The study demonstrates that “Failed Back Surgery Syndrome” is not a single diagnosis but a complex symptom cluster. It highlights the necessity of advanced imaging (CT-myelography or dynamic radiographs) to pinpoint the exact failure point of the primary fixation before attempting revision.
Dr. Vipin Kumar Mishra, Department of Orthopaedics, Government Medical College, Satna, Madhya Pradesh, India. E-mail: vipin9926@gmail.com
Introduction: Low back pain (LBP) ranks among the most common and debilitating musculoskeletal disorders worldwide, affecting about 9.4% of the population and imposing substantial social, economic, and psychological burdens. The aging population has led to a significant rise in chronic LBP and spinal surgical interventions. Nearly 10% of patients continue to experience symptoms beyond 3 months. The biomechanics of the lumbar spine, especially changes in the neutral zone, play a critical role in contributing to instability and pain. Despite advancements in surgical techniques, the success rates of repeat spine surgeries are declining. The objective of the above study was to evaluate the cause and treatment of failed back surgery.
Materials and Methods: This prospective observational analysis was conducted at a tertiary care teaching hospital from January 2021 to June 2022. The study included 26 adults with failed back surgery syndrome (FBSS) following lumbar fixation.
Results: The straight leg raise test (SLRT) showed significant improvement in most patients over time after revision surgery. At the 3-month and 6-month follow-ups post-revision surgery, SLRT was free in the majority of patients (92.3% and 96.2%, respectively). Visual Analog Scale (VAS) and Oswestry disability index (ODI) scores significantly decreased between pre- and post-operative follow-ups at various intervals (P < 0.001). The percentage improvement in VAS and ODI scores was significant from 1 month to 6 months during follow-up. The VAS score improved by 62.5% at 1 month and 89% at the end of 6 months. The ODI score improved by 77% at 1 month to 97.3% at the end of 6 months.
Conclusion: This study demonstrates that revision spinal surgery – specifically utilizing the transforaminal lumbar interbody fusion technique – is a highly effective intervention for the management of FBSS.
Keywords: Failed back surgery syndrome, reoperation, lumbar vertebrae.
Predictors of reintervention success in failed back surgery syndrome (FBSS) following primary lumbar fusion encompass a broad spectrum of clinical, surgical, and patient-specific factors, each intricately linked to the underlying failure modes that necessitate further intervention [1]. A critical determinant is the accurate and timely identification of the primary cause of failure, which may manifest as pseudarthrosis, adjacent segment disease (ASD), hardware-related complications, or persistent nerve root compression [2]. Pseudarthrosis, characterized by a lack of solid bony fusion, often results in instability and pain, requiring revision fusion procedures. ASD involves degenerative changes at spinal levels adjacent to the fused segment, potentially leading to new symptoms and the need for extended fusion [3]. Hardware complications, including loosening, breakage, or malposition, can provoke mechanical pain or neurological symptoms, often necessitating hardware revision or removal [4]. Persistent or recurrent nerve compression, whether due to inadequate initial decompression, scar tissue formation, or foraminal stenosis, demands meticulous surgical re-exploration and neurolysis to alleviate symptoms [5].
In addition to failure mode characterization, patient-related factors substantially influence treatment outcomes. The baseline functional status of patients, encompassing pain severity, neurological deficits, and overall mobility, provides prognostic insight, with better pre-operative function correlating with improved post-operative recovery [6]. Comorbid conditions such as diabetes mellitus, osteoporosis, obesity, and smoking history adversely affect bone healing, immune response, and overall surgical risk, thereby diminishing the likelihood of successful reintervention [7]. Psychological factors, including depression, anxiety, and pain catastrophizing, have been increasingly recognized for their impact on pain perception, rehabilitation engagement, and long-term outcomes, underscoring the necessity of comprehensive psychosocial assessment and intervention as part of the treatment algorithm [8]. Surgical variables related to the initial lumbar fusion also critically determine the trajectory of reintervention success. The extent of fusion – whether single-level or multilevel – affects spinal biomechanics and the risk of adjacent segment degeneration [9]. The surgical approach, such as open posterior fusion versus minimally invasive techniques, influences tissue disruption, scar formation, and recovery dynamics [10]. The adequacy of initial neural decompression is paramount; incomplete decompression can leave residual nerve impingement, perpetuating symptoms and complicating revision surgery. Furthermore, the timing of reintervention is pivotal; early detection and management of failure modes before irreversible anatomical or neurological deterioration significantly enhance the prospects of favorable outcomes [11]. Delayed intervention often correlates with more complex pathology, increased surgical risk, and poorer functional recovery. A multidisciplinary evaluation framework is essential for optimizing reintervention outcomes. This includes advanced imaging modalities such as dynamic radiographs, computed tomography (CT) scans for assessing fusion status, and magnetic resonance imaging (MRI) for soft tissue and neural element evaluation [12]. Clinical examination should be supplemented by patient-reported outcome measures to capture pain, disability, and quality of life comprehensively. Integration of these data points facilitates the development of individualized management plans tailored to the specific failure mode [13]. For example, revision fusion with bone grafting and instrumentation may be indicated for pseudarthrosis, while extension of fusion constructs addresses ASD [14]. Hardware-related issues might require implant removal, replacement, or augmentation, and nerve root compression necessitates targeted decompression or neurolysis.
Optimizing modifiable patient factors before reintervention further improves success rates. This includes glycemic control in diabetic patients, smoking cessation programs, nutritional optimization, and psychological support interventions to address mood disorders and maladaptive pain behaviors [15]. Rehabilitation strategies should be designed to enhance functional capacity and prepare patients for the demands of revision surgery and post-operative recovery. By systematically analyzing the specific etiological triggers and current treatment modalities for FBSS in the context of primary fixation, this study aims to provide clinicians with a comprehensive framework for improving patient selection, surgical planning, and post-operative rehabilitation protocols.
This prospective clinical study was conducted at a tertiary care teaching hospital between January 2021 and June 2022 to evaluate pre- and post-operative outcomes in patients with FBSS. The minimum required sample size was calculated to be 24 considering the proportion of patients with improvement after revision surgery as 87% from the study by Elias et al., assuming 25% relative deviation for an alpha error of 5% and power of 80% the sample size came out to be 26 and hence 26 patients were included in the study of both sexes who met specific clinical and radiological criteria. Ethical permission was taken from IEC before onset of the study (ECR/53/INST/RR-16). Inclusion was restricted to adults aged 20–75 years diagnosed with FBSS following lumbar spinal fixation for degenerative spinal disorders. Eligible participants must have undergone one or more prior lumbar fixations at least 6 months previously, remained symptomatic despite 6 months of conservative management, and expressed a willingness to undergo revision surgery. Diagnosis was confirmed through comprehensive clinical evaluation and multi-modal imaging, including X-ray, CT, and MRI. To ensure a homogenous cohort focused on degenerative pathologies, several exclusion criteria were applied. The study excluded pregnant or lactating women, as well as patients with inflammatory disorders such as ankylosing spondylitis. Furthermore, individuals whose index surgeries were performed for spinal fractures, infections, tumors, or intradural pathologies were omitted. Patients presenting with comorbid major joint involvement, intracranial lesions, or underlying myopathies were also excluded from the final analysis. Upon enrollment, a detailed medical history and standardized physical examination were performed for all participants. Pre-operative and post-operative diagnostic protocols included routine hematological investigations, specifically including C-reactive protein and erythrocyte sedimentation rate to rule out occult infection. Radiographic assessment consisted of lumbosacral spine X-rays with anteroposterior, lateral, and dynamic (flexion/extension) views. Advanced imaging, including CT scans with sagittal reconstruction and MRI of the lumbosacral spine, was utilized as clinically indicated. Clinical outcomes were systematically evaluated at baseline (preoperatively) and at 1, 3, and 6 months postoperatively using two primary instruments: The Visual Analog Scale (VAS) for pain intensity and the Oswestry disability index (ODI) to assess the perceived level of disability across ten activities of daily living.
Most of the study participants were aged between 46 and 60 years (61.5%), with a mean age of 51 ± 11.6 years. The gender distribution was nearly equal, with males comprising 46.2% and females 53.8% of the participants. A majority (69.2%) did not have any comorbidities, while systemic hypertension (19.2%) and diabetes mellitus (15.4%) were the most common comorbid conditions. Degenerative disc disease with instability (34.6%) was the leading cause for the first surgery, followed by prolapsed intervertebral disc with a large protruding disc (30.8%) as shown in Table 1.
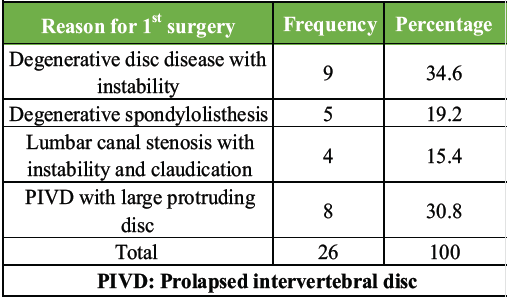
Table 1: Distribution of study participants based on reason for 1st surgery (n=26)
Most patients (84.6%) underwent fixation with transforaminal lumbar interbody fusion (TLIF), with the remaining procedures performed on a few patients. After the first surgery, 53.8% of participants were pain-free for 6 months to a year. The most common revision surgery was for broken, loose, or misplaced implants (34.6%), followed by extended fixation with TLIF (30.7%), Table 2.
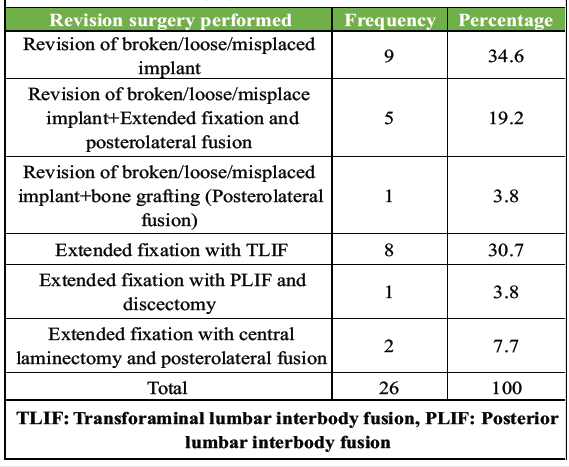
Table 2: Distribution of study participants based on revision surgery performed (n=26)
Before revision surgery, pharmacological and physiotherapy management was the most common conservative approach (42.3%), lasting 6–9 months for all patients. SLRT was positive at 30–40° in 38.4% of patients before revision surgery, but improved significantly post-surgery. At follow-ups 3 and 6 months after revision surgery, most patients (92.3% and 96.2%, respectively) were free of SLR. ODI scores showed significant reductions between pre- and post-operative follow-ups at various intervals (P < 0.001). The VAS score improved by 62.5% at 1 month and 89% at 6 months, while the ODI score improved by 77% at 1 month and 97.3% at 6 months. Preoperatively, neurological deficits (motor or sensory) were present in seven patients (27%), but postoperatively, only one patient had a persistent motor deficit in the bilateral lower limb. All but one patient reported being pain-free by the end of the follow-up. Most patients (88.5%) experienced no complications after revision surgery. No significant association was found between age, reason for the first surgery, type of first surgery, cause of failure of the first surgery, indication for revision surgery, type of revision surgery, and differences in VAS scores between 6 months and pre-operative. However, older patients and those with adjacent segment degeneration as the cause of failure of the first surgery showed better improvements, a statistically significant finding. No significant association was observed between the reason for the first surgery, type of first surgery, indication for revision surgery, type of revision surgery, and differences in ODI scores between 6 months and pre-op. The mean differences in ODI scores for patients aged 15–30 years, 31–45 years, 46–60 years, and over 60 years were 68, 74 ± 8.8, 72.85 ± 8.2, and 62.5 ± 9.8, respectively.
The primary objective of this study was to evaluate the clinical efficacy of revision spinal surgery, specifically focusing on the transition from initial surgical failure to post-operative recovery. Our findings demonstrate that revision surgery, particularly utilizing TLIF, results in a profound and statistically significant improvement in patient-reported outcomes. The most critical takeaway is the drastic reduction in disability and pain levels, with the ODI improving by 97.3% and the VAS score by 89% at the 6-month follow-up (P < 0.001). These results suggest that for patients suffering from FBSS, surgical revision is not merely a salvage procedure but a highly effective intervention for restoring functional independence. This is further evidenced by the resolution of straight leg raise (SLR) positivity, which dropped from 38.4% preoperatively to just 3.8% 6 months post-revision, indicating successful and sustained neural decompression.
When comparing these results to national and international literature, our findings are largely consistent with contemporary trends. The high success rate in pain reduction mirrors recent prospective data where a majority of revision patients achieved clinically significant improvements within the 1st year (Azad et al., 2024) [16]. Furthermore, our identified causes for revision – predominantly implant complications (34.6%) and degenerative instability – align with global studies that cite hardware failure and ASD as the primary drivers for reoperation (Kulkarni et al., 2018; Mahamid et al., 2025) [17,18]. Interestingly, our study observed that older patients and those with ASD showed better relative improvements. While some literature suggests younger patients have better regenerative capacity, our findings align with research suggesting that older populations may perceive a greater quality-of-life gain due to their lower baseline functional status (Behrbalk et al., 2021) [19].
The lack of a significant association between the specific type of revision surgery and the final functional scores suggests that the restoration of spinal stability and decompression is the primary driver of success, regardless of the technical approach used. This supports the clinical consensus that the surgical goal – rather than the specific instrument or approach – dictates the outcome in complex revision cases (Tran et al., 2024) [20]. Furthermore, the low complication rate (11.5%) in our cohort demonstrates that despite the inherent risks of revision surgery, such as scar tissue and distorted anatomy, modern techniques like TLIF provide a safe and reliable profile for stabilization.
Despite these positive outcomes, several limitations must be acknowledged. This study utilized a relatively small sample size from a single center, which may limit the generalizability of the findings to a broader, more diverse population. In addition, the 6-month follow-up period, while sufficient for assessing early recovery, is too short to evaluate long-term risks such as late-stage hardware fatigue or the future development of adjacent segment degeneration, which often manifests several years post-surgery (Lee and Lee, 2019) [21]. Finally, the lack of a control group receiving only conservative management prevents a direct comparison between surgical and non-surgical trajectories for these specific failure modes.
Moving forward, these preliminary findings highlight the need for larger, multicentric longitudinal studies with follow-up periods extending beyond 2 years. Future research should also explore the comparative cost-effectiveness and long-term durability of minimally invasive versus open revision techniques. Such data would be invaluable in refining surgical algorithms and setting realistic expectations for patients undergoing complex secondary spinal interventions.
This study demonstrates that revision spinal surgery – specifically utilizing the TLIF technique – is a highly effective intervention for the management of FBSS. The marked and statistically significant improvements in both the VAS and ODI scores, at 89% and 97.3% respectively, underscore the potential for profound functional restoration and pain relief even in complex clinical scenarios involving mechanical instability or implant failure.
While the surgical complexity of revision procedures is inherently elevated due to distorted anatomy and epidural fibrosis, the favorable complication profile (88.5% complication-free) observed in this cohort suggests that these interventions can be performed safely when tailored to the specific etiology of failure. Furthermore, the significant recovery observed in older patients and those with adjacent segment degeneration challenges the assumption that advanced age or disease progression limits the efficacy of secondary stabilization.
Ultimately, the restoration of spinal stability and definitive neural decompression remain the cornerstones of successful revision outcomes, regardless of the primary surgical indication. Although these short-term results are encouraging, they provide a critical foundation for future longitudinal, multicentric research aimed at optimizing patient selection and surgical timing. These findings offer robust clinical evidence for practitioners to consider revision surgery as a viable and successful trajectory for patients who have failed both primary surgical intervention and conservative management.
Revision surgery for Failed Back Surgery Syndrome is a high-stakes but effective salvage procedure when the underlying etiology is mechanical or structural. Success depends less on the hardware itself and more on the precise identification of the failure mode and the restoration of global spinal alignment.
References
- 1. Bergin SM, Wang TY, Park C, Rajkumar S, Goodwin CR, Karikari IO, et al. Pseudarthrosis rate following anterior cervical discectomy with fusion using an allograft cellular bone matrix: A multi-institutional analysis. Neurosurg Focus 2021;50:E6. [Google Scholar] [PubMed]
- 2. Colonna S, Lo Bue E, Pesaresi A, Dolci L, Gatto A, Ceroni L, et al. Impact of surgical timing on chronic subdural hematoma outcomes: Novel insights from a multicenter study. Neurosurg Rev 2025;48:349. [Google Scholar] [PubMed]
- 3. Haimoto S, Nishimura Y, Hara M, Nakajima Y, Yamamoto Y, Ginsberg HJ, et al. Clinical and radiological outcomes of microscopic lumbar foraminal decompression: A pilot analysis of possible risk factors for restenosis. Neurol Med Chir (Tokyo) 2017;58:49-58. [Google Scholar] [PubMed]
- 4. Marques MF, Fiere V, Obeid I, Charles YP, El-Youssef K, Lahoud A, et al. Pseudarthrosis in adult spine deformity surgery: Risk factors and treatment options. Eur Spine J 2021;30:3225-32. [Google Scholar] [PubMed]
- 5. Mascio A, Comisi C, Cinelli V, Pitocco D, Greco T, Maccauro G, et al. Radiological assessment of Charcot neuro-osteoarthropathy in diabetic foot: A narrative review. Diagnostics (Basel) 2025;15:767. [Google Scholar] [PubMed]
- 6. Mcclure JJ, Desai BD, Ampie L, You W, Smith JS, Buchholz AL. A systematic review of the cost-utility of spinal cord stimulation for persistent low back pain in patients with failed back surgery syndrome. Global Spine J 2021;11 Suppl 1:66S-72. [Google Scholar] [PubMed]
- 7. Park J, Ahn DK, Choi DJ. Treatment concept and technical considerations of biportal endoscopic spine surgery for lumbar spinal stenosis. Asian Spine J 2023;18:301-23. [Google Scholar] [PubMed]
- 8. Park SJ, Park JS, Nam Y, Choi YT, Lee CS. Who will require revision surgery among neurologically intact patients with proximal junctional failure after surgical correction of adult spinal deformity? Spine (Phila Pa 1976) 2021;46:520-9. [Google Scholar] [PubMed]
- 9. Pripotnev S, Mackinnon SE. Revision of carpal tunnel surgery. J Clin Med 2022;11:1386. [Google Scholar] [PubMed]
- 10. Stienen MN, Yoo K, Schonfeld E, Shah V, Abikenari M, Pangal D, et al. Single position lateral anterior lumbar interbody fusion at L5/S1. Neurosurgery 2025;96(3S):S17-25. [Google Scholar] [PubMed]
- 11. Tai DB, Patel R, Lovecchio F, Kwee T, Wouthuyzen-Bakker M. State-of-the-art review: Diagnosis and management of spinal implant infections. Clin Infect Dis 2024;79:e65-71. [Google Scholar] [PubMed]
- 12. Wang G, Pan S. Synergistic effects of psychological resilience training and nutritional support on postoperative recovery, nutritional reconstitution, sleep quality, and long-term survival in gastric cancer: A randomized controlled trial. Ann Surg Oncol 2025; Sep 15. doi: 10.1245/s10434-025-18313-6. Online ahead of print. [Google Scholar] [PubMed] [CrossRef]
- 13. Yamamoto Y, Kawakami M, Minetama M, Nakagawa M, Teraguchi M, Kagotani R, et al. Psychological predictors of satisfaction after lumbar surgery for lumbar spinal stenosis. Asian Spine J 2021;16:270-8. [Google Scholar] [PubMed]
- 14. Yoon SH, Kim BR, Lee SY, Beom J, Choi JH, Lim JY. Influence of comorbidities on functional outcomes in patients with surgically treated fragility hip fractures: A retrospective cohort study. BMC Geriatr 2021;21:283. [Google Scholar] [PubMed]
- 15. Zhou C, Cha T, Wang W, Guo R, Li G. Investigation of alterations in the lumbar disc biomechanics at the adjacent segments after spinal fusion using a combined in vivo and in silico approach. Ann Biomed Eng 2020;49:601-16. [Google Scholar] [PubMed]
- 16. Azad AK, Rahman M, Beg MO. Evaluating the success of revision spine surgery in patients with failed back surgery syndrome: A prospective study. EAS J Orthop Physiother 2024;6:98-105. [Google Scholar] [PubMed]
- 17. Behrbalk E, Uri O, Masarwa R, Alfandari L, Fatal S, Folman Y. Age-related differences in clinical outcomes of lumbar discectomy. Geriatr Orthop Surg Rehabil 2021;12:21514593211066732. [Google Scholar] [PubMed]
- 18. Kulkarni AG, Bangalore Kantharajanna S, Dhruv AN. Minimally invasive transforaminal lumbar interbody fusion: An attractive option for select failed backs. Asian Spine J 2018;12:52-8. [Google Scholar] [PubMed]
- 19. Mahamid A, Jayyusi F, Hodruj M, Mansour A, Fishman D, Behrbalk E. Comparative analysis of primary and revision single-level lumbar fusion surgeries: Predictors, outcomes, and clinical implications using big data. J Clin Med 2025;14:723. [Google Scholar] [PubMed]
- 20. Tran JD, Kubas D, Railey J, MacEwen HL, Helgeson MD, Wagner SC, et al. What are the differences in clinical and surgical outcomes between lumbar spine fusion approaches?-a narrative review. AME Med J 2024;9:15. doi: 10.21037/amj-23-142. [Google Scholar] [PubMed] [CrossRef]
- 21. Lee YC, Lee R. Minimal invasive surgical algorithm for revision lumbar Spinal surgery. J Spine Surg 2019;5:413-24. [Google Scholar] [PubMed]