Solitary plasmacytoma may progress to multiple myeloma despite localized disease at presentation, necessitating long-term vigilant follow-up with laboratory surveillance and sensitive imaging.
Dr. Vishnu Senthil, Department of Orthopedic, Government Roypettah Hospital, Chennai, Tamil Nadu, India. E mail: drvishnusenthil@gmail.com
Introduction: Solitary plasmacytoma (SP) is a rare plasma cell dyscrasia characterized by localized monoclonal plasma cell proliferation without systemic involvement. Although radiotherapy remains the standard treatment, progression to multiple myeloma (MM) is a recognized risk.
Case Report: We report a case of SP initially presenting as a localized bone lesion, diagnosed through imaging, histopathology, and laboratory evaluation. The patient underwent standard treatment and was closely followed. Over time, the disease progressed to MM, confirmed by clinical deterioration, laboratory findings, and radiological evidence.
Conclusion: This case highlights the aggressive potential of SP and reinforces the importance of prolonged surveillance for early detection of progression to MM.
Keywords: Solitary plasmacytoma, multiple myeloma, plasma cell dyscrasia, disease progression.
Solitary plasmacytoma (SP) is defined as a localized mass of neoplastic plasma cells and can be classified into two types based on location: Skeletal plasmacytoma and non-skeletal plasmacytoma. The clinical outcome of SP varies significantly; while many patients achieve a cure with appropriate therapy, some eventually develop disseminated multiple myeloma (MM) years later. Identifying predictors associated with plasma cell malignant proliferation and detecting early signs of SP aggravation are crucial for effective patient management and improved survival outcomes. Generally, clinicians assume that SP patients without high-risk profiles for MM progression will either be cured or experience only a slow transition into MM. This case report describes a case of SP of the lumbar vertebra L3 with a few rapid progression factors, which developed into MM after initial diagnosis despite appropriate treatment. We also review previous literature reports on related factors involved in disseminated MM.
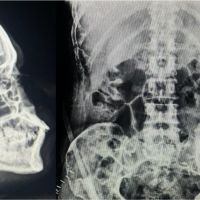
53-year-old male, initially presented with low back pain and was found on computed tomography (CT) scan to have a solitary lytic lesion in the L3 vertebral body (Fig. 1).

Figure 1: Plain radiograph of anterior-posterior (a) and lateral view (b) of lumbar spine showing a L3 lytic lesion (shown by the arrow mark). Axial section of computed tomography scan (c) of lumbar vertebrae showing an expansile lytic lesion at L3.
A biopsy confirmed that this lesion consisted of neoplastic plasma cells positive for CD138, confirming a localized plasma cell neoplasm. Imaging through sagittal and axial sequences demonstrated the destruction of the L3 vertebrae (Fig. 2).

Figure 2: Sagittal cuts (a) shows a T1 sequence solitary hypointense lesion in the body of L3 vertebrae and T2 sequence (b) shows a hyperintense lesion. T2 sequence axial images (c) shows lytic destruction of L3 vertebrae.
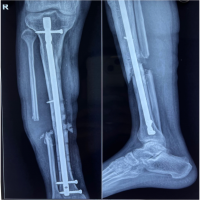
It that time, extensive evaluations, including skeletal survey, bone scan, and bone marrow biopsy, showed no evidence of systemic disease: The bone marrow contained <10% plasma cells, and there was no hypercalcemia, significant cytopenia, or renal dysfunction. These findings met the criteria for a diagnosis of SP, a localized plasma cell tumor. Subsequently, underwent posterior spinal stabilization with pedicle screw fixation at L2 and L4 vertebrae to support the spine structurally (Fig. 3).

Figure 3: Intraoperative image (a) showing the posterior decompression and pedicle screw fixation with biopsy. Post-Operative radiograph of lumbar spine, anterior-posterior view (a) and lateral view (b) with pedicle screws in position.
Laboratory investigations during his initial workup revealed elevated kappa light chain levels and an abnormal kappa/lambda ratio, suggesting a monoclonal (M) protein presence despite the initially localized disease. Over time, the solitary lesion progressed rapidly to systemic involvement, as indicated by a positron emission tomography (PET)-CT scan that demonstrated multiple metabolically active lytic lesions with soft tissue components in various bones, including bilateral clavicles, scapulae, sternum, ribs, vertebrae, sacrum, and pelvis. This progression fulfilled the diagnostic criteria for active MM, characterized by clonal plasma cell infiltration in bone marrow along with myeloma-defining features, specifically multifocal bone lesions identified by advanced imaging. Radiotherapy (30 Gy) was directed at his bone lesions and systemic chemotherapy with bortezomib, thalidomide, and dexamethasone, alongside granulocyte-macrophage colony-stimulating factors ,was administered as supportive treatment. After eight cycles of chemotherapy, re-evaluation showed marked clinical improvement: Serum free light chain ratio normalized, no M protein was detected on serum immunofixation and protein electrophoresis, and PET-CT scan revealed near-complete resolution of metabolic activity despite the persistence of some structural bone lesions. His beta-2 microglobulin level also decreased significantly, consistent with a favorable response and remission status. Following this systemic therapy, the patient was able to mobilize independently at 1-year follow-up (Fig. 4).

Figure 4: Patient at 1-year follow-up showing good outcome with no neurological deficit.
However, follow-up bone marrow biopsies showed a slight increase in plasma cells (up to 3%), indicating an early relapse of the disease. Given this relapse, treatment was modified to a second-line regimen, including cyclophosphamide and bortezomib, along with antiviral and antibacterial prophylaxis with acyclovir and septran to prevent infections during immunosuppression. Further close monitoring with periodic bone marrow evaluations and imaging is ongoing to assess treatment response and guide further management, with consideration for autologous stem cell transplantation if remission is achieved. This case exemplifies the importance of comprehensive initial evaluation, the dynamic progression from localized plasmacytoma to systemic MM, and the necessity for vigilant, individualized treatment strategies to achieve and maintain disease control over time. It also highlights the challenges in managing relapse and the need for adaptable therapeutic regimens to prolong remission and improve patient outcomes.
MM is a malignant disorder resulting from the uncontrolled proliferation of clonal plasma cells in the bone marrow, which produces characteristic clinical manifestations, such as fatigue (due to anemia), bone pain (from osteolytic lesions), hypercalcemia, and renal dysfunction related to M protein production. Diagnosis relies on detecting the M protein in the serum or urine using techniques, such as serum protein electrophoresis, immunofixation, and serum free light chain assays, with immunoglobulin (Ig)G, IgA, or light chains as the most frequent types. In addition, a comprehensive workup includes blood tests, bone marrow examination – demonstrating ≥10% clonal plasma cells – and imaging, such as magnetic resonance imaging (MRI), CT, or PET/CT to evaluate bone lesions and marrow involvement [1,2]. The International Myeloma Working Group (IMWG) defines MM based on this laboratory and clinical findings and includes the presence of myeloma-defining events categorized by the CRAB criteria: Hypercalcemia, renal insufficiency, anemia, and bone lesions [1,3] (Table 1). Prognosis into MM is determined by tumor burden, underlying disease biology, and treatment response, with risk stratification using systems, such as the revised international staging system (ISS) and the durie-salmon system [4]. Assessment is enhanced by cytogenetic analyses, particularly fluorescence in situ hybridization (FISH), which identifies high-risk genetic markers, such as t (4;14), t (14;16), and del (17p), influencing both prognosis and therapeutic choices [5,6]. Treatment typically begins with a combination of regimens, such as bortezomib, lenalidomide, dexamethasone (VRD), followed by autologous stem cell transplantation (ASCT) for eligible patients to deepen responses [7,8]. Maintenance therapies, often using lenalidomide, are applied to extend remission, and newer agents, such as carfilzomib and pomalidomide are employed in relapse, often as part of triplet or quadruplet regimens [7,9]. The overall treatment strategy is personalized based on patient fitness, risk stratification, and previous therapeutic success. MM is a malignancy caused by the proliferation of clonal plasma cells in the bone marrow, resulting in clinical symptoms, such as fatigue, bone pain, anemia, hypercalcemia, and renal dysfunction due to the excessive M protein production. Diagnosis is established through laboratory detection of the M protein by serum protein electrophoresis, immunofixation, and free light chain assays, coupled with a comprehensive workup, including blood tests, bone marrow analysis (demonstrating ≥10% clonal plasma cells) [1,10], and sensitive imaging modalities, such as MRI, CT, or PET/CT, for bone lesions and marrow involvement [1,2]. According to the IMWG, diagnostic criteria include the presence of myeloma-defining events, such as those in the CRAB spectrum: Hypercalcemia, renal insufficiency, anemia, and bone lesions. The prognosis of MM depends on tumor load, biological disease features, and treatment responsiveness, utilizing systems, such as the revised ISS and Durie-Salmon staging, alongside cytogenetic analysis by FISH to identify high-risk chromosomal aberrations [4,11]. Management typically starts with triplet regimens, such as VRD, followed by ASCT for eligible individuals, complemented by consolidation and maintenance therapies to prolong remission. In cases of relapse, newer agents, such as carfilzomib and pomalidomide, often in triplet or quadruplet combinations, expand therapeutic possibilities [9]. Treatment is tailored according to fitness, risk category, and history of response, emphasizing the importance of structured surveillance. Our case was significant clinically because the patient had fulfilled the SP criteria with measurable M burden with abnormal serum-free kappa light chain. While causality cannot be inferred from a single case report. The importance of interpreting biochemical monoclonality when standard systemic criteria are not fulfilled is emphasized. We have monitored progression with available sensitive imaging in our case report with PET-CT multi-focal fluorodeoxyglucose avid lytic lesions, thus establishing solitary myeloma and prompt initiation of systemic therapy.

Table 1: Factors relating to early progression of solitary plasmacytoma to multiple myeloma.
MM exemplifies the evolving nature of disease management and the need for ongoing, individualized treatment strategies for optimal outcomes. His initial presentation with multiple lytic bone lesions and confirmation of neoplastic plasma cells warranted aggressive therapy with the VRD regimen, which achieved a significant metabolic response and near-complete resolution of active disease on imaging. The absence of hypercalcemia and significant cytopenia at diagnosis suggested an intermediate risk profile, and the normalization of laboratory findings further confirmed initial remission. However, the relapse detected through bone marrow biopsy highlights the persistent risk of disease recurrence in MM, necessitating vigilant surveillance and adaptation of therapy. Transition to a second-line regimen incorporating cyclophosphamide, bortezomib, acyclovir, and septran underscores the importance of versatility in therapeutic planning. Continued close monitoring through serial bone marrow assessments and advanced imaging will remain crucial to confirm response, guide further treatment, and optimizing prognosis battling this complex plasma cell malignancy.
Limitations
This case report is intended to highlight an important clinical message rather than correlate with population-level prognosis. Causality cannot be determined due to the observational nature of a case report and the absence of a comparison group. Advanced cytogenic profiling FISH was not available at the time of diagnosis and treatment of the case. Abnormal serum free light chain ratio was noted but serial evaluation from baseline was not uniformly performed. Management and follow-up was defined as per local institution protocols. Long term follow-up is needed to assess the durability of remission and survival outcomes. Imaging modalities evolved during follow-up, may introduce variability in lesion detection and disease burden assessment. Publication bias toward aggressive progressive patterns must be considered.
Early identification of high-risk SP through predictive risk stratification is essential, as biologically aggressive disease may necessitate rapid adaptations of therapy to manage progression and maintain remission.
References
- 1. Rajkumar SV, Dimopoulos MA, Palumbo A, Blade J, Merlini G, Mateos MV, et al. International myeloma working group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol 2014;15:e538-48. [Google Scholar] [PubMed]
- 2. Hillengass J, Usmani S, Rajkumar SV, Durie BG, Mateos MV, Lonial S, et al. International myeloma working group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol 2019;20:e302-12. [Google Scholar] [PubMed]
- 3. Palumbo A, Anderson K. Multiple myeloma. N Engl J Med 2011;364:1046-60. [Google Scholar] [PubMed]
- 4. Greipp PR, San Miguel J, Durie BG, Crowley JJ, Barlogie B, Bladé J, et al. International staging system for multiple myeloma. J Clin Oncol 2005;23:3412-20. [Google Scholar] [PubMed]
- 5. Kumar SK. Risk stratification in multiple myeloma: From systems to biology. Nat Rev Clin Oncol 2017;23:12-15 . [Google Scholar] [PubMed]
- 6. Sonneveld P. Genetics and outcomes of next-generation sequence-based MRD in multiple myeloma. Blood 2021;5:18-23 . [Google Scholar] [PubMed]
- 7. Attal M, Lauwers-Cances V, Hulin C, Leleu X, Caillot D, Escoffre M, et al. Lenalidomide, bortezomib, and dexamethasone with transplantation for myeloma. N Engl J Med 2017;376:1311-20. [Google Scholar] [PubMed]
- 8. Cavo M, Tacchetti P, Patriarca F, Petrucci MT, Pantani L, Galli M, et al. Bortezomib with thalidomide plus dexamethasone compared with thalidomide plus dexamethasone as induction therapy before, and consolidation therapy after, double autologous stem-cell transplantation in newly diagnosed multiple myeloma: a randomised phase 3 study. Lancet 2010;376:2075-85. [Google Scholar] [PubMed]
- 9. Richardson PG, Oriol A, Beksac M, Liberati AM, Galli M, Schjesvold F, et al. Pomalidomide, bortezomib, and dexamethasone for patients with relapsed or refractory multiple myeloma previously treated with lenalidomide (OPTIMISMM): A randomised, open-label, phase 3 trial. Lancet Oncol 2019;20:781-94. [Google Scholar] [PubMed]
- 10. Dispenzieri A, Kyle R, Merlini G, Miguel JS, Ludwig H, Hajek R, et al. International Myeloma Working Group guidelines for serum-free light chain analysis in multiple myeloma and related disorders. Leukemia 2009;23:215-24. [Google Scholar] [PubMed]
- 11. Palumbo A, Avet-Loiseau H, Oliva S, Lokhorst HM, Goldschmidt H, Rosinol L, et al. Revised international staging system for multiple myeloma: A report from international myeloma working group. J Clin Oncol 2015;33:2863-9. [Google Scholar] [PubMed]