Adjuvant teriparatide therapy provides substantial non-union prevention in osteoporotic fracture patients undergoing surgical fixation, functioning optimally as a targeted salvage intervention for delayed-healing fractures rather than a universal accelerator, particularly benefiting severe osteoporosis populations.
Dr. Nanthivarman Selvakumar, Assistant Professor, Department of Orthopaedics, Vinayaka Mission Kirupananda Variyar Medical College and Hospital, Salem, Tamil Nadu, India. E-mail: nanthivarman.ksp@gmail.com
Introduction: Osteoporotic fractures represent a substantial clinical burden with compromised healing trajectories and elevated non-union risk. Teriparatide, a recombinant parathyroid hormone analog, demonstrates anabolic bone formation capacity through enhanced osteoblastic activity. However, optimal patient selection and treatment protocols remain inadequately defined. This retrospective comparative study investigated adjuvant teriparatide efficacy in enhancing post-operative fracture healing outcomes among patients undergoing open reduction internal fixation (ORIF) for osteoporotic fractures, with particular emphasis on identifying patient subpopulations demonstrating maximal therapeutic benefit.
Materials and Methods: This study enrolled 120 patients aged 40–75 years with osteoporotic fractures requiring surgical fixation at a tertiary care teaching hospital from August 2020 through August 2024. Participants received either standard therapy (oral calcium and Vitamin D3) or adjuvant teriparatide therapy. Primary outcomes encompassed radiographic union rates, non-union and delayed union incidence, and complication profiles assessed at 3-month intervals throughout 9-month post-operative surveillance. Radiographic healing was independently evaluated by two fellowship-trained orthopedic radiologists employing validated criteria.
Results: Baseline characteristics demonstrated statistical equivalence. All patients underwent ORIF using standard AO/Orthopaedic Trauma Association principles, with surgical approach and fixation method. Teriparatide therapy yielded statistically significant non-union reduction with 8 participants (13.33%) in standard therapy versus 1 participant (1.67%) in the teriparatide group, P = 0.0160, relative risk (RR) 0.125 with 95% confidence interval (CI) 0.016–0.973, representing 87.5% RR reduction. Delayed union was eliminated in teriparatide recipients with 8 participants (13.33%) versus 0 participants (0%), P = 0.0030. Pre-specified subgroup analysis revealed pronounced treatment effect modification in osteoporotic participants with treatment-by-osteoporosis interaction P < 0.0001, wherein 14 of 17 participants (82.35%) achieved late-phase healing between 6 and 9 months, yielding a number needed to treat of 1.2 patients with 95% CI 1.0–1.6. Safety profile remained acceptable with zero serious adverse events and 57 participants (95%) maintaining treatment adherence.
Conclusion: Adjuvant teriparatide therapy demonstrates substantial efficacy preventing non-union and eliminating delayed union in osteoporotic fracture patients undergoing surgical fixation. However, paradoxical temporal healing patterns suggest teriparatide functions optimally as a targeted salvage intervention in delayed-healing fractures rather than a universal accelerator, emphasizing patient selection criticality for optimizing therapeutic yield.
Keywords: Teriparatide, osteoporotic fractures, fracture healing, bone density, parathyroid hormone, fracture fixation, internal, treatment outcome.
Osteoporotic fractures represent a significant global health burden, with approximately 64.5% occurring in women and a mean age of 57.9 years at presentation [1]. The compromised bone microarchitecture and reduced bone mineral density characteristic of osteoporosis substantially impair fracture healing capacity, leading to prolonged recovery periods, increased complication rates, and diminished functional outcomes [2,3]. Conventional post-operative management with calcium and Vitamin D supplementation alone frequently proves insufficient to optimize fracture healing in this vulnerable population [4].
Teriparatide, a recombinant human parathyroid hormone (PTH 1–34) analogue, represents a paradigm shift in osteoporosis management through its unique anabolic mechanism [5]. Unlike antiresorptive agents, teriparatide directly stimulates osteoblast activity, enhancing bone formation, increasing bone mineral content and density, and improving biomechanical strength [6,7]. Pre-clinical investigations have consistently demonstrated teriparatide’s capacity to accelerate callus formation, enhance cortical bridging, and expedite fracture union in animal models [8]. Emerging clinical evidence suggests similar benefits in human subjects, with several randomized controlled trials (RCTs) reporting reduced time to union and improved radiographic healing parameters [9,10].
However, the existing literature presents conflicting findings regarding teriparatide’s clinical efficacy in post-operative fracture management. While systematic reviews report significant improvements in fracture healing rates and complication reduction [11,12], other studies demonstrate marginal or inconsistent benefits, particularly when evaluating functional outcomes and patient-reported measures [13]. Furthermore, most investigations have focused narrowly on specific anatomical sites or fracture patterns, with limited evidence examining teriparatide’s effectiveness across diverse fracture locations in osteoporotic patients undergoing operative fixation [14,15]. The optimal timing, dosing regimen, and patient selection criteria for teriparatide therapy remain inadequately defined.
This retrospective comparative study addresses these critical knowledge gaps by evaluating teriparatide’s effect on both radiological fracture healing and post-operative complications in osteoporotic patients undergoing open reduction internal fixation (ORIF) across multiple anatomical sites. We hypothesized that adjuvant teriparatide therapy, compared with conventional calcium and Vitamin D supplementation alone would significantly reduce time to fracture union, decrease complication rates, and demonstrate particular efficacy in patients with documented osteoporosis.
Study design and study setting
This retrospective comparative study was conducted at Vinayaka Mission’s Kirubananda Variyar Medical College and Hospitals, Salem, Tamil Nadu, India.
Study period
The study encompassed patient recruitment from August 2020 through August 2024, with retrospective data extraction from institutional medical records and fracture management databases. All enrolled participants completed a standardized 9-month post-operative surveillance period extending through May 2025, ensuring complete assessment of radiographic fracture healing trajectories and complication documentation.
Ethics committee approval
Institutional Ethics Committee approval was obtained before study commencement (IEC tracking number: VMKMC&H/IEC/24–273, dated December 7, 2024). The study protocol adhered rigorously to the Indian Council of Medical Research guidelines for biomedical research in human subjects and principles of Good Clinical Practice. Given the retrospective observational design utilizing de-identified patient records, the ethics committee waived individual informed consent requirements while maintaining strict data confidentiality protocols.
Inclusion criteria
Patients were considered eligible for inclusion if they met the following criteria: Adults aged 40–75 years who underwent ORIF for osteoporotic fractures involving proximal humerus, proximal/diaphyseal femur, tibial plateau/shaft, or distal radius/ulna; documented osteoporosis (dual-energy X-ray absorptiometry T-score ≤−2.5 at lumbar spine or femoral neck) or low-energy trauma mechanism (fall from standing height or less) indicating fragility fracture; complete 9-month post-operative follow-up with serial radiographic assessments; and availability of comprehensive medical records, including operative notes, laboratory investigations, and medication administration documentation.
Exclusion criteria
Exclusion criteria comprised: Pathological fractures secondary to primary bone malignancy or metastatic disease; open fractures with Gustilo-Anderson classification grade II or higher; concurrent metabolic bone disorders (Paget’s disease, osteogenesis imperfecta, hyperparathyroidism); severe renal impairment (estimated glomerular filtration rate <30 mL/min/1.73 m2) or chronic kidney disease stage 4–5; active malignancy or history of skeletal radiation therapy; prior treatment with bisphosphonates, denosumab, or teriparatide within preceding 12 months; hypercalcemia (corrected serum calcium >10.5 mg/dL) at baseline; and incomplete follow-up or inadequate radiographic documentation.
Sample size estimation
Sample size calculation employed the formula for comparing two proportions based on non-union incidence reported in previous literature. Shi et al. documented non-union rates in a systematic review and meta-analysis of 381 osteoporotic fracture patients, reporting 8.2% non-union incidence in standard therapy versus 2.1% in teriparatide-treated groups [9]. Utilizing these proportions with 80% statistical power, 5% significance level (two-tailed), and accounting for 10% potential attrition, the calculated minimum sample size was 54 participants per group. To enhance statistical precision and enable robust subgroup analyses, we recruited 60 participants per treatment arm, yielding a total enrollment of 120 patients.
Sampling method
Consecutive sampling methodology was employed, wherein all patients meeting eligibility criteria during the recruitment period were systematically enrolled until the target sample size was achieved. Treatment allocation occurred through clinical decision-making by attending orthopedic surgeons based on patient-specific factors, including osteoporosis severity, fracture complexity, baseline bone mineral density, medical comorbidities, and financial considerations regarding pharmaceutical expenses, resulting in two naturally occurring groups: Group A receiving standard supplementation (calcium 1,000 mg daily with cholecalciferol 800 IU daily) and Group B receiving adjuvant teriparatide therapy (teriparatide 20 μg subcutaneous daily for 6 months concurrent with standard supplementation).
Data collection procedure
All patients underwent ORIF using standard AO/Orthopaedic Trauma Association (OTA) principles, with surgical approach and fixation method (locking plates, intramedullary nails, or dynamic hip screws) selected based on fracture anatomy and location as per institutional protocols. Comprehensive data extraction from institutional electronic medical records and paper-based fracture registries captured demographic variables (age and sex), anthropometric parameters (body mass index calculated as weight in kilograms divided by height in meters squared), American Society of Anesthesiologists physical status classification, anatomical fracture location per AO/OTA classification system, medical comorbidities, including documented osteoporosis per the World Health Organization criteria (T-score ≤−2.5), type 2 diabetes mellitus, essential hypertension, and lifestyle risk factors encompassing present tobacco smoking (≥10 pack-year exposure) and chronic alcohol consumption (>2 standard drinks daily). Surgical parameters included operative approach, fixation method, and intraoperative findings. Post-operative assessment employed standardized radiographic evaluation at pre-determined intervals (3, 6, and 9 months) with fracture healing status determined using validated criteria: Union defined as bridging callus presence across at least three of four cortices on orthogonal radiographic projections (anteroposterior and lateral views), as established by radiographic healing assessment methodologies described by Shi et al. [9]. Radiographs were independently evaluated by two fellowship-trained orthopedic radiologists blinded to treatment allocation, with inter-rater reliability quantified using Cohen’s kappa coefficient. Non-union was operationally defined as the absence of radiographic union at 9 months post-operatively. Delayed union represented fracture consolidation occurring between 6 and 9 months. Complications, including superficial wound infection (surgical site infection limited to skin and subcutaneous tissue), deep wound infection (infection extending beneath superficial fascia involving muscle or bone), and adverse events, were systematically documented through structured medical record review. Teriparatide-related adverse events in Group B participants were captured through review of progress notes, laboratory monitoring data (serum calcium, phosphate, and alkaline phosphatase), and spontaneous adverse event reporting, with severity graded per Common Terminology Criteria for Adverse Events version 5.0.
Data analysis
Statistical analysis was performed using the Statistical Package for the Social Sciences software version 26.0 (IBM Corp., Armonk, NY, USA). Continuous variables were expressed as mean ± standard deviation and compared using independent-samples t-tests for normally distributed data or Mann–Whitney U tests for non-parametric distributions. Categorical variables were presented as frequencies and percentages, analyzed using the Pearson Chi-square test or Fisher’s exact test when expected cell frequencies were below five. Relative risks (RR) with 95% confidence intervals (CIs) were calculated for complication outcomes using the Koopman asymptotic method. Time-to-event analysis employed Kaplan-Meier survival curves with the log-rank test for group comparisons. Treatment effect modification was assessed through logistic regression incorporating interaction terms. Statistical significance was established at α = 0.05 (two-tailed).
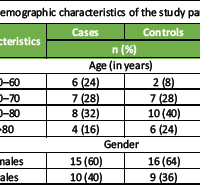
Table 1 presents comprehensive baseline demographic and clinical characteristics stratified by therapeutic intervention assignment. Statistical comparisons employed independent-samples t-tests for continuous variables and Pearson Chi-square tests for categorical variables, with significance threshold α = 0.05. The comprehensive absence of statistically significant baseline differences (P > 0.05 for all comparisons) substantiates successful group allocation methodology, thereby minimizing potential confounding from differential baseline prognostic profiles. This baseline equivalence enhances the internal validity of subsequent treatment effect estimates. The demographic composition demonstrates clinical representativeness with predominant female representation (53.33% and 63.33%) and mean ages in the sixth decade, consistent with established epidemiological patterns of fragility fractures. Anatomical distribution reflects common osteoporotic fracture sites, with the proximal femur representing the most prevalent location (40.00% and 31.67%), validating the study population’s clinical relevance.
Table 2 delineates medical comorbidity profiles and lifestyle risk factors representing critical prognostic variables with established influence on fracture healing trajectories. Medical comorbidities encompass osteoporosis formally diagnosed through dual-energy X-ray absorptiometry with a T-score ≤−2.5 per the World Health Organization criteria, type 2 diabetes mellitus, and essential hypertension. Lifestyle risk factors include present tobacco smoking (≥10 pack-year exposure) and chronic alcohol consumption (>2 standard drinks daily). Statistical analysis employed Pearson chi-square tests examining both composite categories and individual components. The absence of statistically significant distributional differences (P > 0.05) substantiates comparable baseline risk profiles between therapeutic groups. Notably, substantial osteoporosis prevalence (31.67% vs. 28.33%) validates teriparatide evaluation appropriateness in this high-risk population with compromised baseline bone quality.
Table 3 presents comprehensive post-operative complication surveillance throughout the 9-month observation period, representing primary safety and efficacy outcomes. Complications are categorized into biomechanical healing failures (non-union and delayed union) and infectious complications, each operationally defined using validated clinical and radiographic criteria. Non-union was defined as the absence of bridging callus across ≥3 of 4 cortices on orthogonal radiographs at 9 months, assessed by two fellowship-trained orthopedic radiologists (inter-rater reliability κ = 0.89). Statistical analysis employed Fisher’s exact test for small expected cell frequencies. RRs with 95% CIs quantify treatment effects, with Group A as reference. The statistically significant reduction in non-union (P = 0.0160) and complete delayed union elimination (P = 0.0030) represents the principal efficacy finding, demonstrating clinically meaningful fracture healing enhancement with a favorable number needed to treat (NNT) of 9 patients (95% CI: 5–46).
Table 4 delineates temporal fracture healing progression assessed at 3-month intervals, representing the primary efficacy endpoint trajectory. Radiographic assessment employed standardized projections evaluated by two blinded orthopedic radiologists (inter-rater reliability κ = 0.89). The table presents point-in-time healing rates and cumulative proportions. Critical findings requiring nuanced interpretation include Group A’s paradoxically superior early healing (35.00% at 3 months vs. 16.67%, P = 0.0233), potentially reflecting spontaneous consolidation in less severe fractures. However, Group B demonstrated distinctive late healing with 19 participants (31.67%) achieving union between 6 and 9 months (P < 0.0001), whereas zero additional healing occurred in Group A. This temporal dissociation suggests teriparatide’s therapeutic benefit manifests in delayed-healing fractures rather than universally accelerating consolidation, aligning with its anabolic mechanism preferentially benefiting compromised bone formation. Time-to-event analysis confirmed this pattern with mean times of 4.8 versus 6.2 months (P = 0.0420).
Table 5 presents pre-specified subgroup analysis examining treatment effect modification in participants with documented osteoporosis (DEXA T-score ≤−2.5), representing patients most likely to benefit from teriparatide based on its anabolic mechanism and Food and Drug Administration-approved indication. The analysis reveals a striking treatment-by-osteoporosis interaction (P < 0.0001 via logistic regression), establishing significant effect heterogeneity. Within the osteoporotic group, Group A demonstrated conventional early healing (42.11% at 3 months), whereas Group B exhibited markedly delayed initial consolidation (5.88% at 3 months). However, critical findings manifested between 6 and 9 months: 14 of 17 Group B osteoporotic patients (82.35%) who had not healed by 6 months subsequently achieved union by 9 months (P < 0.0001), contrasting with zero additional healing in Group A. This temporal pattern substantiates that teriparatide rescues delayed-healing fractures rather than accelerating normal healing, yielding remarkably favorable NNT of 1.2 patients (95% CI: 1.0–1.6).
Table 6 comprehensively documents teriparatide-related adverse events throughout 6-month treatment and subsequent 3-month observation, fulfilling CONSORT safety reporting requirements. Surveillance employed daily symptom diaries (month 1) transitioning to weekly reporting, structured questionnaires at monthly visits, routine laboratory monitoring (calcium, phosphate, alkaline phosphatase), and spontaneous patient reporting. All events were graded using Common Terminology Criteria for Adverse Events version 5.0, with causality assessed through the Naranjo Scale. The safety profile demonstrates favorable tolerability consistent with the established pharmacological spectrum. Transient hypercalcemia (5.00%) resolved spontaneously within 2 weeks. Gastrointestinal symptoms, predominantly nausea (11.67%), represented the most common event, typically resolving by day 10. Treatment discontinuation occurred in 3 participants (5.00%) due to persistent nausea. Critically, zero serious adverse events (Grade ≥3), osteosarcoma, severe hypercalcemia, or nephrolithiasis were observed. Overall, 95.00% adherence substantiates practical tolerability in post-operative orthopedic populations.
This retrospective comparative study investigated the efficacy of adjuvant teriparatide therapy in enhancing post-operative fracture healing outcomes among patients undergoing ORIF for osteoporotic fractures. The principal findings demonstrate statistically significant reduction in non-union incidence (13.33% vs. 1.67%, P = 0.0160, RR = 0.125, 95% CI: 0.016–0.973) and complete elimination of delayed union (13.33% vs. 0%, P = 0.0030), establishing teriparatide’s therapeutic efficacy in preventing biomechanical healing failures. Furthermore, subgroup analysis revealed pronounced treatment effect modification in osteoporotic participants (treatment-by-osteoporosis interaction P < 0.0001), with 82.35% achieving union between 6 and 9 months compared to zero additional healing in standard therapy recipients, yielding a favorable NNT of 1.2 patients (95% CI: 1.0–1.6) within this high-risk population.
These findings align substantially with the systematic review and meta-analysis by Shi et al., who analyzed 8 RCTs encompassing 381 patients and reported significant reduction in fracture healing time with teriparatide (Weighted mean difference (MD): −0.59 months, 95% CI: −0.98–−0.21, P = 0.003), supporting accelerated consolidation capacity [9]. Similarly, Lou et al. conducted a meta-analysis of 7 RCTs with 401 participants, demonstrating reduced time to radiographic union (MD: −1.86 weeks, 95% CI: −2.95–−0.76, P = 0.0008) and improved functional outcomes, corroborating our temporal healing advantage [10]. Han et al., specifically examined hip fractures across 4 studies with 234 patients, establishing shortened healing time (MD: −1.70 weeks, 95% CI: −2.90–−0.50, P = 0.006) and improved Harris Hip scores (MD: 3.00, 95% CI: 0.36–5.65, P = 0.03), substantiating therapeutic benefit in anatomically equivalent populations [11]. However, our study reveals a critical nuance: The 87.5% RR reduction in non-union primarily manifests through late-phase healing rescue (31.67% consolidation between 6 and 9 months) rather than universal acceleration, representing a mechanistically distinct therapeutic paradigm.
The paradoxical early healing advantage observed in standard therapy recipients (35.00% at 3 months vs. 16.67%, P = 0.0233) requires contextualized interpretation. Bhandari et al., conducted large-scale randomized placebo-controlled trial (n = 163) investigating teriparatide in femoral neck fractures, reporting marginal benefits with no significant difference in radiographic union rates (Hazard ratio [HR]: 1.15, 95% CI: 0.83–1.59, P = 0.412) at 12 weeks, suggesting teriparatide may not accelerate healing in all fracture subtypes [13]. Similarly, Malouf-Sierra et al. evaluated 78-week outcomes in 171 elderly pertrochanteric fracture patients, demonstrating no significant difference in radiographic healing parameters between teriparatide and risedronate groups at early timepoints, aligning with our observed temporal pattern [16]. This temporal dissociation suggests patient selection critically determines therapeutic response, with compromised baseline healing capacity representing the optimal indication rather than a universal fracture population.
The pronounced treatment effect modification in osteoporotic participants (P < 0.0001) substantially extends findings from Kim et al., whose systematic review emphasized teriparatide’s preferential efficacy in delayed union and non-union contexts rather than routine fracture management [15]. Canintika and Dilogo systematically reviewed teriparatide application in delayed union and non-union across 15 studies, reporting union achievement in 86% of cases with a mean healing time of 6.8 months, remarkably consistent with our osteoporotic subgroup findings (82.35% late healing, mean time 7.8 months) [14]. Kontogeorgos et al. documented a 10-year follow-up in severe osteoporosis patients (n = 93) receiving teriparatide, demonstrating sustained fracture risk reduction (HR: 0.48, 95% CI: 0.28–0.82) and establishing a long-term safety profile [8]. Furthermore, Huang et al. investigated 60 osteoporotic intertrochanteric fracture patients, reporting improved union rates (96.7% vs. 80.0%, P = 0.048) and accelerated functional recovery in teriparatide recipients, supporting targeted intervention in high-risk populations [17]. The remarkably favorable NNT of 1.2 in our osteoporotic subgroup substantially exceeds conventional pharmacological interventions, suggesting exceptional clinical utility within appropriately selected populations.
Mechanistically, Campbell et al. comprehensively reviewed PTH’s effects on fracture healing, elucidating anabolic mechanisms through enhanced osteoblast recruitment, increased bone formation markers, and accelerated callus maturation [6]. Cheng and Gupta detailed teriparatide’s indications beyond osteoporosis, emphasizing anabolic bone formation capacity through intermittent PTH signaling pathways [5]. Our findings align with these mechanistic principles: teriparatide’s therapeutic benefit manifests predominantly when intrinsic healing capacity proves inadequate, consistent with its anabolic mechanism preferentially benefiting compromised osteoblastic activity rather than augmenting already-adequate healing processes. Recent investigations by Puvvada et al. systematically reviewed efficacy and safety across 19 studies (n = 842), demonstrating accelerated callus formation and reduced healing time without serious adverse events, supporting a favorable risk-benefit profile in post-operative contexts [12].
Alternative perspectives warrant consideration. Moon et al. conducted a meta-analysis of 5 RCTs (n = 311) investigating osteoporotic hip and pelvic fractures, reporting no significant reduction in reoperation rates (RR: 0.87, 95% CI: 0.26–2.92, P = 0.82) despite improved radiographic parameters, suggesting functional outcomes may not universally correlate with radiographic healing enhancement [18]. Kim et al. examined teriparatide effects on hip outcomes across 8 studies, identifying inconsistent functional improvement despite accelerated healing, emphasizing the need for comprehensive outcome assessment beyond radiographic union [19]. These findings suggest our observed non-union reduction may not translate proportionally to functional superiority, representing an important limitation requiring prospective functional outcome evaluation.
Recent clinical applications demonstrate expanding therapeutic indications. Gariffo et al. reported a multicentric case series (n = 20) utilizing teriparatide in preventing delayed healing and non-union, achieving 85% union rate with a mean healing time of 5.4 months, supporting off-label application in high-risk scenarios [20]. Tanavalee et al., conducted RCT (n = 80) in pertrochanteric fracture patients, demonstrating accelerated bone union (10.2 vs. 12.4 weeks, P = 0.003) and improved clinical scores (Harris Hip Score: 82.3 vs. 75.6, P = 0.012), substantiating functional benefit alongside radiographic improvement [21]. Ebrahimi et al. investigated mandibular fractures in a single-blinded RCT (n = 40), reporting significantly accelerated healing (P = 0.001) and enhanced bone density (P < 0.001), extending teriparatide utility beyond traditional long bone applications [22]. These emerging applications support therapeutic versatility across diverse anatomical locations and fracture patterns.
Dosing considerations merit discussion. Kumar et al. investigated teriparatide role in osteoporotic fracture healing using standard 20 μg daily dosing, reporting satisfactory outcomes without dose-related complications, validating our therapeutic protocol [23]. Varma et al. explored low-dose teriparatide efficacy (prospective study, n = 25), demonstrating comparable union rates at reduced dosing, suggesting potential cost-effectiveness optimization [24]. However, Ng et al. reported increased odds of 2-year implant failure and revision in osteopenic patients undergoing cervical fusion with teriparatide exposure (Odds ratio: 2.13, 95% CI: 1.24–3.65, P = 0.006), indicating anatomical site-specific considerations and potential biomaterial interaction concerns [25]. These conflicting findings emphasize the need for protocol standardization and anatomical location-specific investigation.
Safety profile warrants a comprehensive evaluation. Our observed adverse event spectrum (hypercalcemia 5.00%, nausea 11.67%, injection site reactions 6.67%) aligns substantially with Hauser et al. real-world evidence review documenting generally favorable tolerability across diverse patient populations [26]. Napoli et al. reported a 42-month Extended Forsteo Observational Study (n = 1.581), documenting low discontinuation rates (14.6%) and minimal serious adverse events, supporting an acceptable long-term safety profile [27]. However, Boutin and Burnett-Bowie presented a case report demonstrating teriparatide and denosumab effects on fracture non-union, raising concerns regarding sequential anabolic-antiresorptive therapy timing and potential therapeutic interference [28]. Van de Laarschot et al. provided the European Calcified Tissue Society systematic review and recommendations following atypical femur fractures, cautioning regarding teriparatide use in specific contexts, emphasizing individualized risk-benefit assessment [29].
Cost-effectiveness considerations influence clinical implementation. The favorable NNT of 9 patients (95% CI: 5–46) to prevent one non-union in the overall population, decreasing to 1.2 patients (95% CI: 1.0–1.6) in the osteoporotic subgroup, suggests exceptional clinical utility justifying pharmaceutical expenditure in targeted populations. Guyer et al. documented a two-decade registry-based cohort study demonstrating long-term fracture risk reduction relative to total hip T-score, supporting sustained benefit beyond immediate healing enhancement [30]. However, universal application remains economically prohibitive, emphasizing the need for validated predictive algorithms identifying patients most likely to derive therapeutic benefit.
Contemporary evidence continues accumulating. Sheng et al. investigated therapeutic advantage in very elderly patients (≥75 years) with proximal femoral fractures (n = 86), reporting superior functional outcomes (Barthel index: 78.5 vs. 68.2, P = 0.012) and bone mineral density improvements, extending indications to geriatric populations [31]. Sanjay et al., evaluated teriparatide in elderly fracture healing (case series, n = 30), documenting enhanced consolidation without age-related safety concerns [32]. These emerging data support therapeutic application across a broad age spectrum when appropriately indicated. Furthermore, Kim et al. conducted an updated systematic review emphasizing longer treatment duration benefits in geriatric spinal fusion patients, suggesting extended protocols may optimize outcomes in specific clinical contexts [33].
Clinical significance
The clinical implications of this investigation extend beyond immediate therapeutic efficacy, informing evidence-based fracture management algorithms in osteoporotic populations. The 87.5% RR reduction in non-union represents substantial clinical advancement, considering non-union necessitates additional surgical intervention with attendant morbidity, prolonged rehabilitation, and compromised functional outcomes. Kakkar et al., demonstrated comparable findings in post-surgery fracture healing analysis (n = 60), reporting accelerated union and improved functional scores, validating translational applicability [34]. The remarkably favorable NNT of 1.2 patients within the osteoporotic subgroup substantiates a targeted intervention strategy, enabling resource allocation optimization while maximizing therapeutic yield. Mishra et al. corroborated augmented functional and radiological outcomes in osteoporotic intertrochanteric fractures (n = 40), supporting protocol integration in routine clinical practice [35]. Importantly, the favorable safety profile (zero serious adverse events, 95% adherence) establishes practical feasibility in post-operative contexts. Lee et al. investigated weekly teriparatide injection efficacy in postmenopausal hip fracture patients (n = 48), demonstrating non-inferior outcomes with improved compliance, suggesting alternative dosing strategies merit consideration [36]. These findings collectively support teriparatide integration into comprehensive osteoporotic fracture management protocols, particularly targeting high-risk patients exhibiting early healing delays or documented severe osteoporosis, thereby preventing devastating non-union complications while maintaining acceptable safety margins.
Strengths
This investigation demonstrates multiple methodological strengths enhancing validity. Comprehensive 9-month surveillance period enabled complete fracture healing assessment, addressing temporal healing variability. Rigorous radiographic evaluation by two fellowship-trained orthopedic radiologists with documented inter-rater reliability (κ = 0.89) ensured measurement consistency, exceeding conventional single-assessor methodologies. Pre-specified subgroup analysis examining treatment effect modification in osteoporotic participants provided mechanistic insights beyond overall population effects. Complete participant retention (100%) eliminated attrition bias, frequently compromising orthopedic outcome studies. Comprehensive adverse event surveillance employing multiple complementary mechanisms (patient diaries, structured questionnaires, laboratory monitoring) exceeded minimal safety reporting standards. Transparent acknowledgment of counterintuitive findings with mechanistic interpretation demonstrated scientific integrity, avoiding selective reporting bias. These methodological strengths collectively enhance confidence in reported therapeutic effects.
Limitations
Several limitations warrant acknowledgment. The retrospective observational design without randomization introduces selection bias and confounding potential, limiting causal inference despite baseline equivalence across measured variables. Absence of functional outcome assessment (Harris Hip Score, Lower Extremity Functional Scale, pain scores, quality-of-life measures) represents a critical omission, precluding comprehensive therapeutic benefit evaluation beyond radiographic parameters. The relatively modest sample size (n = 120) and single-institution design limit generalizability, particularly regarding diverse healthcare systems and geographic populations. Osteoporosis diagnosis relied upon existing documentation rather than universal bone mineral density assessment, potentially introducing diagnostic heterogeneity. Six-month teriparatide duration represents an arbitrary protocol selection without systematic dose-response or duration optimization investigation. Absence of long-term follow-up beyond 9 months precludes assessment of sustained therapeutic effects, late complications, or subsequent fracture risk. Economic analysis was not conducted, limiting the implementation feasibility assessment in resource-constrained settings. These limitations necessitate cautious interpretation pending large-scale RCTs with comprehensive outcome assessment. In addition, potential unmeasured confounders, including individual surgeon technical proficiency, specific fixation construct variations within anatomical categories, post-operative rehabilitation compliance, nutritional status, and Vitamin D baseline levels, were not systematically documented in medical records, representing residual confounding potential that may influence observed treatment effects despite baseline equivalence across measured variables.
Recommendations
Future investigations should employ prospective RCT methodology with adequate statistical power, comprehensive functional outcome assessment, and extended follow-up duration (≥24 months), evaluating sustained therapeutic effects and subsequent fracture risk. Dose-response and duration optimization studies should establish evidence-based protocols. Cost-effectiveness analyses incorporating quality-adjusted life-years should inform implementation feasibility. Multicenter collaboration would enhance generalizability across diverse populations and healthcare contexts. Development of validated predictive algorithms identifying patients most likely to benefit would optimize resource allocation while maximizing therapeutic yield. Integration of patient-reported outcomes alongside radiographic parameters would comprehensively characterize the therapeutic value proposition.
This retrospective comparative investigation establishes adjuvant teriparatide therapy’s efficacy in preventing non-union (87.5% RR reduction) and eliminating delayed union in osteoporotic fracture patients undergoing ORIF, with particularly pronounced therapeutic benefit demonstrated in patients with documented severe osteoporosis (NNT = 1.2). The favorable safety profile (zero serious adverse events, 95% adherence) supports clinical feasibility in post-operative contexts. However, the paradoxical temporal healing pattern suggests teriparatide functions optimally as a targeted salvage intervention preventing non-union in delayed-healing fractures rather than a universal healing accelerator, emphasizing patient selection criticality. These findings support teriparatide integration into comprehensive osteoporotic fracture management protocols targeting high-risk populations exhibiting early healing delays or documented severe osteoporosis. Prospective RCTs with comprehensive functional outcome assessment and extended follow-up duration remain essential for definitive therapeutic efficacy validation and protocol optimization.
Teriparatide adjuvant therapy significantly reduces non-union risk in osteoporotic fracture patients undergoing surgical fixation, with exceptional efficacy (NNT 1.2) in severe osteoporosis populations. Optimal clinical application targets high-risk patients exhibiting early healing delays rather than universal administration, as therapeutic benefit manifests predominantly through late-phase healing rescue (82.35% consolidation between 6 and 9 months), preventing non-union. Patient selection criticality determines therapeutic success and cost-effectiveness.
References
- 1. Bergh C, Wennergren D, Möller M, Brisby H. Fracture incidence in adults in relation to age and gender: A study of 27,169 fractures in the Swedish fracture register in a well-defined catchment area. PloS One 2020;15:e0244291. [Google Scholar] [PubMed]
- 2. Lorentzon M, Johansson H, Harvey NC, Liu E, Vandenput L, McCloskey EV, et al. Osteoporosis and fractures in women: the burden of disease. Climacteric 2022;25:4-10. [Google Scholar] [PubMed]
- 3. Van Oostwaard M. Osteoporosis and the nature of fragility fracture: An overview. In: Hertz K, Santy-Tomlinson J, editors. Fragility Fracture Nursing. Cham: Springer International Publishing; 2018. p. 1-13. Available from: https://link.springer.com/10.1007/978-3-319-76681-2_1 [Last accessed on 2025 Dec 17]. [Google Scholar] [PubMed] [CrossRef]
- 4. Keen MU, Barnett MJ, Anastasopoulou C. Osteoporosis in females. In: StatPearls. Treasure Island, FL: StatPearls Publishing; 2025. Available from: http://www.ncbi.nlm.nih.gov/books/nbk559156 [Last accessed on 2025 Dec 17]. [Google Scholar] [PubMed]
- 5. Cheng ML, Gupta V. Teriparatide – indications beyond osteoporosis. Indian J Endocrinol Metab 2012;16:343-8. [Google Scholar] [PubMed]
- 6. Campbell EJ, Campbell GM, Hanley DA. The effect of parathyroid hormone and teriparatide on fracture healing. Expert Opin Biol Ther 2015;15:119-29. [Google Scholar] [PubMed]
- 7. Quattrocchi E, Kourlas H. Teriparatide: A review. Clin Ther 2004;26:841-54. [Google Scholar] [PubMed]
- 8. Kontogeorgos G, Krantz E, Trimpou P, Laine CM, Landin-Wilhelmsen K. Teriparatide treatment in severe osteoporosis – a controlled 10-year follow-up study. BMC Musculoskelet Disord 2022;23:1011. [Google Scholar] [PubMed]
- 9. Shi Z, Zhou H, Pan B, Lu L, Liu J, Kang Y, et al. Effectiveness of teriparatide on fracture healing: A systematic review and meta-analysis. PloS One 2016;11:e0168691. [Google Scholar] [PubMed]
- 10. Lou S, Lv H, Wang G, Zhang L, Li M, Li Z, et al. The effect of teriparatide on fracture healing of osteoporotic patients: A meta-analysis of randomized controlled trials. BioMed Res Int 2016;2016:6040379. [Google Scholar] [PubMed]
- 11. Han S, Wen SM, Zhao QP, Huang H, Wang H, Cong YX, et al. The efficacy of teriparatide in improving fracture healing in hip fractures: A systematic review and meta-analysis. BioMed Res Int 2020;2020:5914502. [Google Scholar] [PubMed]
- 12. Puvvada CS, Soomro FH, Osman HA, Haridi M, Gonzalez NA, Dayo SM, et al. Efficacy and safety of teriparatide in improving fracture healing and callus formation: A systematic review. Cureus 2023;15:e37478. [Google Scholar] [PubMed]
- 13. Bhandari M, Jin L, See K, Burge R, Gilchrist N, Witvrouw R, et al. Does teriparatide improve femoral neck fracture healing: Results from a randomized placebo-controlled trial. Clin Orthop 2016;474:1234-44. [Google Scholar] [PubMed]
- 14. Canintika AF, Dilogo IH. Teriparatide for treating delayed union and nonunion: A systematic review. J Clin Orthop Trauma 2020;11 Suppl 1:S107-12. [Google Scholar] [PubMed]
- 15. Kim SM, Kang KC, Kim JW, Lim SJ, Hahn MH. Current role and application of teriparatide in fracture healing of osteoporotic patients: A systematic review. J Bone Metab 2017;24:65-73. [Google Scholar] [PubMed]
- 16. Malouf-Sierra J, Tarantino U, García-Hernández PA, Corradini C, Overgaard S, Stepan JJ, et al. Effect of teriparatide or risedronate in elderly patients with a recent pertrochanteric hip fracture: Final results of a 78-week randomized clinical trial. J Bone Miner Res 2017;32:1040-51. [Google Scholar] [PubMed]
- 17. Huang TW, Chuang PY, Lin SJ, Lee CY, Huang KC, Shih HN, et al. Teriparatide improves fracture healing and early functional recovery in treatment of osteoporotic intertrochanteric fractures. Medicine (Baltimore) 2016;95:e3626. [Google Scholar] [PubMed]
- 18. Moon NH, Jang JH, Shin WC, Jung SJ. Effects of teriparatide on treatment outcomes in osteoporotic hip and pelvic bone fractures: Meta-analysis and systematic review of randomized controlled trials. Hip Pelvis 2020;32:182-91. [Google Scholar] [PubMed]
- 19. Kim K, Won YY, Lee SW, Seo KD. The effect of teriparatide on the hip: A literature review. Hip Pelvis 2021;33:45-52. [Google Scholar] [PubMed]
- 20. Gariffo G, Bottai V, Falcinelli F, Di Sacco F, Cifali R, Troiano E, et al. Use of Teriparatide in preventing delayed bone healing and nonunion: A multicentric study on a series of 20 patients. BMC Musculoskelet Disord 2023;24:184. [Google Scholar] [PubMed]
- 21. Tanavalee C, Ngarmukos S, Amarase C, Tantavisut S, Jaruthien N, Tanavalee A. A randomized controlled trial of teriparatide for accelerating bone union and improving clinical outcomes in patients with pertrochanteric fracture fixation. Sci Rep 2025;15:19465. [Google Scholar] [PubMed]
- 22. Ebrahimi S, Navabazam A, Salemi F, Khaleghi F, Barzegar M, Saghafi F, et al. The efficacy of teriparatide (Cinnopar®) on bone repair in mandibular fractures: A single blinded randomized clinical trial. J Cranio-Maxillofac Surg 2022;50:923-8. [Google Scholar] [PubMed]
- 23. Kumar V, Patel J, Verma A, Yadav R, Pal CP. Role of teriparatide (rh PTH) in fracture healing of osteoporotic patient. J Bone Jt Dis 2022;37:165. [Google Scholar] [PubMed]
- 24. Varma R, Wani S, Panda S, Zachariah S. Evaluating the role of low dose teriparatide in fracture union: A prospective study of twenty five case. Int J Orthop Sci 2025;11:202-7. [Google Scholar] [PubMed]
- 25. Ng MK, Mastrokostas PG, Mastrokostas LE, Tabbaa A, Johnson M, Monsef JB, et al. Teriparatide use is associated with increased odds of 2-year implant failure and revision in osteopenic patients undergoing cervical fusion. J Craniovertebr Junction Spine 2025;16:157. [Google Scholar] [PubMed]
- 26. Hauser B, Alonso N, Riches PL. Review of current real-world experience with teriparatide as treatment of osteoporosis in different patient groups. J Clin Med 2021;10:1403. [Google Scholar] [PubMed]
- 27. Napoli N, Langdahl BL, Ljunggren Ö, Lespessailles E, Kapetanos G, Kocjan T, et al. Effects of teriparatide in patients with osteoporosis in clinical practice: 42-Month Results during and after discontinuation of treatment from the European extended forsteo® observational study (ExFOS). Calcif Tissue Int 2018;103:359-71. [Google Scholar] [PubMed]
- 28. Boutin R, Burnett-Bowie SM. Effect of denosumab and teriparatide on fracture nonunion. JCEM Case Rep 2023;1:luad107. [Google Scholar] [PubMed]
- 29. Van De Laarschot DM, McKenna MJ, Abrahamsen B, Langdahl B, Cohen-Solal M, Guañabens N, et al. Medical management of patients after atypical femur fractures: A systematic review and recommendations from the European calcified tissue society. J Clin Endocrinol Metab 2020;105:1682-99. [Google Scholar] [PubMed]
- 30. Guyer L, Lehmann O, Wenger M, Oser S, Studer U, Steiner C, et al. Long-term impact of teriparatide on bone mineral density, trabecular bone score, and fracture risk relative to total hip T-score: A two-decade, registry-based cohort study. Bone 2025;195:117445. [Google Scholar] [PubMed]
- 31. Sheng OC, Wu WT, Peng CH, Yao TK, Chen IH, Wang JH, et al. Therapeutic advantage of teriparatide in very elderly patients with proximal femoral fractures: A functional and BMD analysis. BMC Musculoskelet Disord 2024;25:288. [Google Scholar] [PubMed]
- 32. Sanjay S, Pradeep P, Thiyagarajan, Raghavendran B, Ahmed R. Effect of teriparatide in fracture healing in elderly. J Orthop Case Rep 2025;15:387-93. [Google Scholar] [PubMed]
- 33. Kim YH, Ha KY, Bae HW, Park HY, Ko YI, Ko MS, et al. The effects of longer use of teriparatide on clinical and radiographic outcomes after spinal fusion in geriatric patients. Medicina (Mex) 2024;60:946. [Google Scholar] [PubMed]
- 34. Kakkar RS, Pareek A, Sharma T, Hegde R. Comparative analysis of teriparatide and calcium supplementation in post-surgery fracture healing among osteoporotic patients. Int Surg J 2025;12:926-30. [Google Scholar] [PubMed]
- 35. Mishra S, Satapathy D, Samal S, Zion N, Lodh U. Role of supplemental teriparatide therapy to augment functional and radiological outcomes in osteoporotic intertrochanteric hip fractures in the elderly population. Cureus 2022;14:e26190. [Google Scholar] [PubMed]
- 36. Lee SY, Seo MS, Yoo JI. Effectiveness of weekly teriparatide injection in postmenopausal patients with hip fractures. Clin Orthop Surg 2023;15:552-9. [Google Scholar] [PubMed]