Vertebral fragility fractures with normal bone mineral density should raise suspicion of systemic mastocytosis, and kyphoplasty-guided biopsy can be a key diagnostic tool in unexplained cases.
Dr. Joaquín Zúñiga Soria, Universidad del Desarrollo – Clínica Alemana de Santiago, Santiago, Chile. Av. Vitacura 5951, Santiago, Chile. E-mail: jzunigasoria.med@gmail.com
Introduction: Indolent systemic mastocytosis (ISM) is a rare clonal mast-cell disorder that may present with skeletal involvement, driven by mast-cell mediator–induced osteoclast activation. Vertebral fragility fractures can occur even in the presence of normal bone mineral density (BMD), making early diagnosis challenging.
Case Report: A 56-year-old woman with a prior history of cutaneous mastocytosis and persistently elevated serum tryptase presented with acute thoracolumbar pain after minimal trauma. Initial imaging revealed a T11 compression fracture with normal BMD on dual-energy X-ray absorptiometry. Kyphoplasty resulted in symptomatic improvement. Ten weeks later, she developed a second atraumatic L1 vertebral fracture. Given the atypical fracture pattern and mastocytosis history, she underwent L1 kyphoplasty with concurrent osseous biopsy. Histopathology showed mast-cell infiltrates with CD25 expression, and molecular testing identified the KIT D816V mutation. Bone marrow biopsy confirmed multifocal mast-cell infiltration, establishing a diagnosis of ISM. The patient was treated with intravenous bisphosphonates and multidisciplinary follow-up. At the last follow-up, she demonstrated pain resolution and no new fractures.
Discussion: This case underscores that ISM may present solely with skeletal manifestations despite normal BMD. Mast-cell mediators, including tryptase, histamine, tumour necrosis factor-alpha, and interleukin-6 promote osteoclastogenesis and microarchitectural bone fragility. Kyphoplasty-guided biopsy is a key diagnostic opportunity in unexplained vertebral fractures.
Conclusion: ISM should be considered in patients with atypical vertebral fractures, particularly when BMD is normal. Serum tryptase, KIT mutation testing, and bone biopsy enable timely diagnosis and treatment to prevent further skeletal morbidity.
Keywords: Indolent systemic mastocytosis, vertebral fragility fractures, secondary osteoporosis, KIT D816V mutation, tryptase, bone biopsy, normal bone mineral density, kyphoplasty biopsy, mast cell–mediated bone loss.
Systemic mastocytosis (SM) is a rare clonal mast-cell disorder characterised by abnormal mast-cell proliferation and infiltration of extracutaneous organs, most commonly the bone marrow, liver, gastrointestinal tract, and skeletal system [1,2]. Indolent SM (ISM) represents the most frequent adult subtype and typically follows a chronic course, though significant morbidity may occur from mast-cell mediator release and target-organ involvement [2-10]. Skeletal involvement is reported in up to 70% of SM patients and constitutes a major contributor to disability [3-5]. Bone manifestations range from osteopenia and osteoporosis to focal osteosclerosis and fragility fractures. Importantly, bone fragility in SM may occur despite normal bone mineral density (BMD), reflecting structural deterioration and trabecular microarchitectural compromise rather than mineral loss alone [4,5,13,14]. Mast-cell derived mediators—including tryptase, histamine, tumour necrosis factor-alpha (TNF-α), interleukin-1 (IL-1), and IL-6—stimulate osteoclastogenesis and impair osteoblast activity, promoting accelerated bone resorption and structural weakness [6-8]. Thus, vertebral compression fractures after minimal trauma should prompt consideration of SM, particularly in patients with prior cutaneous mastocytosis or elevated serum tryptase [16]. Conventional metabolic bone evaluation may underestimate fracture risk, and diagnostic confirmation requires integration of serum tryptase, KIT D816V mutation testing, and histopathologic demonstration of mast-cell infiltrates with aberrant immunophenotype (CD25+/CD2+) [6,9,11,12]. Because focal skeletal infiltration can precede systemic diagnosis, vertebral augmentation procedures offer a diagnostic opportunity via targeted bone biopsy in selected cases [17-19]. We present a patient with cutaneous mastocytosis who developed consecutive vertebral fragility fractures despite normal BMD, ultimately leading to the diagnosis of ISM. This case highlights the need to suspect mastocytosis in patients with unexplained vertebral fractures and preserved BMD and underscores the diagnostic utility of kyphoplasty-guided biopsy.
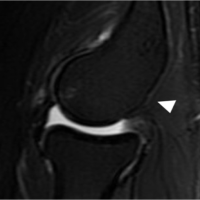
A 56-year-old woman with a history of cutaneous mastocytosis diagnosed 20 years earlier and chronically elevated serum tryptase (baseline 38 ng/mL) presented with sudden onset mid-thoracic pain after lifting a light object. She denied systemic symptoms, including flushing, syncope, gastrointestinal complaints, constitutional symptoms, or prior low-trauma fractures. Her medical history was otherwise unremarkable, and she was not on antiresorptive therapy. Spine radiographs and magnetic resonance imaging (MRI) revealed an acute T11 compression fracture with mild retropulsion but no spinal cord compromise (Fig. 1). Dual-energy X-ray absorptiometry (DXA) demonstrated normal BMD (lumbar T-score: −0.3; femoral neck T-score: −0.5). Laboratory evaluation showed normal calcium, phosphorus, Vitamin D, renal function, and parathyroid hormone. Given severe pain refractory to medical management, she underwent T11 kyphoplasty with symptomatic improvement. No biopsy was obtained at that time. Ten weeks later, the patient developed new severe lumbar pain without trauma. MRI demonstrated a new acute L1 vertebral compression fracture (Fig. 2). The repeat metabolic bone work-up remained unremarkable, including Vitamin D and calcium. Due to the recurrence of atraumatic vertebral fractures in the context of longstanding mastocytosis and normal BMD, targeted biopsy was planned at the time of L1 kyphoplasty. During the procedure, transpedicular bone biopsy was obtained (Fig. 3). Histology revealed dense clusters of mast cells with spindle morphology, positive for tryptase and aberrant CD25 expression. Molecular testing detected the KIT D816V mutation, confirming clonal mast-cell disease. Subsequent bone marrow biopsy demonstrated multifocal mast-cell infiltrates with the same immunophenotype, establishing a diagnosis of ISM. The patient initiated intravenous bisphosphonate therapy and multidisciplinary follow-up with hematology and metabolic bone disease specialists. At 12-month follow-up, she reported complete pain resolution, had returned to baseline physical activity, and experienced no new fractures. Follow-up imaging demonstrated stable vertebral alignment without further compression (Fig. 4).
ISM is a clonal mast-cell disorder characterized by aberrant mast-cell accumulation and mediator release. Although cutaneous involvement is typical, skeletal manifestations are common and clinically meaningful, affecting up to 70% of patients [1,2]. The spectrum ranges from osteopenia and osteoporosis to focal osteosclerosis and fragility fractures [3,4,5]. Notably, vertebral fractures may occur despite normal BMD, as shown in this case, reflecting microarchitectural compromise rather than loss of mineral content alone [4,6].
Mast-cell–mediated bone fragility:
Bone involvement in mastocytosis arises predominantly from mast-cell activation and mediator release. Tryptase, histamine, prostaglandins, TNF-α, IL-1, and IL-6 stimulate RANKL-dependent osteoclastogenesis and impair osteoblast function [6-8]. In addition, KIT D816V-driven constitutive signalling promotes mast-cell proliferation and cytokine secretion via STAT5, PI3K, and MAPK pathways, amplifying bone turnover and trabecular deterioration [9,10,12]. These mechanisms explain why standard DXA may appear normal despite substantial structural weakness, underscoring the importance of evaluating trabecular integrity using trabecular bone score or opportunistic computed tomography Hounsfield units [13-15].
Diagnostic considerations:
Diagnosis requires integration of clinical suspicion, biochemical testing, molecular studies, and histopathology. Persistent serum tryptase elevation (>20 ng/mL) is suggestive but not universal and may be normal in a minority of patients [1,16]. Bone biopsy during vertebral augmentation offers a unique diagnostic opportunity in patients with unexplained fractures [17-19]. In this case, kyphoplasty-guided biopsy was crucial in confirming mast-cell infiltration with aberrant CD25 expression and the KIT D816V mutation, fulfilling WHO criteria [9,11,12].
Differential diagnosis:
Vertebral fragility fractures with normal BMD necessitate evaluation for secondary causes, including:
- Mastocytosis
- Multiple myeloma (including non-secretory)
- Monoclonal gammopathy of skeletal significance
- Glucocorticoid-induced osteoporosis
- Hyperparathyroidism
- Osteomalacia with preserved calcium/phosphorus
- Marrow infiltrative disorders (lymphoma, leukemia)
- Metabolic bone diseases with preserved BMD (e.g., osteogenesis imperfecta in adults).
Distinguishing mastocytosis from myeloma is essential; hallmark features include mast-cell aggregates, CD25 positivity, and KIT mutation rather than plasma-cell infiltrates and monoclonal protein expression [9,11,17].
Management and outcomes:
Management focuses on fracture stabilization, optimization of bone health, and mast cell–directed therapy. Bisphosphonates and, in selected cases, anabolic agents such as teriparatide have shown benefit [20]. Targeted KIT inhibitors, including midostaurin and avapritinib, have demonstrated efficacy [21,22]. Our patient achieved durable pain relief and radiographic stability after kyphoplasty and initiation of intravenous bisphosphonates, with no new fractures at 1-year follow-up.
Clinical implications:
This case reinforces key clinical points:
- Vertebral fractures may represent the first systemic manifestation of ISM.
- Normal DXA does not exclude mast-cell–mediated bone fragility.
- Persistent or recurrent atraumatic vertebral fractures warrant targeted evaluation with serum tryptase and KIT mutation analysis.
- Kyphoplasty-guided biopsy is a valuable diagnostic tool in unexplained fractures.
- Early diagnosis may prevent further skeletal morbidity and enable appropriate hematologic follow-up.
ISM should be considered in patients presenting with atraumatic vertebral fragility fractures, particularly when BMD is preserved, as standard densitometry may underestimate mast-cell–mediated impairment of trabecular microarchitecture. Persistent elevation of serum tryptase, a history of cutaneous mastocytosis, or unexplained recurrent fractures should prompt targeted diagnostic work-up, including KIT D816V testing and bone biopsy, ideally performed at the time of vertebral augmentation. Histopathologic demonstration of aberrant mast-cell infiltrates with CD25 expression remains essential for definitive diagnosis. Early recognition allows implementation of fracture prevention strategies, antiresorptive therapy, and multidisciplinary monitoring to mitigate long-term skeletal morbidity. This case underscores the clinical value of kyphoplasty-guided biopsy in patients with unexplained vertebral fractures and highlights the need for heightened clinical suspicion of SM when axial fragility occurs despite normal BMD.
Systemic mastocytosis should be suspected in patients presenting with recurrent or atypical vertebral fractures despite normal bone mineral density. Serum tryptase levels, KIT mutation analysis, and bone biopsy—ideally obtained during vertebral augmentation—are essential for early diagnosis and prevention of further skeletal complications.
References
- 1. <p>Valent P, Akin C, Metcalfe DD. Mastocytosis: 2023 update on diagnosis, risk stratification and management. Blood 2023;141:2721-36.</p> [Google Scholar] [PubMed]
- 2. <ol start="2"> [Google Scholar] [PubMed]
- 3. <li>Matito A, Morgado JM, Sánchez-Muñoz L, Álvarez-Twose I, Mollejo M, Orfao A, et al. Management of indolent systemic mastocytosis in daily practice. Allergy 2015;70:897-906.</li> [Google Scholar] [PubMed]
- 4. <li>Rossini M, Zanotti R, Bonadonna P, Artuso A, Caruso B, Viapiana O, et al. Bone mineral density, bone turnover markers and fractures in patients with indolent systemic mastocytosis. Bone 2011;49:880-5.</li> [Google Scholar] [PubMed]
- 5. <li>Van Anrooij B, Kluin-Nelemans HC, Van Der Veer E, Arends S, Van Doormaal JJ, Oude Elberink JNG, et al. High prevalence of vertebral fractures despite normal bone mineral density in systemic mastocytosis. Bone 2016;90:21-4.</li> [Google Scholar] [PubMed]
- 6. <li>Van Der Veer E, Arends S, Kluin-Nelemans HC, Van Doormaal JJ, Oude Elberink JNG, De Monchy JGR, et al. Bone involvement in mastocytosis: Pathogenesis, clinical features and management. Calcif Tissue Int 2019;104:469-83.</li> [Google Scholar] [PubMed]
- 7. <li>Escribano L, Orfao A, Díaz-Agustín B, Villarrubia J, Cerveró C, López A, et al. Immunophenotypic and molecular diagnosis of systemic mastocytosis. J Allergy Clin Immunol 2001;107:411-26.</li> [Google Scholar] [PubMed]
- 8. <li>Kushnir-Sukhov NM, Brittain E, Brown M, Theoharides TC, Metcalfe DD, Kalesnikoff J, et al. Tryptase levels and cytokine-mediated osteoclast activation in mastocytosis. J Immunol 2006;176:4025-34.</li> [Google Scholar] [PubMed]
- 9. <li>Pagani M, Zini M, Bonecchi R, Galli SJ, Grimbaldeston MA, Maurer M, et al. Bone turnover in mastocytosis: Link between mast cells and bone loss. Bone Res 2023;11:32-41.</li> [Google Scholar] [PubMed]
- 10. <li>Arock M, Sotlar K, Akin C, Broesby-Olsen S, Hoermann G, Escribano L, et al. KIT mutation and mast-cell disease: Biology and clinical implications. Blood 2015;125:581-9.</li> [Google Scholar] [PubMed]
- 11. <li>Pardanani A. Systemic mastocytosis in adults: 2023 update on diagnosis and management. Blood 2023;142:567-78.</li> [Google Scholar] [PubMed]
- 12. <li>Horny HP, Metcalfe DD, Bennett JM, Bain BJ, Akin C, Escribano L, et al. WHO classification of mast cell disorders. Histopathology 2016;68:351-70.</li> [Google Scholar] [PubMed]
- 13. <li>Garcia-Montero AC, Jara-Acevedo M, Teodósio C, Sánchez ML, Núñez R, Prados A, et al. KIT mutation in mast cells and other bone marrow hematopoietic cell lineages in systemic mast cell disorders. Blood 2006;108:2366-72.</li> [Google Scholar] [PubMed]
- 14. <li>Silva BC, Broy SB, Boutroy S, Schousboe JT, Shepherd JA, Leslie WD, et al. Trabecular bone score: A new DXA-derived index for bone microarchitecture. J Clin Endocrinol Metab 2014;99:E2520-8.</li> [Google Scholar] [PubMed]
- 15. <li>Zebaze R, Ghasem-Zadeh A, Mbala A, Seeman E. Bone fragility despite normal bone mineral density: Cortical porosity and trabecular disruption. J Bone Miner Res 2010;25:787-94.</li> [Google Scholar] [PubMed]
- 16. <li>Schousboe JT, Shepherd JA, Bilezikian JP, Baim S. Executive summary of the ISCD position development conference on bone densitometry. J Clin Densitom 2013;16:455-66.</li> [Google Scholar] [PubMed]
- 17. <li>Brockow K, Jofer C, Ring J, Darsow U, Biedermann T, Schmid-Grendelmeier P, et al. Long-term follow-up of cutaneous mastocytosis. J Invest Dermatol 2008;128:1619-26.</li> [Google Scholar] [PubMed]
- 18. <li>Shah NP, Berta SC, Lee SK, Phillips FM, An HS, Vaccaro AR, et al. Diagnostic yield of vertebral biopsy in unexplained vertebral fractures. Clin Spine Surg 2019;32:314-9.</li> [Google Scholar] [PubMed]
- 19. <li>Park YS, Lee SH, Lee HY, Kim KT, Suk KS, Lee JH, et al. Value of biopsy during vertebroplasty for unsuspected malignancy. AJNR Am J Neuroradiol 2011;32:2023-7.</li> [Google Scholar] [PubMed]
- 20. <li>Hodges SD, Humphreys SC, Eck JC, Covington LA. Incidental diagnoses during spine biopsy at vertebroplasty. Spine J 2004;4:537-40.</li> [Google Scholar] [PubMed]
- 21. <li>Rossini M, Viapiana O, Orsolini G, Idolazzi L, Gatti D, Adami S, et al. Efficacy of bisphosphonates in systemic mastocytosis-related osteoporosis. Calcif Tissue Int 2016;99:173-9.</li> [Google Scholar] [PubMed]
- 22. <li>Gotlib J, Kluin-Nelemans HC, George TI, Akin C, Sotlar K, Hermine O, et al. Efficacy and safety of midostaurin in advanced systemic mastocytosis. N Engl J Med 2016;374:2530-41.</li> [Google Scholar] [PubMed]
- 23. <li>DeAngelo DJ, Radia D, George TI, Robinson S, Hexner EO, Scherber RM, et al. Avapritinib in advanced systemic mastocytosis. Blood 2021;137:3650-63. [Google Scholar] [PubMed]