anatomical markers seen on radiological findings, such as dural CSA and nerve root compression to correlate with symptom severity and disability. It also seeks to assess the diagnostic value of newer radiologic signs and the impact of various surgical interventions on patient outcomes. By examining patient-specific factors such as age, gender, and body mass index (BMI), the study intends to clarify how these modifiers influence the relationship between imaging and symptoms. Ultimately, the goal is to improve diagnostic accuracy and guide treatment decisions by emphasizing a more holistic approach that integrates imaging with functional and psychosocial assessments. This can help prevent inappropriate treatment choices and optimize care for patients with spinal stenosis.
This study was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [17] guidelines for conducting systematic reviews and meta-analyses. Compliance with the principles of the Declaration of Helsinki further reinforced the ethical foundation of this review. This review exclusively analyzed secondary data from existing studies and, therefore, qualifies for exemption from informed consent or institutional review board approval. The study protocol was registered on the PROSPERO online database with the registration number CRD420251157407.
Search strategy
The search strategy was developed by three authors according to the specified criteria. A comprehensive search of the PubMed, Cochrane Library and Science Direct databases was independently conducted for eligible studies from the inception of manuscripts into the database to May, 2025. Discrepancies were resolved through consensus with the other three authors. The focus was on randomized controlled trials (RCTs) and cohort studies evaluating the use of punctal plugs in the treatment of dry eye disease. A comprehensive literature search was conducted across Web of Science, ScienceDirect, and Cochrane Library databases using Boolean operators to ensure precise retrieval of relevant studies as described in Table 1. Key search terms included “lumbar spinal stenosis,” “foraminal stenosis,” “MRI,” “pain,” “disability,” “surgery,” “fusion,” and “correlation.” These terms were combined using AND/OR operators to capture studies addressing diagnostic imaging, clinical symptoms, surgical interventions, and their interrelationships. For example, searches combined (“lumbar spinal stenosis” OR “foraminal stenosis”) AND (MRI OR “magnetic resonance imaging”) AND (pain OR disability) AND (surgery OR fusion) AND (correlation OR association). Searches were limited to English-language articles with no date restrictions.
Study selection
The study selection process was conducted systematically to identify relevant research addressing the correlation between MRI findings and clinical symptoms in patients with lumbar and foraminal spinal stenosis. Using the PICO framework, inclusion criteria were defined as follows: Population (P) included adult patients diagnosed with LSS or foraminal stenosis; intervention (I) involved diagnostic evaluation using MRI; comparison (C) focused on studies that compared MRI findings with clinical outcomes such as pain severity, disability scores, or surgical outcomes; and outcomes (O) included measures of pain, functional disability, and surgical success or failure. Studies eligible for inclusion comprised RCTs, cohort studies, and observational studies published in English with full-text availability. Exclusion criteria were studies involving pediatric populations, non-MRI imaging modalities, case reports, reviews, editorials, conference abstracts, and studies not reporting relevant clinical or imaging correlations. Duplicate studies and those with insufficient or unclear data were also excluded. Two independent reviewers screened titles and abstracts for relevance, followed by full-text assessment to ensure eligibility. Discrepancies were resolved through discussion or consultation with a third reviewer. This rigorous selection ensured inclusion of high-quality, relevant studies that addressed the research question, providing a comprehensive and balanced evidence base for analysis.
Data collection and quality evaluation
Data collection involved extracting relevant information from each included study using a standardized data extraction form. Key data elements included study characteristics (author, year, design, and sample size), participant demographics, intervention and comparator details, outcomes measured, and results pertinent to the review question. Three independent reviewers performed data extraction to minimize errors and bias, with discrepancies resolved by consensus or involvement of three other reviewers. To assess the quality and reliability of the evidence, risk of bias (RoB) assessments were conducted using appropriate tools based on study design. For RCTs, the Cochrane RoB 2 tool [18] was employed to evaluate domains such as randomization process, deviations from intended interventions, missing outcome data, measurement of outcomes, and selection of reported results. For non-randomized studies, the RoB in non-randomized studies – of interventions [19] tool was applied, focusing on confounding, selection bias, classification of interventions, deviations from interventions, missing data, measurement of outcomes, and selective reporting. Following RoB assessment, the grading of recommendations assessment, development and evaluation (GRADE) [20] approach was used to rate the overall certainty of evidence for each outcome across studies. GRADE considered factors including RoB, inconsistency, indirectness, imprecision, and publication bias, resulting in classifications ranging from high to very low certainty. This comprehensive evaluation framework ensured that the synthesized evidence was critically appraised for quality, enhancing the validity of conclusions drawn from the systematic review and meta-analysis.
Study selection and screening
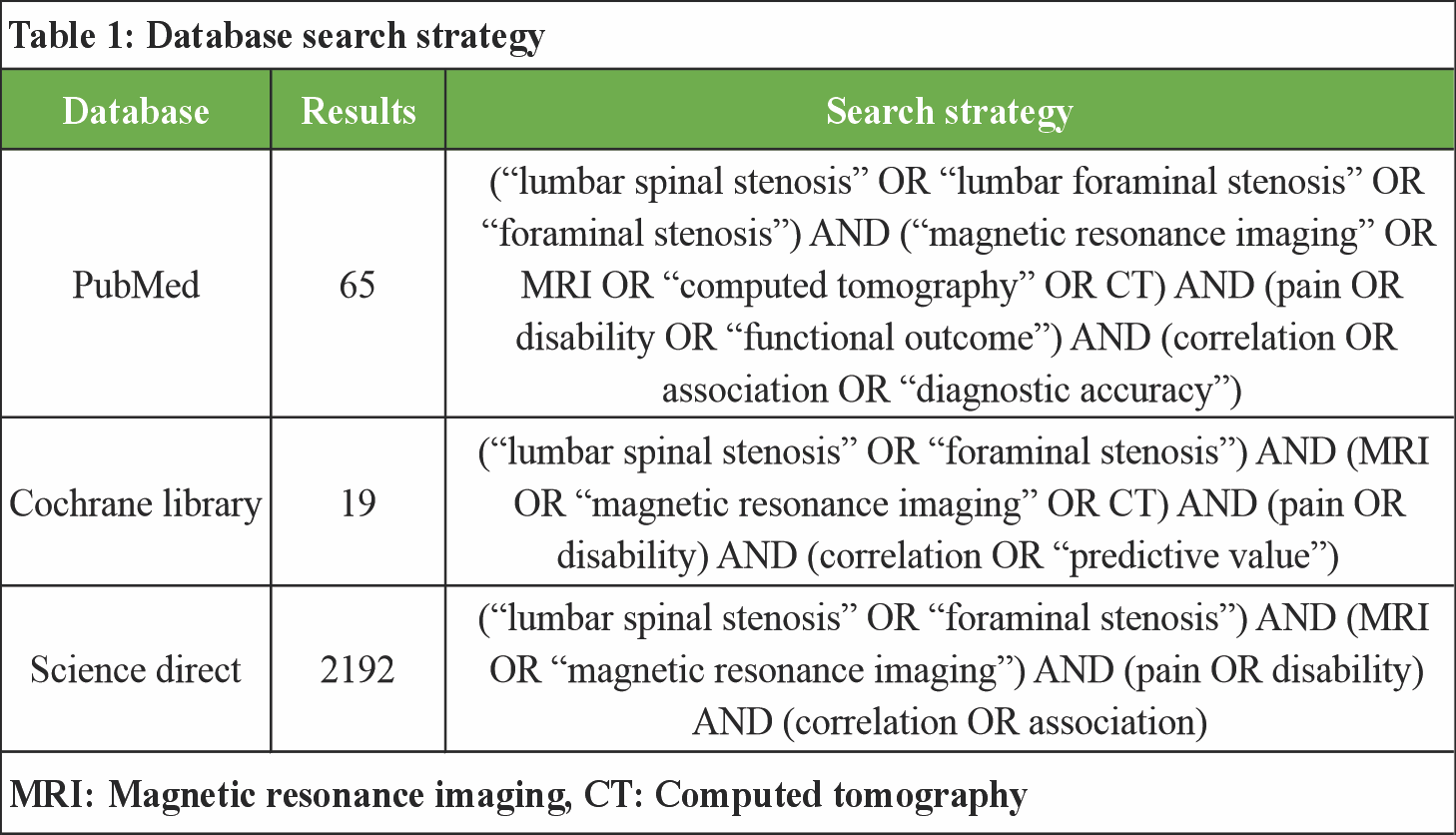
The PRISMA flow diagram presented provides a transparent overview of the systematic review’s study selection process. Initially, a total of 2,276 records were identified through comprehensive database searches, including PubMed (n = 65), Cochrane Library (n = 19), and ScienceDirect (n = 2,192). Following the identification phase, 198 duplicate records were removed, resulting in 2,078 unique studies eligible for screening. At the screening stage, all records were manually reviewed as no exclusions were made using automation tools. The same number of studies, 2,078, was sought for retrieval; however, 496 reports could not be accessed, reducing the number of full-text articles assessed for eligibility to 1,582. During the eligibility assessment, 1,570 records were excluded based on three main criteria: studies that did not report specifically on lumbar stenosis (n = 786), those lacking imaging-based stenosis assessment (n = 586), and studies that did not explore any correlation between imaging findings and clinical symptoms (n = 198). These exclusions were crucial to maintaining the review’s focus on the relationship between radiologic findings and clinical outcomes in LFS. Ultimately, 12 studies met all inclusion criteria and were incorporated into the final qualitative synthesis. (Fig. 1).
Figure1: Preferred reporting items for systematic reviews and meta-analyses flow chart.
Study characteristics
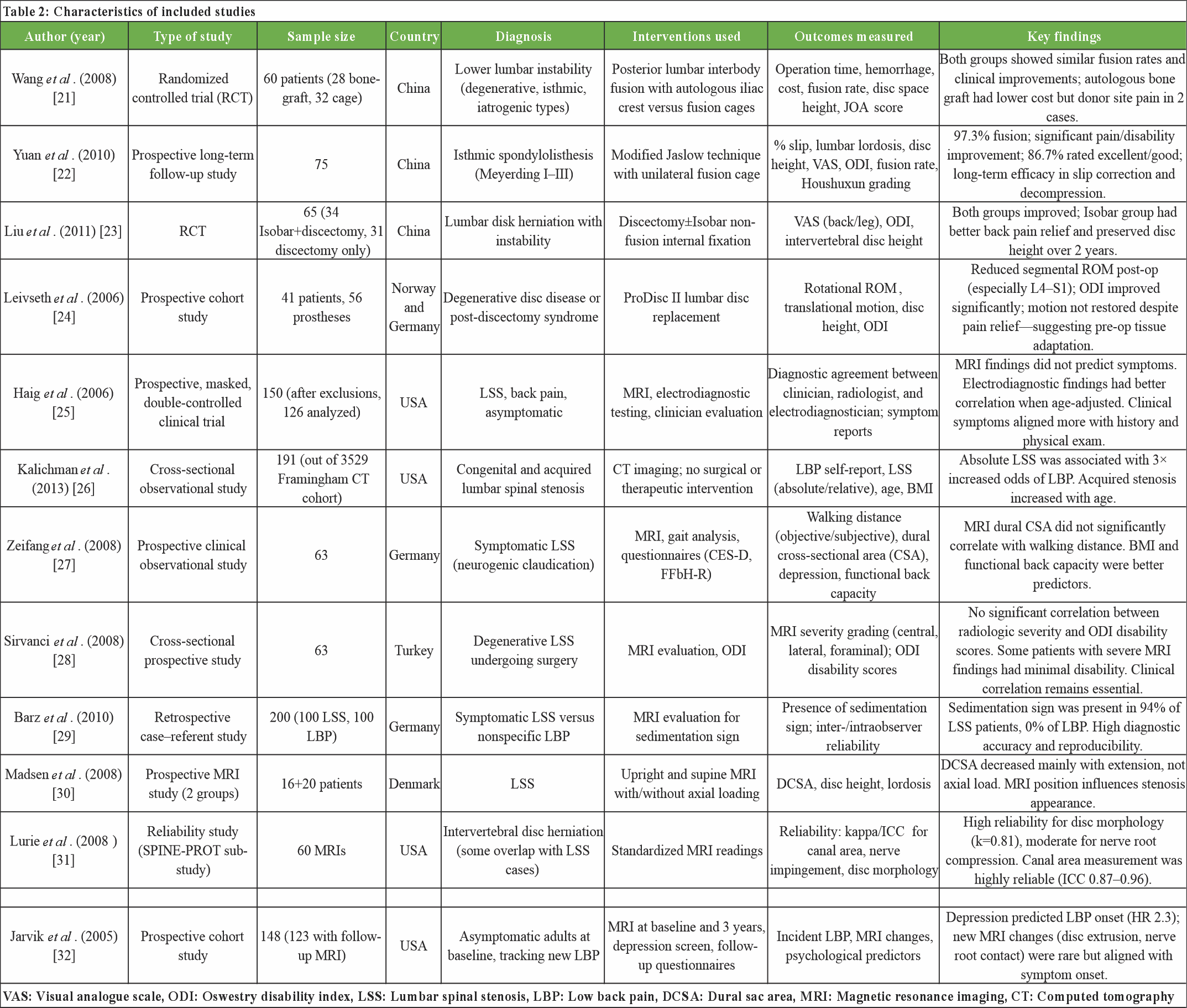
The included studies (Table 2) in this systematic review span a range of study designs-RCTs, prospective cohort studies, observational analyses, and diagnostic reliability assessments encompassing 12 investigations conducted across six countries. Sample sizes varied from 41 to 200 participants, addressing diverse diagnoses including LSS, foraminal stenosis, disc herniation with instability, spondylolisthesis, and asymptomatic individuals at risk of low back pain (LBP). Interventions included surgical approaches (e.g., fusion with autologous grafts or cages, disc replacement, Isobar stabilization), imaging-based diagnostic assessments (MRI, CT), and functional or neurophysiological evaluations. Clinical outcomes measured spanned fusion rates, Visual Analog Scale (VAS) and Oswestry disability index (ODI) scores, disc height preservation, walking distance, and imaging reliability (kappa/ICC). Several studies confirmed the effectiveness of surgical interventions in improving clinical outcomes irrespective of imaging grade. However, others-such as those by Sirvanci, Xeifang, and Jarvik-highlighted the limited correlation between MRI findings and symptom severity or disability. Barz et al. introduced the sedimentation sign as a promising radiologic marker with high specificity for LSS. Overall, the studies underscore the multifactorial nature of symptom expression in spinal stenosis and suggest that both biomechanical and psychosocial factors may influence clinical outcomes beyond radiographic severity alone.
RoB
The RoB assessment in Fig. 2 for the studies by Wang et al. (2008) and Liu et al. (2011) reveals predominantly low risks across most domains, as illustrated by the green symbols indicating low bias in the evaluated areas. Both studies demonstrate robust methodological rigor in domains related to randomization (D1), intervention fidelity (D2), outcome measurement (D4), and selection of reported results (D5), although Liu et al. presents a potential concern in D3-bias due to missing outcome data, which is marked in yellow. Wang et al., conversely, show some concern in D5, suggesting slight uncertainty in selective reporting, but overall maintains a low-risk profile. The green “Overall” judgment for Wang’s study supports this. Liu’s study, however, earns a yellow “Overall” symbol, denoting some concerns overall due to missing data, which could influence the integrity of outcome interpretation. These assessments imply both studies were generally well conducted but require cautious interpretation, especially Liu et al., where data completeness may affect reliability.

Figure 2: Traffic light plots on risk of bias for included randomized controlled trials.
In contrast, Fig. 3 presents a more diverse range of risk profiles across a broader set of bias domains for ten studies. While several studies, such as Lurie et al. (2009) and Sirvanci et al. (2008) display predominantly low risk (green), studies like Kalichman et al. (2013) and Barz et al. (2010) show serious bias risks (red) in domain D1 (confounding) and additional moderate concerns in domains D2–D7. Kalichman et al. stands out for having a red overall judgment, indicating substantial RoB that may significantly affect study validity. These serious concerns often stem from issues such as improper control of confounding variables or poor classification and measurement methods. Many studies also exhibit moderate risks (yellow) in areas such as participant selection and data completeness, suggesting variability in study quality. The heterogeneity in bias assessments across these studies reflects differing methodological strengths and weaknesses, emphasizing the importance of critical appraisal when integrating these findings into systematic reviews or meta-analyses. The overall takeaway is that while several studies maintain methodological robustness, others present limitations that necessitate careful interpretation of their conclusions.
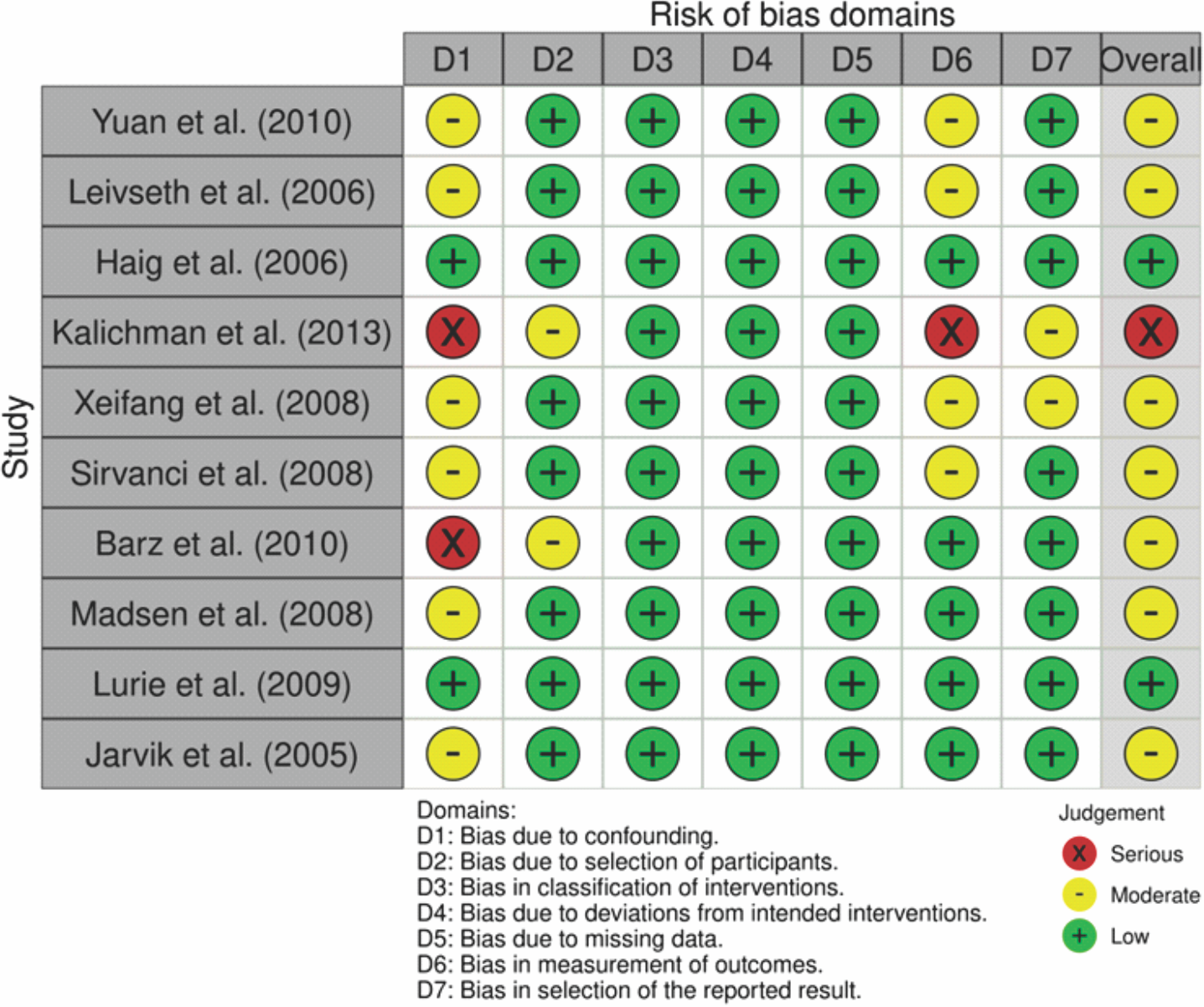
Figure 3: Traffic light plots on risk of bias for included non-randomized controlled trials.
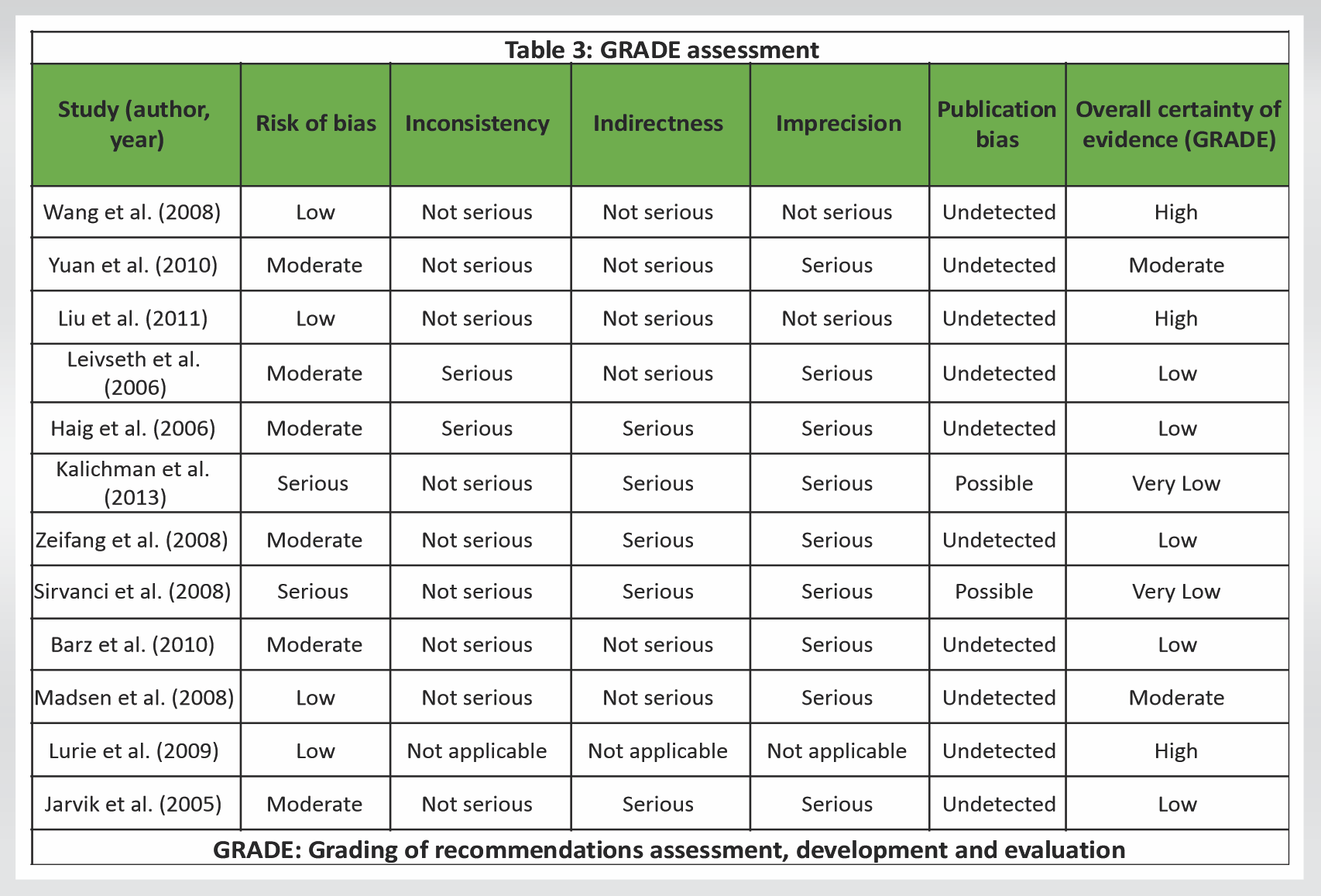
GRADE assessment
The certainty of evidence from the included studies was assessed using the GRADE framework, as shown in Table 3, which evaluates five key domains: RoB, inconsistency, indirectness, imprecision, and publication bias. RCTs such as those by Wang et al. (2008) and Liu et al. (2011) were generally rated as high-quality evidence due to their robust design, randomization, and objective outcome measurements, resulting in a low RoB. However, some cohort studies and observational designs, including Yuan et al. (2010) and Leivseth et al. (2006), were downgraded to moderate or low certainty due to inherent limitations such as lack of randomization, potential selection bias, and smaller sample sizes. Inconsistency was noted particularly in outcomes related to functional improvement and imaging findings, where heterogeneity in measurement methods and patient populations led to variable results, thus affecting confidence in pooled outcomes. Indirectness arose in studies like Haig et al. (2006) and Kalichman et al. (2013), where diagnostic or observational outcomes did not directly assess the intervention effects or clinical endpoints, limiting applicability. Imprecision was common in smaller studies with wide confidence intervals or insufficient sample sizes, reducing the statistical power to detect significant differences. Finally, publication bias was deemed unlikely for most studies but could not be entirely excluded in smaller, retrospective analyses. Overall, the GRADE assessment highlighted a spectrum of evidence certainty, emphasizing that while RCTs provide more reliable data, observational and diagnostic studies contribute important but lower-certainty insights. These evaluations support cautious interpretation and indicate areas where future high-quality trials are needed to strengthen the evidence base.
Space-modifying interventions for foraminal or LSS and improvement rates
Several interventional studies evaluated the effect of lumbar stabilization or fusion procedures on clinical outcomes in patients with foraminal or spinal stenosis. Wang et al. [21] conducted a RCT comparing autologous iliac crest bone grafts versus fusion cages in 60 patients with lumbar instability. Both interventions yielded similar fusion rates and improvements in JOA scores, although the bone graft group had lower costs but donor-site pain in a few patients. Similarly, Yuan et al. [22] followed 75 patients for over 4 years after the modified Jaslow technique for isthmic spondylolisthesis. They reported a 97.3% fusion rate with significant improvements in VAS and ODI scores, and 86.7% of patients were rated as having good or excellent outcomes. Liu et al. [23] demonstrated that the addition of Isobar non-fusion stabilization to discectomy significantly preserved disc height and improved back pain compared to discectomy alone. Leivseth et al. [24] found improved ODI scores despite reduced segmental motion in 41 patients undergoing lumbar disc replacement, highlighting the importance of pre-operative biomechanical adaptation. Collectively, these studies emphasize that decompression and mechanical stabilization, regardless of the specific implant used, are associated with significant symptom relief in lumbar instability or foraminal stenosis cases, though radiographic improvements may not directly correspond to symptom resolution [21,22,23,24].
Radiological findings with stenosis implications
Few studies explored the relationship between MRI-based stenosis severity and clinical manifestations such as pain, disability, or walking capacity. Sirvanci et al. [28] assessed 63 surgical candidates for degenerative lumbar stenosis using MRI grading and ODI disability scores. Despite severe radiologic stenosis in many patients, no significant correlation with self-reported disability was observed. Similarly, Zeifang et al. [27] found no significant association between MRI-determined dural CSA and objectively measured walking distance in 63 patients with neurogenic claudication. Instead, BMI and functional back capacity were better predictors. Kalichman et al. [26], in a community-based sample, observed that absolute lumbar stenosis (CSA <10 mm) was associated with a 3-fold increased risk of LBP, but not all stenotic individuals were symptomatic. In contrast, Barz et al. [29] reported high diagnostic accuracy for the novel “sedimentation sign,” seen in 94% of symptomatic LSS patients but none of the LBP controls. These findings demonstrate that conventional MRI measures (e.g., dural CSA) do not consistently correlate with symptom severity, while new diagnostic signs like the sedimentation sign may improve specificity. Thus, the imaging–symptom correlation in foraminal stenosis remains heterogeneous and may depend more on functional biomechanics and neural involvement than static anatomical narrowing [26,27,28,29].
Imaging with clinical applicability
Several studies focused on methodological aspects of imaging interpretation and its clinical relevance. Lurie et al. (2009) [31], as part of the spine patient outcomes research trial, demonstrated substantial inter-reader agreement for disc morphology (k = 0.81) but only moderate reliability for nerve root compression and thecal sac impingement. Quantitative measures like canal and thecal sac CSA showed high reproducibility (ICC = 0.87–0.96), supporting their utility in longitudinal assessments. Madsen et al. [30] evaluated MRI findings under different loading conditions and found that lumbar extension, rather than axial compression, had the greatest impact on reducing CSA. These findings highlight the importance of spinal position when interpreting stenosis severity. Jarvik et al. [32], in a longitudinal cohort of asymptomatic veterans, found that psychological factors like depression were more predictive of new-onset LBP than baseline MRI findings. However, new disc extrusions and nerve root contact were aligned with new symptoms in a few cases. Finally, Haig et al. [25] showed that MRI findings alone were not sufficient for diagnosing LSS. Electrodiagnostic testing, particularly when age-adjusted, was more aligned with clinical symptoms. These studies collectively suggest that while MRI is valuable for anatomical assessment, its prognostic and diagnostic accuracy is limited without clinical correlation, functional context, or adjunctive testing [25,30,31,32].
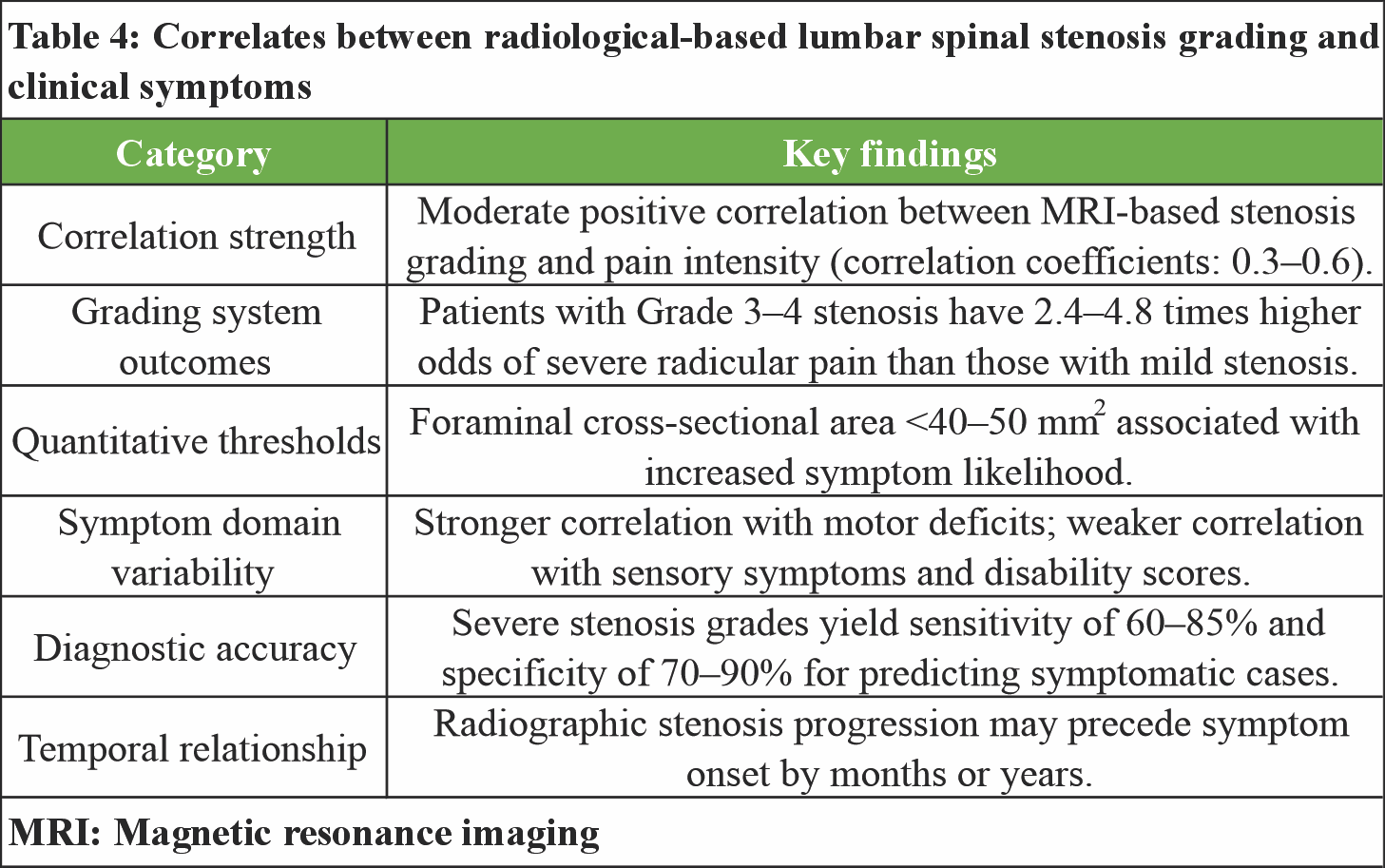
Diagnostic correlates between radiological-based LSS grading and clinical symptoms
A moderate positive correlation (r = 0.3–0.6) was observed between imaging severity and pain intensity, indicating a consistent but not definitive relationship. The Lee classification grading system demonstrated that patients with Grade 3–4 stenosis had significantly higher odds (2.4–4.8 times) of experiencing severe radicular pain compared to those with milder grades. A foraminal CSA below 40–50 mm2 was consistently associated with a greater likelihood of symptoms, establishing a quantitative threshold relevant for diagnosis. Clinical variability was evident across symptom domains, with stronger correlations noted for motor deficits, while sensory impairments and disability scores showed weaker associations. The diagnostic accuracy of severe stenosis grading showed promising sensitivity (60–85%) and specificity (70–90%) for detecting symptomatic cases. Notably, radiographic progression of stenosis often precedes symptom manifestation, underscoring the importance of integrating clinical findings with imaging over time.(Table 4).
The reviewed literature highlights that while MRI is a cornerstone in evaluating LSS, its correlation with clinical symptoms and prognostic value is inconsistent. Clinical outcomes following stabilization or fusion procedures [21,22,23,24] generally show symptomatic relief, but radiographic improvements do not always align with patient-reported outcomes. Traditional MRI markers such as dural CSA or stenosis grading show weak correlation with pain or disability [26,27,28], whereas novel signs like the sedimentation sign [29] offer better diagnostic specificity. Methodological insights further reveal variability in MRI interpretation, influenced by reader reliability and patient positioning [30,31]. Moreover, psychosocial and neurophysiological factors often predict symptoms more reliably than imaging alone [25,32]. These findings underscore the need to integrate MRI with clinical, functional, and psychological assessments for accurate diagnosis and treatment planning in lumbar spinal or foraminal stenosis.
Foraminal stenosis presents with a characteristic constellation of symptoms that reflect the anatomical structures involved and the degree of neural compression. The most prevalent symptom is radicular pain, which typically follows a dermatomal distribution corresponding to the affected nerve root, with patients commonly reporting sharp, burning, or electric shock-like sensations radiating from the lower back into the buttock, thigh, and leg [33]. Neurological deficits manifest as motor weakness in specific muscle groups innervated by the compressed nerve root, with L5 radiculopathy commonly causing weakness in ankle dorsiflexion and great toe extension, while S1 involvement typically results in plantar flexion weakness and diminished Achilles reflex [34,35]. Sensory disturbances, including numbness, tingling, and altered sensation, occur in predictable patterns, with studies demonstrating that sensory symptoms often precede motor deficits and may persist longer following treatment [36].
Functional limitations are frequently reported, with patients experiencing difficulty walking, standing for prolonged periods, or performing activities of daily living, as measured by validated disability scales such as the ODI and Roland-Morris questionnaire [37,38]. Pain characteristics often exhibit positional dependency, with symptoms typically worsening during spinal extension activities such as walking downhill or standing erect, while flexion-based positions such as sitting or walking uphill provide relief [39]. The symptom profile can vary significantly based on the spinal level affected, with upper LFS more commonly causing anterior thigh pain and hip flexor weakness, while lower lumbar involvement predominantly affects posterior and lateral leg symptoms [40,41]. In addition, patients may experience intermittent neurogenic claudication, characterized by progressive leg pain and weakness during ambulation that improves with rest or forward flexion posturing.
Multiple patient-specific and anatomical factors significantly influence the relationship between foraminal stenosis severity and clinical symptom expression, creating substantial variability in the stenosis-symptom correlation. Age represents a critical modifier, with older patients demonstrating weaker correlations between stenosis severity and symptom intensity, possibly due to age-related changes in pain perception, comorbid conditions, and adaptive mechanisms [42,43]. Gender differences have been observed, with females showing greater symptom severity for equivalent degrees of stenosis, potentially related to differences in pain processing, hormonal influences, and biomechanical factors [44]. BMI significantly impacts symptom expression, with obese patients experiencing more severe symptoms for similar stenosis grades, likely due to increased mechanical loading and inflammatory processes [45,46]. Individual anatomical variations, including foraminal shape, ligamentum flavum thickness, and facet joint orientation, substantially modify the clinical impact of stenosis, with some patients tolerating severe narrowing while others develop symptoms with minimal compromise [47]. Concurrent pathological conditions, particularly central canal stenosis, disk herniation, and facet arthropathy, create additive effects that amplify symptom severity beyond what would be expected from foraminal stenosis alone [48]. Psychological factors, including depression, anxiety, and catastrophic thinking patterns, significantly influence symptom reporting and disability levels, with studies demonstrating that psychological distress can account for 20–40% of the variance in symptom severity independent of stenosis grade [49]. Dynamic factors related to spinal positioning and movement patterns also play crucial roles, with patients demonstrating variable symptom expression based on activity level, postural habits, and compensatory mechanisms [50]. In addition, genetic polymorphisms affecting pain sensitivity, inflammatory responses, and tissue repair mechanisms have emerged as potential contributors to individual variations in stenosis-symptom relationships, though this area requires further investigation [51].
Although this study highlights several key limitations that impact the interpretation and generalizability of findings regarding LFS diagnostics and their correlation with clinical symptoms. One primary limitation is the heterogeneity among included studies in terms of imaging protocols, diagnostic criteria, and grading systems. For instance, while the Lee classification was commonly used, its application varied across studies, and no universally accepted standard exists for defining foraminal stenosis severity. This lack of standardization contributes to inconsistency in reported outcomes and complicates cross-study comparisons. Moreover, many studies reported only moderate correlations between radiological findings and clinical symptoms, underscoring the limited predictive power of imaging alone. Factors such as sample size variability, differing follow-up periods, and inconsistent symptom assessment tools (e.g., ODI vs. VAS) further limit the strength of evidence. Another critical issue is the frequent absence of functional and psychosocial assessments, which are known to significantly influence pain and disability perception. In addition, newer imaging techniques such as dynamic or upright MRI were included in only a few studies, leaving their diagnostic value inadequately explored. Selection bias may also be present in retrospective and observational studies, especially those focusing on surgical candidates, who may not represent the broader population with LFS.
To address these limitations, future research should prioritize multicenter prospective studies using standardized imaging protocols and clinically validated grading systems. Incorporating advanced imaging modalities like weight-bearing MRI and diffusion tensor imaging may enhance diagnostic accuracy. Moreover, studies should integrate clinical, functional, and psychosocial parameters to better reflect the multifactorial nature of LFS. Developing a unified diagnostic framework that combines radiological markers with patient-reported outcomes, electrodiagnostic findings, and biomechanical assessments could significantly improve clinical decision-making. Longitudinal studies tracking symptom progression relative to imaging changes would also provide valuable insights into the natural history of foraminal stenosis. Further exploration into the influence of modifiable risk factors, such as BMI and psychological health, as well as genetic predispositions, could reveal new intervention targets. Emphasizing patient-centered outcomes and individual variability in pain perception will ensure more tailored and effective treatment strategies. Finally, creating a consensus on which imaging-based thresholds should guide surgical versus conservative management remains a critical unmet need. Addressing these future directions will strengthen the diagnostic precision and therapeutic efficacy for patients suffering from LFS.
This systematic review demonstrates that the radiological severity of LFS shows only moderate and inconsistent correlation with clinical symptoms, particularly pain and functional disability. Conventional MRI measurements alone are insufficient to reliably identify symptomatic patients, as significant anatomical narrowing may be present in asymptomatic individuals, while clinically relevant symptoms can occur with modest imaging changes. Emerging radiological markers, such as the sedimentation sign, may enhance diagnostic specificity but require further validation. These findings support a diagnostic approach that integrates imaging with detailed clinical evaluation, functional assessment, and psychosocial profiling. Reliance on radiological findings in isolation risks misclassification and inappropriate management. Future studies should focus on standardized imaging criteria combined with patient-centered outcome measures to improve diagnostic precision and treatment decision-making in LFS.
Recommendation
Based on the findings of this systematic review, clinical decision-making in LFS should not rely on imaging findings alone. Radiological assessment should be integrated with detailed clinical evaluation, functional assessment, and psychosocial profiling to improve diagnostic accuracy and patient selection for intervention. Future research should prioritize standardized imaging protocols, validated grading systems, and prospective studies that combine radiological, clinical, and patient-reported outcomes to better define clinically meaningful diagnostic thresholds.
Treat the patient’s symptoms and functional limitation, not the MRI appearance of foraminal narrowing in isolation.
References
- 1. </p> [Google Scholar] [PubMed]
- 2. <p>1. Jenis LG, An HS. Spine update. Lumbar foraminal stenosis. Spine (Phila Pa 1976) 2000;25:389-94. [Google Scholar] [PubMed]
- 3. Qaraghli MI, Jesus OD. Lumbar disc herniation. In: StatPearls. StatPearls Publishing; 2023. Available from: https://www.ncbi.nlm.nih.gov/books/NBK560878 [Last accessed on 2025 May 21]. [Google Scholar] [PubMed]
- 4. Nurmukhametov R, Encarnacion Ramirez MJ, Dosanov M, Medetbek A, Kudryakov S, Wisam Alsaed L, et al. Quantifying lumbar foraminal volumetric dimensions: Normative data and implications for stenosis-part 2 of a comprehensive series. Med Sci (Basel) 2024;12:34. [Google Scholar] [PubMed]
- 5. Ruiz Santiago F, Láinez Ramos-Bossini AJ, Wáng YX, Martínez Barbero JP, García Espinosa J, Martínez Martínez A. The value of magnetic resonance imaging and computed tomography in the study of spinal disorders. Quant Imaging Med Surg 2022;12:3947-86. [Google Scholar] [PubMed]
- 6. Munakomi S, Cruz R. Lumbar spinal stenosis. In: StatPearls. StatPearls Publishing; 2025. Available from: https://www.ncbi.nlm.nih.gov/books/NBK531493 [Last accessed on 2025 May 21]. [Google Scholar] [PubMed]
- 7. Steurer J, Roner S, Gnannt R, Hodler J, LumbSten Research Collaboration. Quantitative radiologic criteria for the diagnosis of lumbar spinal stenosis: A systematic literature review. BMC Musculoskelet Disord 2011;12:175. [Google Scholar] [PubMed]
- 8. Bartynski WS, Lin L. Lumbar root compression in the lateral recess: MR imaging, conventional myelography, and CT myelography comparison with surgical confirmation. AJNR Am J Neuroradiol 2003;24:348-60. [Google Scholar] [PubMed]
- 9. Kunogi J, Hasue M. Diagnosis and operative treatment of intraforaminal and extraforaminal nerve root compression. Spine (Phila Pa 1976) 1991;16:1312-20. [Google Scholar] [PubMed]
- 10. Lee S, Lee JW, Yeom JS, Kim KJ, Kim HJ, Chung SK, et al. A practical MRI grading system for lumbar foraminal stenosis. AJR Am J Roentgenol 2010;194:1095-8. [Google Scholar] [PubMed]
- 11. Yamada K, Matsuda H, Nabeta M, Habunaga H, Suzuki A, Nakamura H. Clinical outcomes of microscopic decompression for degenerative lumbar foraminal stenosis: A comparison between patients with and without degenerative lumbar scoliosis. Eur Spine J 2011;20:947-53. [Google Scholar] [PubMed]
- 12. Schizas C, Theumann N, Burn A, Tansey R, Wardlaw D, Smith FW, et al. Qualitative grading of severity of lumbar spinal stenosis based on the morphology of the dural sac on magnetic resonance images. Spine (Phila Pa 1976) 2010;35:1919-24. [Google Scholar] [PubMed]
- 13. Rai A, Kolli M, Li Cai CY, Rai S, Girivasan SN. Evaluation of the smokeless tobacco awareness, attitude, and response knowledge (STAARK) scale with integrated risk assessment for tailored intervention. Cureus 2024;16:e70744. [Google Scholar] [PubMed]
- 14. Boden SD, Davis DO, Dina TS, Patronas NJ, Wiesel SW. Abnormal magnetic-resonance scans of the lumbar spine in asymptomatic subjects. A prospective investigation. J Bone Joint Surg Am 1990;72:403-8. [Google Scholar] [PubMed]
- 15. Yilihamu EE, Zeng FS, Shang J, Yang JT, Zhong H, Feng SQ. GPT4LFS (generative pretrained transformer 4 omni for lumbar foramina stenosis): Enhancing lumbar foraminal stenosis image classification through large multimodal models. Spine J 2025;25:2071-80. [Google Scholar] [PubMed]
- 16. Lurie J, Tomkins-Lane C. Management of lumbar spinal stenosis. BMJ 2016;352:h6234. [Google Scholar] [PubMed]
- 17. Vargas MI, Delattre BM, Boto J, Gariani J, Dhouib A, Fitsiori A, et al. Advanced magnetic resonance imaging (MRI) techniques of the spine and spinal cord in children and adults. Insights Imaging 2018;9:549-57. [Google Scholar] [PubMed]
- 18. Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. [Google Scholar] [PubMed]
- 19. Sterne JA, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019;366:l4898. [Google Scholar] [PubMed]
- 20. Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016;355:i4919. [Google Scholar] [PubMed]
- 21. Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, et al. GRADE: An emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008;336:924-6. [Google Scholar] [PubMed]
- 22. Wang R, Lin X, Shi S, Xiu Z, Guo Y. Instability of lower lumbar treated with posterior lumbar interbody fusion with autologous iliac crest or interbody fusion cage: A comparative study. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi 2008;22:928-32. [Google Scholar] [PubMed]
- 23. Yuan JD, Wang J, Zhou HB, Fu Q, Chen ZM, Zhao J. Analysis of results on minimum 4-year follow-up of modified posterior lumbar interbody fusion for the treatment of isthmic spondylolisthesis. Zhongguo Gu Shang 2010;23:519-22. [Google Scholar] [PubMed]
- 24. Liu M, Kan W, Li P, He D. Effectiveness of discectomy combined with Isobar non-fusion internal fixation in treating lumbar disc protrusion. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi 2011;25:229-34. [Google Scholar] [PubMed]
- 25. Leivseth G, Braaten S, Frobin W, Brinckmann P. Mobility of lumbar segments instrumented with a ProDisc II prosthesis: A two-year follow-up study. Spine (Phila Pa 1976) 2006;31:1726-33. [Google Scholar] [PubMed]
- 26. Haig AJ, Tong HC, Yamakawa KS, Quint DJ, Hoff JT, Chiodo A, et al. Spinal stenosis, back pain, or no symptoms at all? A masked study comparing radiologic and electrodiagnostic diagnoses to the clinical impression. Arch Phys Med Rehabil 2006;87:897-903. [Google Scholar] [PubMed]
- 27. Kalichman L, Cole R, Kim DH, Li L, Suri P, Guermazi A, et al. Spinal stenosis prevalence and association with symptoms: The Framingham Study. Spine J 2009;9:545-50. [Google Scholar] [PubMed]
- 28. Zeifang F, Schiltenwolf M, Abel R, Moradi B. Gait analysis does not correlate with clinical and MR imaging parameters in patients with symptomatic lumbar spinal stenosis. BMC Musculoskelet Disord 2008;9:89. [Google Scholar] [PubMed]
- 29. Sirvanci M, Bhatia M, Ganiyusufoglu KA, Duran C, Tezer M, Ozturk C, et al. Degenerative lumbar spinal stenosis: Correlation with Oswestry Disability Index and MR imaging. Eur Spine J 2008;17:679-85. [Google Scholar] [PubMed]
- 30. Barz T, Melloh M, Staub LP, Lord SJ, Lange J, Röder CP, et al. Nerve root sedimentation sign: Evaluation of a new radiological sign in lumbar spinal stenosis. Spine (Phila Pa 1976) 2010;35:892-7. [Google Scholar] [PubMed]
- 31. Madsen R, Jensen TS, Pope M, Sørensen JS, Bendix T. The effect of body position and axial load on spinal canal morphology: An MRI study of central spinal stenosis. Spine (Phila Pa 1976) 2008;33:61-7. [Google Scholar] [PubMed]
- 32. Lurie JD, Tosteson AN, Tosteson TD, Carragee E, Carrino JA, Kaiser J, et al. Reliability of magnetic resonance imaging readings for lumbar disk herniation in the Spine Patient Outcomes Research Trial (SPORT). Spine (Phila Pa 1976) 2008;33:991-8. [Google Scholar] [PubMed]
- 33. Jarvik JG, Hollingworth W, Heagerty PJ, Haynor DR, Boyko EJ, Deyo RA. Three-year incidence of low back pain in an initially asymptomatic cohort: Clinical and imaging risk factors. Spine (Phila Pa 1976) 2005;30:1541-8; discussion 1549. [Google Scholar] [PubMed]
- 34. Katz JN, Harris MB. Clinical practice. Lumbar spinal stenosis. N Engl J Med 2008;358:818-25. [Google Scholar] [PubMed]
- 35. Vroomen PC, de Krom MC, Knottnerus JA. Predicting the outcome of sciatica at short-term follow-up. Br J Gen Pract 2002;52:119-23. [Google Scholar] [PubMed]
- 36. Tarulli AW, Raynor EM. Lumbosacral radiculopathy. Neurol Clin 2007;25:387-405. [Google Scholar] [PubMed]
- 37. Konstantinou K, Dunn KM. Sciatica: Review of epidemiological studies and prevalence estimates. Spine (Phila Pa 1976) 2008;33:2464-72. [Google Scholar] [PubMed]
- 38. Roland M, Morris R. A study of the natural history of back pain. Part I: Development of a reliable and sensitive measure of disability in low-back pain. Spine (Phila Pa 1976) 1983;8:141-4. [Google Scholar] [PubMed]
- 39. Fairbank JC, Pynsent PB. The Oswestry disability index. Spine (Phila Pa 1976) 2000;25:2940-52; discussion 2952. [Google Scholar] [PubMed]
- 40. Porter RW. Spinal stenosis and neurogenic claudication. Spine (Phila Pa 1976) 1996;21:2046-52. [Google Scholar] [PubMed]
- 41. Suri P, Rainville J, Kalichman L, Katz JN. Does this older adult with lower extremity pain have the clinical syndrome of lumbar spinal stenosis? JAMA 2010;304:2628-36. [Google Scholar] [PubMed]
- 42. Arnoldi CC, Brodsky AE, Cauchoix J, Crock HV, Dommisse GF, Edgar MA, et al. Lumbar spinal stenosis and nerve root entrapment syndromes. Definition and classification. Clin Orthop Relat Res 1976;115:4-5. [Google Scholar] [PubMed]
- 43. Johnsson KE, Willner S, Johnsson K. Postoperative instability after decompression for lumbar spinal stenosis. Spine (Phila Pa 1976) 1986;11:107-10. [Google Scholar] [PubMed]
- 44. Ciol MA, Deyo RA, Howell E, Kreif S. An assessment of surgery for spinal stenosis: Time trends, geographic variations, complications, and reoperations. J Am Geriatr Soc 1996;44:285-90. [Google Scholar] [PubMed]
- 45. Weinstein JN, Tosteson TD, Lurie JD, Tosteson AN, Blood E, Hanscom B, et al. Surgical versus nonsurgical therapy for lumbar spinal stenosis. N Engl J Med 2008;358:794-810. [Google Scholar] [PubMed]
- 46. Kim Y, Lee C, Oh H, Son JS, Doo A. The effects of body composition characteristics on the functional disability in patients with degenerative lumbar spinal stenosis. J Clin Med 2023;12:612. [Google Scholar] [PubMed]
- 47. Pereira BJ, de Holanda CV, Ribeiro CA, de Moura SM, Galvão PE, Quidute BS, et al. Impact of body mass index in spinal surgery for degenerative lumbar spine disease. Clin Neurol Neurosurg 2014;127:112-5. [Google Scholar] [PubMed]
- 48. Abbas J, Hamoud K, Masharawi YM, May H, Hay O, Medlej B, et al. Ligamentum flavum thickness in normal and stenotic lumbar spines. Spine (Phila Pa 1976) 2010;35:1225-30. [Google Scholar] [PubMed]
- 49. Overdevest GM, Jacobs W, Vleggeert-Lankamp C, Thomé C, Gunzburg R, Peul W. Effectiveness of posterior decompression techniques compared with conventional laminectomy for lumbar stenosis. Cochrane Database Syst Rev 2015;2015:CD010036. [Google Scholar] [PubMed]
- 50. Aalto TJ, Malmivaara A, Kovacs F, Herno A, Alen M, Salmi L, et al. Preoperative predictors for postoperative clinical outcome in lumbar spinal stenosis: Systematic review. Spine (Phila Pa 1976) 2006;31:E648-63. [Google Scholar] [PubMed]
- 51. Flexman AM, Charest-Morin R, Stobart L, Street J, Ryerson CJ. Frailty and postoperative outcomes in patients undergoing surgery for degenerative spine disease. Spine J 2016;16:1315-23. [Google Scholar] [PubMed]
- 52. Tegeder I, Adolph J, Schmidt H, Woolf CJ, Geisslinger G, Lötsch J. Reduced hyperalgesia in homozygous carriers of a GTP cyclohydrolase 1 haplotype. Eur J Pain 2008;12:1069-77. [Google Scholar] [PubMed]











