This article demonstrates that photobiomodulation therapy (PBMT) can modulate both systemic and local inflammatory mediators in knee osteoarthritis, reducing biomarkers associated with cartilage degradation while improving clinical outcomes such as pain and joint mobility. It highlights the potential biochemical mechanisms underlying PBMT analgesic effects and supports its use as a non-invasive adjunct therapy for osteoarthritis management.
Dr. Marucia Chacur, Department of Anatomy, Neuroanatomy Functional of Pain Laboratory, Institute of Biomedical Sciences, University of Sao Paulo, Sao Paulo, Brazil E-mail:chacurm@icb.usp.br
Introduction: Knee osteoarthritis (KOA) is a common degenerative joint disease characterized by pain, inflammation, and progressive cartilage degradation. Current treatments often provide limited relief, encouraging the exploration of adjuvant, non-invasive modalities such as photobiomodulation therapy (PBMT). Although PBMT has demonstrated analgesic and anti-inflammatory potential, its biochemical effects in KOA remain underexplored.
Case Report: This pilot clinical investigation included 30 patients with clinically and radiographically confirmed KOA, allocated into two groups (PBMT, n = 15; placebo, n = 15). All participants received identical treatment protocols, with the placebo group undergoing sham irradiation. Synovial fluid and blood samples were collected before and after the intervention to assess inflammatory and cartilage metabolism biomarkers, including interleukins (IL-1, IL-6, and IL-10), tumor necrosis factor-alpha (TNF-α), matrix metalloproteinases (MMP-3 and MMP-13), collagen type II C-telopeptide (CTX-II), and prostaglandin E2 (PGE2).
Results: PBMT produced significant biochemical and clinical modulation compared with placebo. In serum, PBMT reduced IL-1, IL-6, TNF-α, and PGE2 levels while increasing IL-10 concentrations. In synovial fluid, significant reductions were observed in MMP-3, MMP-13, CTX-II, IL-1, IL-6, and TNF-α, accompanied by increased IL-10 levels. Clinically, PBMT participants reported substantial pain reduction and improved joint mobility.The observed reductions in inflammatory cytokines and cartilage degradation biomarkers indicate that PBMT exerts both systemic and intra-articular modulatory effects. The increase in IL-10 suggests activation of anti-inflammatory regulatory pathways, which may contribute to the clinical improvements observed. These findings reinforce the hypothesis that PBMT acts not only through symptomatic relief but also through modulation of disease-related biochemical mechanisms.
Conclusion: PBMT demonstrates beneficial local and systemic effects on inflammatory and cartilage degradation pathways in KOA, supporting its potential as an adjunctive therapeutic strategy. Larger controlled clinical trials are warranted to confirm these findings and establish standardized treatment parameters.
Keywords: Knee osteoarthritis, photobiomodulation therapy, inflammation, interleukins.
Osteoarthritis (OA) is a chronic inflammatory disease that can impact any joint, leading to the progressive degeneration of cartilage. Knee osteoarthritis (KOA) is the most prevalent form of OA, accounting for 85% of OA cases and affecting 37% of individuals over the age of 60 years [1]. The prevalence of KOA is expected to continuously escalate due to population aging and increases in obesity rates. It is characterized by several pathological changes, including synovitis, cartilage degeneration, subchondral bone remodeling, and osteophyte formation. These changes contribute to joint pain, swelling, stiffness, physical impairments, reduced quality of life, and potential disability [2,3]. At the molecular level, these structural alterations are largely driven by an imbalance between anabolic and catabolic processes within the joint. Proteolytic enzymes, particularly matrix metalloproteinases (MMPs), play a central role in mediating extracellular matrix breakdown, making them key contributors to cartilage degradation in KOA [4]. MMPs are enzymes crucial for various physiological processes; however, elevated levels have been consistently observed in the synovial fluid of KOA patients. Research by Lohmander et al. demonstrated that synovial fluid contains increased levels of aggrecan fragments and cartilage proteins, which are markers of matrix metabolism, following initial joint trauma, indicating heightened joint tissue degradation [5,6]. These findings highlight the pivotal role of MMP activity and matrix-derived fragments in the pathophysiology of KOA and support their use as relevant biomarkers for disease monitoring and therapeutic targeting. MMP-3 (stromelysin-1) is a significant enzyme in the MMP cascade due to its ability to degrade various cartilage components, including gelatin, aggrecan, and collagen types III, IV, IX, and X. It also activates proMMPs 1, 7, 8, 9, and 13. In addition, collagenases (MMP-1, MMP-8, and MMP-13) can degrade intact collagen type II, a primary component of articular cartilage, which is further degraded by gelatinases [7,8]. MMP activity is regulated at multiple levels, with gene expression being particularly important and influenced by various growth factors and cytokines [3,9]. During inflammation, immune cells such as macrophages, lymphocytes, and neutrophils infiltrate the joint, leading to synovial fluid accumulation. Cytokines and lipid-derived inflammatory mediators in synovial fluid have been widely investigated, particularly in rheumatoid arthritis and OA [10]. Pre-clinical evidence suggests that interleukin-6 (IL-6) plays a critical role in central sensitization, chronic inflammation, and autoimmunity. In human studies, IL-6 has been linked to KOA onset, structural progression, and reduced physical function [11]. Similarly, changes in inflammatory mediators, including IL-1, tumor necrosis factor-alpha (TNF-α), nitric oxide (NO), and MMPs, promote matrix component loss and progressive cartilage destruction [12,13]. Biochemical analyses consistently confirm that synovial membrane inflammation is central to OA pathophysiology. Pro-inflammatory cytokines such as IL-1β and TNF-α, secreted mainly by macrophages, exacerbate joint inflammation and stimulate synovial cells to release additional mediators (cytokines, chemokines, and MMPs, notably MMP-3, -8, and -13) [14]. These processes compromise collagen type II integrity, increase collagen type II C-telopeptide (CTX-II) levels, and drive articular tissue degeneration and joint space narrowing [15,16]. Current treatments for KOA, including conservative therapies (e.g., exercise and lifestyle modifications), pharmacological management, and surgical interventions, primarily aim to alleviate pain and preserve function [17]. However, conservative strategies often provide limited efficacy, and pharmacological or surgical options may not be suitable for all patients, underscoring the need for alternative or complementary approaches. Over the past three decades, research has explored photobiomodulation therapy (PBMT) as a potential non-pharmacological treatment for KOA in both human patients and animal models. Findings suggest that PBMT can reduce pain and modulate inflammatory responses associated with OA progression [17,18]. Although its mechanisms remain under investigation, PBMT appears to influence key molecular pathways involved in joint inflammation and matrix degradation. For example, Nambi demonstrated that PBMT reduced pain and inhibited the proliferation of collagen type II C-telopeptide, as well as MMP-3, MMP-8, and MMP-13, compared with placebo treatments, suggesting its potential benefit even in later stages of AO [19]. Despite encouraging results, the clinical efficacy of PBMT from a biochemical perspective remains controversial, requiring further investigation. Therefore, this study aims to evaluate the effects of PBMT in patients with KOA. Specifically, we will assess its impact on the concentrations of IL-1, IL-6, IL-10, TNF-α, MMP-3, MMP-13, collagen type II C-telopeptide (CTX-II), and prostaglandin E2 (PGE2) in synovial fluid and blood samples.
Study population
The current study was approved by the Ethics Committee on Research of the Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo (CAAE: 93744418.5.0000.0068) and conducted in accordance with the principles of the Declaration of Helsinki. Written informed consent was obtained from all participants before inclusion. The trial was registered at ClinicalTrials.gov (NCT03924128). Data were collected at the Pain Clinic of the Department of Anesthesia, Hospital das Clínicas, Faculdade de Medicina da Universidade de São Paulo. Participants were screened for eligibility and enrolled after meeting the inclusion criteria. The study was conducted over 10 months, including baseline evaluation, treatment sessions, and post-treatment assessments. This case series is derived from a previously published randomized, double-blind, placebo-controlled pilot trial conducted by our research group [20]. For the present analysis, we used biological samples (synovial fluid and serum) obtained from 15 patients per group (PBMT and placebo) from that prior study to perform an extended investigation of the inflammatory response to PBMT, by comparing pre- and post-treatment measures with placebo controls. This secondary analysis provides a more detailed exploration of PBMT’s biochemical effects, complementing the previously reported clinical outcomes.
Inclusion criteria
Patients of both sexes, aged 40–90 years, diagnosed with chronic KOA according to the American College of Rheumatology criteria, and classified as grade III or IV according to the Kellgren–Lawrence radiological grading system, were included in this study.
Exclusion criteria
Patients with cancer, neurological deficits, fractures, cardiac diseases, or other conditions affecting the knee joint were excluded from the study.
Treatment procedure
Treatment was administered twice a week on the same days over a period of 5 weeks with the system Light-Aid (Bright Photomedicine, SP, Brazil), operated at a continuous wave of 850 nm or with a placebo probe of the same appearance and display. The probes were numbered A (active) and B (placebo). Treatment was administered in contact with the skin over the patient’s knee. Both knees were treated, but statistical analysis was done in the knee with the highest pain intensity on the numerical rating scale. Placebo treatment was identical to PBM treatment, in a way that the device was turned on, but the probe was disabled without emitting any energy, ensuring that neither the operator nor the subject could identify which group it belonged to. The probe device comprises 100 light-emitting diodes (LEDs) divided into 4 LED clusters with 25 LEDs each, which covered patients’ whole knee surface. The treatment was applied using a constant 5–8 min to deliver a total of 526J–1402J each session and a power density of 60–100 mW/cm² [20].
Laboratory examinations
Blood samples were collected from the antecubital vein, and synovial fluid was aspirated from the inflamed knee joints of all participants in the fasting state between 7:00 and 11:00 AM. After clotting, blood samples were centrifuged, and the serum was stored at −80°C until analysis. Concentrations of IL-1, IL-6, IL-10, TNF-α, MMP-3, MMP-13, CTX-II, and PGE2 in both serum and synovial fluid were determined using commercially available sandwich enzyme-linked immunosorbent assay kits (R&D Systems, CA, USA) following the manufacturer’s instructions. All measurements were performed in triplicate, and the mean values were used for statistical analysis.
Statistical analysis
GraphPad Prism was used to perform statistical analyses. Biomarker results were analyzed using one-way analysis of variance with repeated measures, followed by a Sidak-corrected post hoc test to determine possible differences between groups. A value of P < 0.05 was considered statistically significant in this study, and data were presented as means ± standard deviation.
Impact of PBMT on blood biomarkers in KOA
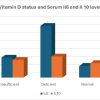
In the placebo group (PBMT device turned off), no significant changes were observed in blood biomarkers between pre- and post-intervention measurements (Fig. 1). In contrast, patients who received PBMT showed significant changes in specific markers. Serum IL-10 levels were significantly increased after PBMT compared with pre-treatment (P < 0.01), whereas IL-1β levels were significantly reduced (P <0.03). Similarly, serum MMP-3 concentrations were significantly increased following PBMT (P < 0.04). No statistically significant differences were found in TNF-α, IL-6, or MMP-13 levels between pre- and post-treatment. These results indicate that PBMT selectively modulates anti-inflammatory (IL-10) and matrix remodeling (MMP-3 and IL-1β) biomarkers in blood, whereas other inflammatory mediators remain unaffected.
Effects of PBMT on synovial fluid biomarkers in KOA
Analysis of synovial fluid revealed that PBMT produced selective effects on certain biomarkers (Fig. 2). Post-treatment, TNF-α levels were significantly reduced compared with baseline (P < 0.04), whereas MMP-3 levels were significantly increased (P < 0.05). No significant changes were observed in IL-1β, IL-6, IL-10, MMP-13, PGE2, or CTX-II concentrations following PBMT. These findings suggest that PBMT may attenuate local pro-inflammatory activity (via TNF-α reduction) while enhancing MMP-3 activity related to cartilage remodeling, without significantly altering other inflammatory or catabolic mediators in the synovial environment.
According to the World Health Organization, approximately 528 million people suffer from OA, with the knee being the most commonly affected joint, followed by the hip and hand. KOA is therefore a highly prevalent condition, affecting around 32.5 million adults in the United States alone, and symptomatic KOA impacts nearly 14% of the population [21,22,23]. It is characterized by progressive cartilage degeneration, synovial inflammation, and chronic pain, all of which contribute to significant disability and reduced quality of life. Current therapeutic approaches, including conservative management, pharmacological treatment, and surgical intervention, are primarily aimed at alleviating symptoms rather than modifying the underlying disease process [24]. However, their overall efficacy remains limited, underscoring the urgent need for innovative therapeutic strategies. PBMT has emerged as a promising non-invasive intervention, with both pre-clinical and clinical evidence suggesting beneficial effects on inflammation, pain modulation, and tissue repair mechanisms in KOA [25,26]. The pathophysiology of KOA involves a complex interplay between pro-inflammatory cytokines, MMPs, and biochemical markers of cartilage degradation. Elevated levels of cytokines such as IL-1β, IL-6, and TNF-α amplify joint inflammation and promote cartilage breakdown, while enzymes including MMP-3 and MMP-13 mediate extracellular matrix degradation. Moreover, CTX-II has been widely recognized as a biomarker of cartilage turnover and disease progression[4,27]. In this study, we demonstrated that PBMT induces selective changes in both systemic and local biomarkers associated with KOA. In serum, PBMT significantly increased IL-10 levels, a cytokine with potent anti-inflammatory properties that regulates immune responses and counteracts the activity of IL-1β and TNF-α. Elevated IL-10 has been shown to attenuate cartilage destruction and synovial inflammation in experimental OA models [28,29], supporting the hypothesis that PBMT may enhance endogenous protective pathways. Consistent with this, we observed a significant reduction in serum IL-1β after PBMT. IL-1β is recognized as a master regulator of cartilage catabolism and synovial inflammation, driving the production of degradative enzymes such as MMPs and aggrecanases [30,31,32,33,34]. The observed decrease in IL-1β, therefore, highlights a plausible mechanism through which PBMT could mitigate KOA progression. Notably, previous studies have also reported that PBMT can reduce IL-1β and increase IL-10 in both animal models of arthritis and clinical contexts [24,33,34], although the available evidence remains scarce. These results reinforce the therapeutic potential of PBMT while underscoring the need for further studies with larger cohorts and standardized protocols to corroborate these immunomodulatory effects. An unexpected yet noteworthy finding was the significant increase in MMP-3 levels in both serum and synovial fluid after PBMT. MMP-3 (stromelysin-1) plays a dual role in cartilage metabolism: On one hand, it contributes to extracellular matrix degradation by cleaving proteoglycans and non-collagenous proteins, and by activating proMMPs such as MMP-1 and MMP-9 [4,35]. On the other hand, MMP-3 has been implicated in matrix remodeling and repair responses, suggesting that its upregulation may not exclusively reflect destructive processes but could also indicate increased tissue turnover and adaptive remodeling [27,36]. Elevated MMP-3 levels in synovial fluid and serum have been consistently associated with OA progression and are considered sensitive biomarkers for joint tissue metabolism and therapeutic monitoring [5,37]. In this context, the paradoxical increase observed after PBMT may represent a transient compensatory response linked to tissue repair rather than pathological degradation. Importantly, the clinical significance of elevated MMP-3 following PBMT remains uncertain and warrants further investigation, particularly through longitudinal studies assessing its relationship with structural outcomes and functional improvement. Taken together, our results suggest that PBMT may modulate KOA pathophysiology by simultaneously enhancing anti-inflammatory responses (↑ IL-10, ↓ IL-1β, and ↓ TNF-α) and influencing matrix remodeling dynamics (↑ MMP-3). These findings align with previous studies showing that PBMT reduces pro-inflammatory cytokines and oxidative stress while upregulating anti-inflammatory mediators in both pre-clinical models and clinical settings of musculoskeletal disorders [33,34,38]. Furthermore, PBMT has been reported to stimulate tissue repair processes, including angiogenesis, collagen synthesis, and mitochondrial bioenergetics, which may contribute to improved joint homeostasis [18]. The paradoxical increase in MMP-3 observed here raises the possibility that PBMT not only suppresses inflammation but also activates remodeling pathways, consistent with its reported role in accelerating tissue healing. Nonetheless, the dual nature of MMP-3, implicated in both cartilage degradation and repair, underscores the need for cautious interpretation. Longitudinal and mechanistic studies will be essential to clarify whether PBMT-induced increases in MMP-3 represent beneficial remodeling or detrimental catabolic activity, and how this dynamic relates to long-term structural and functional outcomes in KOA.
This case series highlights that PBMT can selectively modulate both systemic and local inflammatory and cartilage-related biomarkers in patients with KOA, supporting its proposed anti-inflammatory and tissue-modulating actions. The observed increase in IL-10 and modulation of MMP-3, alongside reductions in IL-1β and TNF-α, suggest that PBMT may contribute to a shift toward a more balanced inflammatory environment within the joint. Although not all biomarkers responded uniformly, particularly IL-6, MMP-13, CTX-II, and PGE2, the findings provide valuable insights into the complex biochemical effects of light therapy in degenerative joint disease. Clinically, these results add to the growing body of evidence supporting PBMT as a safe, non-invasive, and potentially disease-modifying adjunctive therapy for KOA. From a broader perspective, this report advances our understanding of how light-based interventions may influence inflammatory networks and cartilage metabolism, offering new perspectives for the management of OA and related musculoskeletal disorders. Further studies with larger cohorts and extended follow-up are warranted to confirm these findings and clarify their implications for long-term joint protection and functional recovery.
Photobiomodulation therapy may represent a safe, non-invasive adjunct treatment for knee osteoarthritis, capable of reducing inflammatory activity and cartilage degradation while improving pain and joint function, supporting its integration into multimodal clinical management strategies.
References
- 1. Hunter DJ, Bierma-Zeinstra S. Osteoarthritis. Lancet 2019;393:1745-59. [Google Scholar] [PubMed]
- 2. Perrone V, Veronesi C, Ciappetta M, Lucatelli D, Cinti Luciani A, Degli Esposti L. An up-to-date description of the use of non-steroidal anti-inflammatory drugs (NSAIDs) in Italy: Evidence from real clinical practice. Adv Ther 2025;42:2354-68. [Google Scholar] [PubMed]
- 3. Zhou Y, Liu Y, Guo Y, Liu X, Zhou Y. Analysis of the current status and factors influencing post-traumatic stress disorder in elderly patients with knee osteoarthritis: A cross-sectional study. Sci Rep 2025;15:10253. [Google Scholar] [PubMed]
- 4. Murphy G, Nagase H. Progress in matrix metalloproteinase research. Mol Aspects Med 2008;29:290-308. [Google Scholar] [PubMed]
- 5. Lohmander LS, Saxne T, Heinegard DK. Release of cartilage oligomeric matrix protein (COMP) into joint fluid after knee injury and in osteoarthritis. Ann Rheum Dis 1994;53:8-13. [Google Scholar] [PubMed]
- 6. Lohmander LS, Ionescu M, Jugessur H, Poole AR. Changes in joint cartilage aggrecan after knee injury and in osteoarthritis. Arthritis Rheum 1999;42:534-44. [Google Scholar] [PubMed]
- 7. Srivastava V, Harsulkar A, Aphale S, Märtson A, Kõks S, Kulkarni P, et al. Functional attributes of synovial fluid from osteoarthritic knee exacerbate cellular inflammation and metabolic stress, and fosters monocyte to macrophage differentiation. Biomedicines 2025;13:878. [Google Scholar] [PubMed]
- 8. Murphy JM, Dixon K, Beck S, Fabian D, Feldman A, Barry F. Reduced chondrogenic and adipogenic activity of mesenchymal stem cells from patients with advanced osteoarthritis. Arthritis Rheum 2002;46:704-13. [Google Scholar] [PubMed]
- 9. Wang W, Du Y, Zhang X. Nimesulide and sodium hyaluronate ameliorate the inflammatory process and pain severity in traumatic knee osteoarthritis. Inflammopharmacology 2025;33:3135-42. [Google Scholar] [PubMed]
- 10. Barden J, Derry S, McQuay HJ, Moore AR. Bias from industry trial funding? A framework, a suggested approach, and a negative result. Pain 2006;121:207-18. [Google Scholar] [PubMed]
- 11. Lee YC, Lu B, Bathon JM, Haythornthwaite JA, Smith MT, Page GG, et al. Pain sensitivity and pain reactivity in osteoarthritis. Arthritis Care Res (Hoboken) 2011;63:320-7. [Google Scholar] [PubMed]
- 12. Uchida K, Takano S, Matsumoto T, Nagura N, Inoue G, Itakura M, et al. Transforming growth factor activating kinase 1 regulates extracellular matrix degrading enzymes and pain-related molecule expression following tumor necrosis factor-α stimulation of synovial cells: An in vitro study. BMC Musculoskelet Disord 2017;18:283. [Google Scholar] [PubMed]
- 13. Takano S, Uchida K, Itakura M, Iwase D, Aikawa J, Inoue G, et al. Transforming growth factor-β stimulates nerve growth factor production in osteoarthritic synovium. BMC Musculoskelet Disord 2019;20:204. [Google Scholar] [PubMed]
- 14. Ismail HM, Didangelos A, Vincent TL, Saklatvala J. Rapid activation of transforming growth factor β-activated kinase 1 in chondrocytes by phosphorylation and K63-linked polyubiquitination upon injury to animal articular cartilage. Arthritis Rheumatol 2017;69:565-75. [Google Scholar] [PubMed]
- 15. Demir-Dora D, Tuna S, Kurtoglu ED, Gursoy S, Balci N, Tezman S, et al. Evaluation of the efficacy and safety of collasel pro® type I and type III hydrolyzed collagen peptides in the treatment of osteoarthritis: A double-blind, placebo-controlled, randomized clinical trial. J Clin Med 2025;14:3655. [Google Scholar] [PubMed]
- 16. Genc AS, Yilmaz AK, Anil B, Korkmaz Salkılıç E, Akdemir E, Güzel B, et al. The effect of supplementation with type I and type III collagen peptide and type II hydrolyzed collagen on pain, quality of life and physical function in patients with meniscopathy: A randomized, double-blind, placebo-controlled study. BMC Musculoskelet Disord 2025;26:17. [Google Scholar] [PubMed]
- 17. Li BM, Zhang CK, He JH, Liu YQ, Bao XY, Li FH. The effects of photobiomodulation on knee function, pain, and exercise tolerance in older adults: A meta-analysis of randomized controlled trials. Arch Phys Med Rehabil 2024;105:593-603. [Google Scholar] [PubMed]
- 18. Hamblin MR. Mechanisms and applications of the anti-inflammatory effects of photobiomodulation. AIMS Biophys 2017;4:337-61. [Google Scholar] [PubMed]
- 19. Nambi G. Does low level laser therapy has effects on inflammatory biomarkers IL-1β, IL-6, TNF-α, and MMP-13 in osteoarthritis of rat models-a systemic review and meta-analysis. Lasers Med Sci 2021;36:475-84. [Google Scholar] [PubMed]
- 20. Pinto NC, Pires De MV, Ferreira NL, Braga NA, Aldred A, Gomes G, et al. Customized photobiomodulation modulates pain and alters thermography pattern in patients with knee osteoarthritis: A randomized double-blind pilot study. Photobiomodul Photomed Laser Surg 2022;40:698-707. [Google Scholar] [PubMed]
- 21. Dong Y, Yan Y, Zhou J, Zhou Q, Wei H. Evidence on risk factors for knee osteoarthritis in middle-older aged: A systematic review and meta analysis. J Orthop Surg Res 2023;18:634. [Google Scholar] [PubMed]
- 22. Cieza A, Causey K, Kamenov K, Hanson SW, Chatterji S, Vos T. Global estimates of the need for rehabilitation based on the global burden of disease study 2019: A systematic analysis for the global burden of disease study 2019. Lancet 2021;396:2006-17. [Google Scholar] [PubMed]
- 23. Long H, Liu Q, Yin H, Wang K, Diao N, Zhang Y, et al. Prevalence trends of site-specific osteoarthritis from 1990 to 2019: Findings from the global burden of disease study 2019. Arthritis Rheumatol 2022;74:1172-83. [Google Scholar] [PubMed]
- 24. Araújo HR, Trindade AM, Jacob TC, Freitas GG, Barbosa SF, Goiás MT, et al. Comprehensive review of conservative and surgical treatment strategies for knee osteoarthritis: Efficacy, safety, and emerging therapies. Glob J Orthop 2025;1:24-37. [Google Scholar] [PubMed]
- 25. Pérez SE, Niebla JR, Pérez LG, León RG, Mejías AL, Tejera DM, et al. Effectiveness of photobiomodulation therapy in the management of fibromyalgia syndrome: A systematic review. Appl Sci 2025;15:4161. [Google Scholar] [PubMed]
- 26. Liu H, Cheema U, Player DJ. Photobiomodulation therapy (PBMT) in skeletal muscle regeneration: A comprehensive review of mechanisms, clinical applications, and future directions. Photodiagnosis Photodyn Ther 2025;53:104634. [Google Scholar] [PubMed]
- 27. Burrage PS, Mix KS, Brinckerhoff CE. Matrix metalloproteinases: Role in arthritis. Front Biosci 2006;11:529-43. [Google Scholar] [PubMed]
- 28. Fernandes JC, Martel-Pelletier J, Pelletier JP. The role of cytokines in osteoarthritis pathophysiology. Biorheology 2002;39:237-46. [Google Scholar] [PubMed]
- 29. Saraiva M, Vieira P, O’Garra A. Biology and therapeutic potential of interleukin-10. J Exp Med 2020;217 (1):e20190418. [Google Scholar] [PubMed]
- 30. Lubberts E. Th17 cytokines and arthritis. Semin Immunopathol 2010;32:43-53. [Google Scholar] [PubMed]
- 31. Goldring MB, Otero M. Inflammation in osteoarthritis. Curr Opin Rheumatol 2011;23:471-8. [Google Scholar] [PubMed]
- 32. Kapoor M, Martel-Pelletier J, Lajeunesse D, Pelletier JP, Fahmi H. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat Rev Rheumatol 2011;7:33-42. [Google Scholar] [PubMed]
- 33. Tomazoni SS, Costa LO, Joensen J, Stausholm MB, Naterstad IF, Leal-Junior EC, et al. Effects of photobiomodulation therapy on inflammatory mediators in patients with chronic non-specific low back pain: Protocol for a randomized placebo-controlled trial. Medicine (Baltimore) 2019;98:e15177. [Google Scholar] [PubMed]
- 34. Tomazoni SS, Leal-Junior EC, Frigo L, Pallotta RC, Teixeira S, De Almeida P, et al. Isolated and combined effects of photobiomodulation therapy, topical nonsteroidal anti-inflammatory drugs, and physical activity in the treatment of osteoarthritis induced by papain. J Biomed Opt 2016;21:108001. [Google Scholar] [PubMed]
- 35. Vincenti MP, Brinckerhoff CE. Transcriptional regulation of collagenase (MMP-1, MMP-13) genes in arthritis: Integration of complex signaling pathways for the recruitment of gene-specific transcription factors. Arthritis Res 2002;4:157-64. [Google Scholar] [PubMed]
- 36. Davidson RK, Waters JG, Kevorkian L, Darrah C, Cooper A, Donell ST, et al. Expression profiling of metalloproteinases and their inhibitors in synovium and cartilage. Arthritis Res Ther 2006;8:R124. [Google Scholar] [PubMed]
- 37. Eckstein F, Hochberg MC, Guehring H, Moreau F, Ona V, Bihlet AR, et al. Long-term structural and symptomatic effects of intra-articular sprifermin in patients with knee osteoarthritis: 5-year results from the FORWARD study. Ann Rheum Dis 2021;80:1062-9. [Google Scholar] [PubMed]
- 38. Tomazoni SS, Frigo L, Ferreira TC, Casalechi HL, Teixeira S, De Almeida P, et al. Effects of photobiomodulation therapy and topical non-steroidal anti-inflammatory drug on skeletal muscle injury induced by contusion in rats-part 2: Morphological and functional aspects. Lasers Med Sci 2017;32:1879-87. [Google Scholar] [PubMed]