In the elderly rheumatoid arthritis patients with high disease activity, we should be cautious of a fracture cascade.
Dr. Rie Kurose, Department of Orthopaedic Surgery, Hirosaki Memorial Hospital, 59-1 Sakaizeki Nishida, Hirosaki, Japan. E-mail: riekuro@hirosaki-kinen.or.jp
Introduction:Patients with rheumatoid arthritis (RA) have generalized bone loss and deterioration of bone quality, leading to a relatively higher risk of vertebral and non-vertebral fractures. It has been previously reported that patients who cause fragility fractures such as vertebral fractures, had statistically higher scores of RA disease activity, but there are no case reports.
Case Report:The case described in this report is an elderly woman with RA who was receiving treatment including glucocorticoids. Her state of RA activity had been in remission for a long time, and she was also receiving treatment to prevent osteoporosis. However, the worsening of the disease activity triggered a fracture cascade starting from a vertebral fracture and her activities of daily living deteriorated rapidly.
Conclusion:This report highlights the difficulties in treating osteoporosis and preventing fragility fractures in an elderly patient with RA and the importance of managing RA disease activity.
Keywords: Elderly patient, rheumatoid arthritis, fracture cascade, disease activity, osteoporosis.
Rheumatoid arthritis (RA) is a chronic, autoimmune, and systemic inflammatory disease. The increased expression of inflammatory cytokines (tumor necrosis factor [TNF-α], interleukin [IL]-6, IL-1, etc.) promotes the expression of the receptor activator of nuclear factor-kappa B ligand (RANKL) in osteoblasts, activated T cells, and synovial fibroblasts, leading to osteoclast differentiation and activation, which causes periarticular bone loss. In addition to systemic inflammation, the use of glucocorticoids which inhibit bone formation, increase bone resorption, and decrease bone quality, and the decreasing in mechanical stress caused by local immobility increase the risk of generalized bone loss and osteoporosis. It has been reported that fragility fractures are more common in patients with RA compared to individuals in the general population [1]. Fragility fractures have been recognized as a disease that not only deteriorates the activities of daily living (ADL) but also affects the prognosis of life. The risk of fragility fractures is higher in an elderly RA due to aging, physical dysfunction associated with joint destruction, and long-term glucocorticoid use. In this study, we report the case of an elderly RA who had been in remission for more than a decade and was treated for osteoporosis; however, a sudden fracture cascade was triggered by worsening of the disease activity, which rapidly deteriorated her ADL.
A 86-year-old woman was diagnosed with RA at the age of 60 years. She had been receiving treatment with methotrexate (MTX) at a dose of 4–10 mg/week and the remission was maintained for approximately 10 years. In addition, she had been treated with prednisolone (PSL) at a dose of 2–5 mg/day for approximately 10 years. However, she had been treated with a reduced dose of MTX since 5 years ago due to renal dysfunction. Two years ago, she stopped taking MTX due to a few medication errors, and golimumab (GLM) 100 mg/month, which is an anti-TNF- α antibody, was started. After 6 months of GLM treatment, her medication was changed to baricitinib (BARI) 2 mg/day because there was no symptomatic improvement. Subsequently, she maintained in a state of remission to low disease activity. After 1 year and 3 months of BARI treatment, she experienced palpitations and nausea. She underwent a checkup with an internal medicine doctor; however, nothing was found. At that time, she stopped taking BARI based on her personal judgment. Instead of BARI, iguratimod (IGU) 25 mg/day was prescribed, but she stopped taking IGU in a few days later due to itching symptoms on her entire body. Finally, she had been treated with PSL only, without any new medication after she stopped taking BARI. After 3 months of only PSL intake, she visited our hospital due to generalized pain and difficulty in performing physical movement. There were no falls or trauma, but she could not walk. The physical examination revealed swelling and tenderness in the bilateral wrist joints and low back pain. Regarding blood examination, C-reactive protein (CRP) was 4.5 mg/dL (normal range: 0–0.3 mg/dL), erythrocyte sedimentation rate 60 mm/h (normal range: 0–15 mm/h), rheumatoid factor 233.6 U/mL (normal range: 0–15 U/mL), and serum matrix metalloproteinase-3 176 ng/mL (normal range, male: 36.9–121 ng/mL; female: 17.3–59.7 ng/mL). Bone mineral density (g/cm2) results were 0.412 and 0.683 at the right femoral neck and the lumbar spine (L2-L4), respectively. Radiography and magnetic resonance imaging (MRI) revealed L2 compression fracture and sacral fracture (Fig. 1).  A L3 compression fracture was also found, but it was asymptomatic and obsolete. Furthermore, 3 years ago, she fell and injured a left femoral neck fracture and underwent bipolar hip arthroplasty at another hospital. Malignancy and infectious diseases were ruled out after the examination results. After admission to our hospital, BARI was administered again to improve the RA disease activity and no heart palpitations or nausea was recorded. Furthermore, teriparatide was initiated to enhance osteoporosis treatment while controlling that her renal function did not deteriorate further (Fig. 2).
A L3 compression fracture was also found, but it was asymptomatic and obsolete. Furthermore, 3 years ago, she fell and injured a left femoral neck fracture and underwent bipolar hip arthroplasty at another hospital. Malignancy and infectious diseases were ruled out after the examination results. After admission to our hospital, BARI was administered again to improve the RA disease activity and no heart palpitations or nausea was recorded. Furthermore, teriparatide was initiated to enhance osteoporosis treatment while controlling that her renal function did not deteriorate further (Fig. 2). 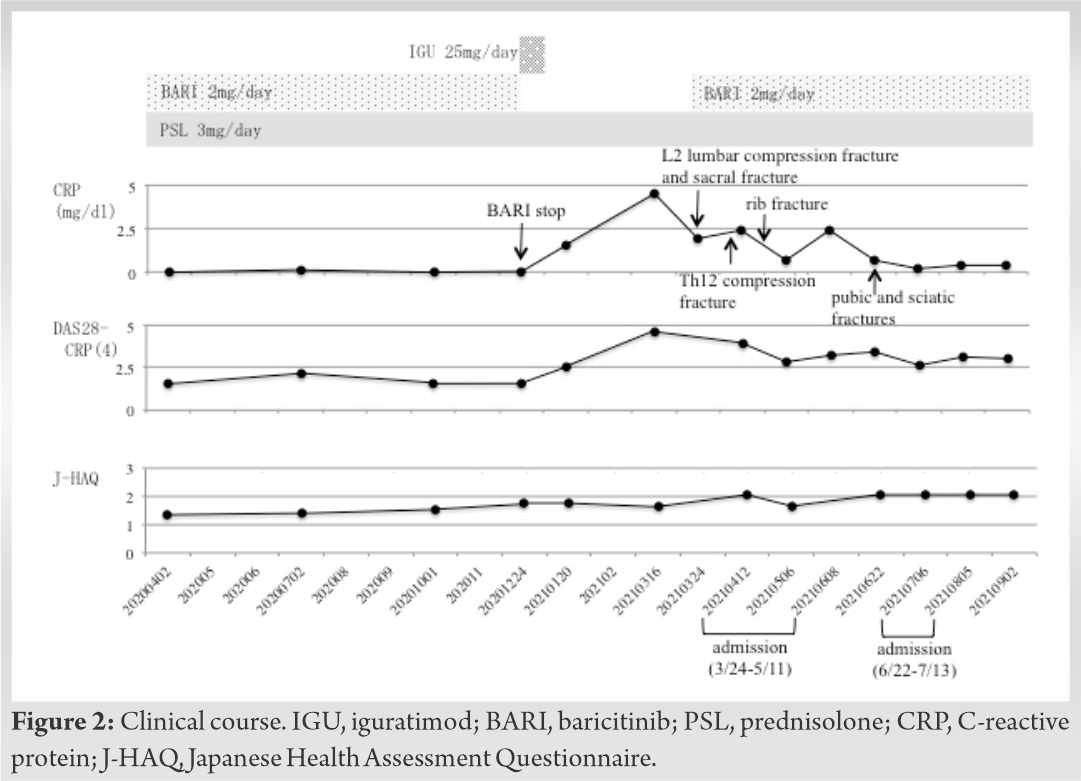 Two weeks after admission, a follow-up computed tomography (CT) was performed and revealed a new non-traumatic thoracic compression fracture at Th12. One month after admission, she lost her balance, while she was transferred from a wheelchair in the ward and hit the right side of her chest on the fence, resulting in the fracture of a right rib. Three weeks following this accident, she could walk by pushing a wheel barrow and was discharged to the nursing facility. However, 1 month after she was discharged, she fell in the bathroom of the nursing facility and could not walk. Radiography and MRI revealed left pubic and sciatic fractures, and she was readmitted to our hospital. Although daily rehabilitation was performed, the progress was very slow due to the pain at the fracture sites. She could not walk even with a cane. She was discharged to the nursing facility in a wheelchair.
Two weeks after admission, a follow-up computed tomography (CT) was performed and revealed a new non-traumatic thoracic compression fracture at Th12. One month after admission, she lost her balance, while she was transferred from a wheelchair in the ward and hit the right side of her chest on the fence, resulting in the fracture of a right rib. Three weeks following this accident, she could walk by pushing a wheel barrow and was discharged to the nursing facility. However, 1 month after she was discharged, she fell in the bathroom of the nursing facility and could not walk. Radiography and MRI revealed left pubic and sciatic fractures, and she was readmitted to our hospital. Although daily rehabilitation was performed, the progress was very slow due to the pain at the fracture sites. She could not walk even with a cane. She was discharged to the nursing facility in a wheelchair.
RA is an autoimmune disease, in which bone destruction is induced by persistent inflammation, causing periarticular and generalized bone loss. Regarding the cause of periarticular bone loss, it is thought to be related to synovitis [2, 3], and it has been recently reported that synovial fibroblasts and plasma cells express RANKL and induce osteoclastogenesis [4]. Regarding the causes of generalized bone loss, it is thought to be related to aging, glucocorticoids use, and physical immobility [5]. In addition, generalized bone loss is influenced by several other factors such as poor nutritional status, sarcopenia, and frailty [6]. Therefore, the risk of fragility fractures is significantly higher in patients with RA than that in healthy individuals [7, 8]. Furthermore, fragility fractures in RA are not limited to the vertebral and proximal femur but can occur anywhere in the body, including the pelvis and metaphysis [9]. Some reports pointed out that incidence of vertebral fractures in RA patients was associated with high RA disease activity [10, 11]. On the other hand, some studies have reported that the incidence of non-vertebral fractures in RA patients was high regardless of RA disease activity [12]. This suggests that it is important to manage RA disease activity. In the elderly RA, the risk of fragility fractures is more severe due to aging, physical dysfunction associated with joint destruction, and long-term use of glucocorticoids and the complications of fragility fractures in elderly individuals cause a decrease in physical performance, which leads to a reduced ability in walking and balance, causing recurrent falls and a fracture cascade. In our case, the patient had been in clinical remission for more than 10 years, but she experienced vertebral fractures and a fracture cascade when her disease activity became worse. As a result, she could not walk with a cane over a short period of approximately 4 months and her ADL deteriorated rapidly. According to a recent report, the previous fracture is the highest risk factor for fragility fractures [13]. In this case, fractures cascade occurred in spite of osteoporosis treatment. In the elderly RA, it is important to manage RA activity and we need to pay attention occurrences of fragility fractures.
Elderly RA have various comorbidities including renal dysfunction such as this case and therefore available medications are limited. Further studies are needed on the timing of medication initiation, choice of medications, and dose of medications for osteoporosis treatment in elderly RA. In addition to RA management, we need to pay attention occurrences of fragility fractures to avoid worsening ADL in elderly RA.
In the elderly patients with RA, it is necessary to manage RA activity and pay attention occurrences of fragility fractures.
References
- 1.Huusko TM, Korpela M, Karppi P, Avikainen V, Kautiainen H, Sulkava R. Threefold increased risk of hip fractures with rheumatoid arthritis in central Finland. Ann Rheum Dis 2001;60:521-2. [Google Scholar]
- 2.Sambrook PN, Reeve J. Bone disease in rheumatoid arthritis. Clin Sci 1988;74:225-30. [Google Scholar]
- 3.Furumitsu Y, Inaba M, Yukioka K, Yukioka M, Kumeda Y, Azuma Y, et al. Levels of serum and synovial fluid pyridinium crosslinks in patients with rheumatoid arthritis. J Rheumatol 2000;27:64-70. [Google Scholar]
- 4.Komatsu N, Win S, Yan M, Huynh NC, Sawa S, Tsukasaki M, et al. Plasma cells promote osteoclastogenesis and periarticular bone loss in autoimmune arthritis. J Clin Invest 2021;131:e143060. [Google Scholar]
- 5.Kumeda Y, Inaba M, Goto H, Nagata M, Henmi Y, Furumitsu Y, et al. Increased thickness of the arterial intimamedia detected by ultrasonography in patients with rheumatoid arthritis. Arthritis Rheum 2002;46:1489-97. [Google Scholar]
- 6.Inaba M, Mori H, Katsumata T, Goto H, Ishimura E, Kawagishi T, et al. Hyperparathyroidism is augmented by ovar-iectomy in Nagase an albuminemic rats. J Nutr 2000;130:1543-7. [Google Scholar]
- 7.van Staa TP, Geusens P, Bijlsma JW, Leufkens HG, Cooper C. Clinical assessment of the long-term risk of fracture in patients with rheumatoid arthritis. Arthritis Rheum 2006;54:3104-12. [Google Scholar]
- 8.Wright NC, Lisse JR, Walitt BT, Eaton CB, Chen Z, Women’s Health Initiative Investigators. Arthritis increases the risk for fractures-results from the Women’s Health Initiative. J Rheumatol 2011;38:1680-8. [Google Scholar]
- 9.Michel BA, Bloch DA, Wolfe F, Fries JF. Fractures in rheumatoid arthritis: An evaluation of associated risk factors. J Rheumatol 1993;20:1666-9. [Google Scholar]
- 10.Ishida O, Furuya T, Inoue E, Ochi K, Ikari K, Taniguchi A, et al. Risk factors for established vertebral fractures in Japanese patients with rheumatoid arthritis: Results from a large prospective observational cohort study. Mod Rheumatol 2015;25:373-8. [Google Scholar]
- 11.Guañabens N, Olmos JM, Hernández JL, Cerdà D, Calleja CH, López JA, et al. Vertebral fractures are increased in rheumatoid arthritis despite recent therapeutic advances: A case-control study. Osteop Int 2021;32:1333-42. [Google Scholar]
- 12.Ochi K, Inoue E, Furuya T, Ikari K, Toyama Y, Taniguchi A, et al. Ten-year incidence of self-reported non-vertebral fractures in Japanese patients with rheumatoid arthritis: Discrepancy between disease activity control and the incidence of non-vertebral fracture. Osteoporos Int 2015;26:961-8. [Google Scholar]
- 13.Lin PH, Yu SF, Chen JF, Chen YC, Lai HM, Chiu WC, et al. Risk factor analysis of fragility fractures in rheumatoid arthritis: A 3-year longitudinal, real-world, observational, cohort study. PLoS One 2021;16:e0255542. [Google Scholar]







