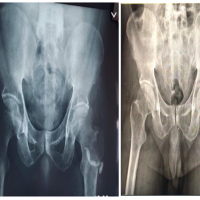
Melioidosis can occur even in young, immunocompetent, non-diabetic individuals, not just in traditionally high-risk groups. Burkholderia pseudomallei, though rare, should be included in the differential diagnosis in endemic regions in case of trauma-induced infections.
Dr. Zobiakhlui Chhakchhuak, Department of Microbiology, Ebenezer Medical Center, Aizawl, Mizoram. E-mail: zobiakhlui@gmail.com
Introduction: Melioidosis, a potentially fatal infectious human disease labelled the “silent killer disease", caused by Burkholderia pseudomallei, is an environmental Gram-negative bacillus endemic to tropical regions. To our knowledge, this is one of the few reported cases of the rare melioidosis infection from the Mizoram state bordering Myanmar and Bangladesh.
Case Report: We report a case of culture-confirmed B. pseudomallei osteomyelitis in a 29-year-old non-diabetic, immunocompetent adult from an endemic region of Myanmar.
Conclusion: The diagnosis of melioidosis requires a high index of suspicion among clinicians and clinical microbiologists, particularly in endemic areas. This report aims to highlight the complexity of the organism in view of the diagnostic challenges and the critical importance of clinical suspicion of melioidosis.
Keywords: Burkholderia pseudomallei, melioidosis, osteomyelitis.
Burkholderia pseudomallei is the causative agent of melioidosis or Whitmore’s disease, which is widely found in the environment. Human infections may result from exposure to the organism through inhalation, non-intact skin, or ingestion. B. pseudomallei causes a wide range of infections from localized abscesses such as osteomyelitis to pulmonary manifestations [1]. Melioidosis is a rare entity in Northeast India, and reports of it are rare. Data regarding melioidosis infections are more commonly reported from tropical countries of Southeast Asia and along the coastal regions of India [2,3]. Historically, it was first discovered by Alfred Whitmore and CS Krishnaswami in 1911 at Rangoon, Myanmar [1].
A 29-year-old male was brought to the emergency department with complaints of pain in the left knee. There was a history of progressive pain around the left knee, causing difficulty in walking. The left knee was tender and slightly swollen upon physical examination. The patient is from a small village in Myanmar where farming is the main source of livelihood. He came to the hospital in Mizoram, the northeastern part of India, to seek higher medical facilities due to restrained resources back in Myanmar. There was a reported history of trauma to the leg 3 months before admission. He was non-diabetic, and no other comorbidities were noted. His blood investigations were all within the reference range except for the inflammatory markers, i.e., C-reactive protein and erythrocyte sedimentation rate (Table 1). Magnetic resonance imaging of the left knee documented patchy areas of abnormal marrow signal intensity involving the lower end of the femur, medial and lateral femoral condyles, diaphysis, and metaphysis of the upper tibia. Moderate joint effusion with mild synovial thickening extending into suprapatellar and bilateral patellofemoral recesses was also noted. Other possible infectious causes, such as tuberculous osteomyelitis, were ruled out by the cartridge-based nucleic acid amplification test. Incision and debridement, along with corticotomy for the left upper tibia, were performed (Fig. 1). Pus cells and Gram-negative bacilli were seen on the Gram stain. The thick caseous pus that was evacuated from the operation resulted in the growth of B. pseudomallei in microbial culture. Identification of the organism was done using the Vitek 2 compact system (BioMérieux Inc., France). Antibiotic susceptibility testing (AST) of the organism was performed using the Kirby–Bauer disc method on Mueller–Hinton agar following EUCAST guidelines [4]. Meropenem (10 µg), doxycycline (30 µg), trimethoprim-sulfamethoxazole (1.25/23.75 µg), and ceftazidime (10 µg) antibiotic discs were used. Gentamicin (10 µg), colistin (10 µg), and amoxicillin–clavulanic acid (20/10 µg) antibiotic discs were used to aid in screening of B. pseudomallei. The isolated organism was found to be susceptible to meropenem and doxycycline. Trimethoprim–sulfamethoxazole and ceftazidime were within the intermediate zone. Colistin and gentamicin showed no zone of inhibition around the discs, while the organism was sensitive to the amoxicillin–clavulanic acid disc (Table 2). The patient was given intravenous amoxicillin–potassium clavulanate and oral trimethoprim–sulfamethoxazole and was discharged in stable condition with advice to continue oral trimethoprim–sulfamethoxazole. However, the patient was re-admitted almost 2 months after his initial discharge due to a recurring fever and unresolved swelling of the operated site. He was re-operated on, and the evacuated pus specimen was sent for microbial culture, which yielded the same organism. A saucerisation operation was done along with the implantation of a stimulant and meropenem antibiotic beads (Fig. 2). He was discharged after a month in stable condition after a sterile pus culture report with a marked decrease in the inflammatory markers. The patient did not turn up for review, hence lost to follow-up.
Pseudomallei is an emerging opportunistic infection, where its diagnosis can be quite challenging due to its wide range of clinical manifestations mimicking other infections. A melioidosis case is defined by the presence of clinical and epidemiological criteria confirmed either by culture or polymerase chain reaction [1]. Melioidosis may manifest as osteomyelitis, septic arthritis, soft-tissue infection, pneumonia, and multiorgan involvement in severe cases [5,6]. Vithiya et al. reported 22% melioidosis-associated osteomyelitis in a 10-year retrospective study from India [7], whereas Wu et al. observed osteomyelitis in 56.8% of cases in a comparable study from China [8]. Melioidosis is known to have an acute presentation (80.9%) [7]. However, a rare chronic case has also been documented [9]. One of the main contributors to B. pseudomallei infection is diabetes mellitus, followed by other pre-existing conditions such as thalassaemia and renal diseases. [3,6] Nonetheless, non-diabetic melioidosis is not unusual, as observed in our case. Occupational exposure and geographical origin seemed to be crucial links in our patient. These are the most plausible reasons for the cause of melioidosis infection in our patient. Pseudomallei is a Gram-negative, non-fermenter, oxidase-positive bacterium that shows resistance to aminoglycosides, colistin, and polymyxin. The 2024 revised Darwin melioidosis recommendations include intravenous ceftazidime or meropenem for a minimum of 2 weeks to 8 weeks in complicated melioidosis infections such as osteomyelitis, septic arthritis, neurological melioidosis, and genitourinary infections. Apart from antibiotic coverage, surgical drainage is indicated in most abscesses. Osteomyelitis often requires repeated and aggressive surgical drainage, as done in our patient. Darwin treatment guidelines also recommended an eradication therapy with oral trimethoprim–sulfamethoxazole along with folic acid for at least a further 3 months and monitoring of adverse effects of prolonged trimethoprim–sulfamethoxazole [10]. The prolonged treatment guidelines are due to the high recurrent rate of the infection, either due to reinfection or relapse. However, with the advancement of diagnosis and AST, a declining trend in recurrence rate is anticipated.
Pseudomallei infection is presumed to have higher rates than reported, due to a lack of knowledge about the organism and its variable non-specific clinical manifestations. Hence, many cases remain undiagnosed or misdiagnosed. It is imperative to raise awareness in order to tackle the silent endemic caused by this organism.
Melioidosis can present as osteomyelitis or septic arthritis, often mimicking other infections and delaying diagnosis. A high index of suspicion, timely culture confirmation, aggressive surgical management, and prolonged eradication therapy are key to reducing complications and recurrence.
References
- 1. </p> [Google Scholar] [PubMed]
- 2. <ol> [Google Scholar] [PubMed]
- 3. <li>CD Alert. Delhi: National Centre for Disease Control, Directorate General of Health Services, Government of India; 2019. Available from: <a href="https://ncdc.mohfw.gov.in/wp-content/uploads/2024/05/meliodosis-cd_alert-duly-launched-by-hon_ble-hfm-on-annual-day-on-30th-july-2019.pdf_x0005_">https://ncdc.mohfw.gov.in/wp-content/uploads/2024/05/meliodosis-cd_alert-duly-launched-by-hon_ble-hfm-on-annual-day-on-30th-july-2019.pdf</a></li> [Google Scholar] [PubMed]
- 4. <li>Raina S, Gautam D, Kumar R, Sisodia K, Mukhopadhyay C, Kaur H, <em>et al</em>. Unearthing the burden of melioidosis in North India- an emerging threat in a non-endemic region. Curr Res Microb Sci 2025;8:100344.</li> [Google Scholar] [PubMed]
- 5. <li>Gupta N, Kumar TP, Sethi A, Kumari P, Kaur H, Mukhopadhyay C. Melioidosis in India: A systematic review of individual cases. IJID Reg 2026;18:100843.</li> [Google Scholar] [PubMed]
- 6. <li>Dance DA, Wuthiekanun V, Baird RW, Norton R, Limmathurotsakul D, Currie BJ. Interpreting <em>Burkholderia pseudomallei</em> disc diffusion susceptibility test results by the EUCAST method. Clin Microbiol Infect 2021;27:827-9.</li> [Google Scholar] [PubMed]
- 7. <li>Chang CY. Pulmonary melioidosis. QJM 2021;114:900.</li> [Google Scholar] [PubMed]
- 8. <li>Warapitiya DS, Subasinghe S, De Silva RF, Piyarisi DL, Jayatilleke K. Severe sepsis with multiorgan failure due to melioidosis: A lesson to learn. Case Rep Med 2021;8:5563214.</li> [Google Scholar] [PubMed]
- 9. <li>Vithiya G, Rajalakshmi Preeti G, Shunmuga Sundaram P, Rajendran T. Musculoskeletal melioidosis-a retrospective review of 22 cases from a tertiary care centre in South Tamilnadu. Indian J Med Microbiol 2024;52:100750.</li> [Google Scholar] [PubMed]
- 10. <li>Wu H, Wang X, Zhou X, Chen S, Mai W, Huang H, <em>et al</em>. Osteomyelitis and septic arthritis due to <em>Burkholderia pseudomallei</em>: A 10-year retrospective melioidosis study from South China. Front Cell Infect Microbiol 2021;11:654745.</li> [Google Scholar] [PubMed]
- 11. <li>Alexander V, Koshy M, Shenoy R, Sudarsanam TD. Chronic multifocal osteomyelitis: A rare presentation of melioidosis. J Family Med Prim Care 2018;5:1133-5.</li> [Google Scholar] [PubMed]
- 12. <li>Currie BJ, Janson S, Meumann E, Martin GE, Ewin T, Marshal CS. The 2024 revised Darwin melioidosis treatment guideline. North Territ Dis Control Bull 2023;30:3-12.</li> [Google Scholar] [PubMed]
- 13. </ol> [Google Scholar] [PubMed]
- 14. <p> [Google Scholar] [PubMed]