In distal humerus non-union with preserved mechanical stability and absence of infection, biological augmentation using platelet-rich plasma may offer a conservative treatment option in patients unwilling or unsuitable for revision surgery.
Dr. Anirudh Dwajan, Department of Orthopaedics, All India Institute of Medical Sciences, Bilaspur, Himachal Pradesh, India. E-mail: anirudhdwajan@gmail.com
Introduction: Distal humerus fractures are uncommon injuries with a known risk of non-union despite surgical fixation. Revision surgery with bone grafting remains standard, but some patients may decline reoperation or may be unfit for surgery. Platelet-rich plasma (PRP), an autologous biological product rich in growth factors, may enhance healing in selected cases.
Case Report: A woman in her 30s developed an aseptic non-union of the distal humerus 6 months after internal fixation for a complex fracture. As she declined revision surgery, she was managed non-surgically with autologous PRP injections. Seven injections were administered over 4 consecutive days using anatomical landmarks. Pain progressively reduced, function improved, and radiographs demonstrated bridging callus and cortical continuity by 6 months.
Conclusion: In stable fixation with aseptic atrophic non-union, PRP may act as a minimally invasive biological adjunct to support bone healing. Careful patient selection and monitoring are essential for success.
Keywords: Distal humerus, Non-union, platelet-rich plasma, orthobiologics, bone healing, biological therapy.
Fractures of the distal humerus are relatively uncommon, comprising approximately 2% of all fractures, with an estimated incidence of 5–6 cases/100,000 adults annually. These injuries display a bimodal distribution: Younger individuals typically sustain them through high-energy trauma, such as motor vehicle collisions or sports injuries; whereas older adults, often with osteoporotic bone, more commonly sustain them following low-energy falls. With an ageing global population, the frequency of these injuries in elderly patients is projected to rise substantially in the coming decades [1]. Open reduction and internal fixation with pre-contoured locking plates is the mainstay of treatment for displaced distal humerus fractures. Despite advancements in surgical technique and implant design, non-union remains a relatively frequent complication. Reported rates of non-union following internal fixation of distal humerus fractures range between 8% and 25%, which is significantly higher than those reported for proximal or diaphyseal humerus fractures [2,3]. Management of distal humerus non-union typically involves revision surgery with hardware removal, re-fixation, and bone grafting. However, this approach may not be feasible for all patients due to personal, medical, or socioeconomic reasons. In selected cases, alternative biological strategies may offer potential benefits. Platelet-rich plasma (PRP), derived from autologous blood and concentrated in growth factors, has been explored as a supportive adjunct in bone healing. Although evidence remains limited, PRP has shown promise in enhancing osteogenesis, particularly in cases of delayed healing or non-union. This case describes the successful use of PRP as a non-surgical treatment option in a patient with humeral non-union who declined revision surgery [4].
Patient information
A woman in her 30s from South Asia presented to our tertiary orthopedic unit with persistent pain, restricted range of motion, and functional impairment in her left upper limb. These symptoms had been present for several months following surgical fixation of a distal humerus intercondylar fracture sustained after a fall on an outstretched hand while descending stairs. At the time of injury, she was evaluated at a local trauma center, where radiographs revealed a displaced intercondylar distal humerus fracture with a concurrent fracture of the proximal shaft of the ipsilateral ulna (Fig. 1). She underwent open reduction and internal fixation using dual plating for the distal humerus and plating for the ulnar fracture (Fig. 2). The limb was immobilized in a splint for 10 days, followed by supervised physiotherapy. The ulnar fracture showed progressive healing and achieved union by 3 months post-operatively.
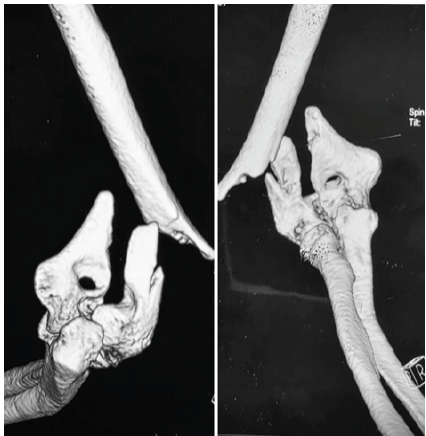
Figure 1: Pre-operative 3D computed tomography reconstructions showing a displaced intercondylar fracture of the distal humerus with associated ipsilateral proximal ulnar shaft fracture.
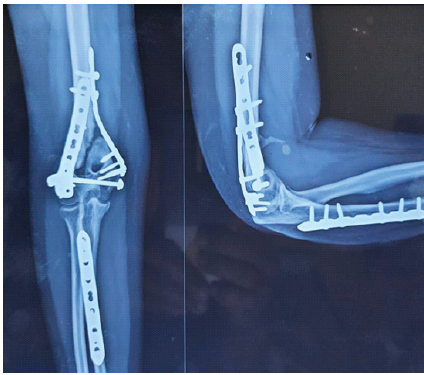
Figure 2: Immediate post-operative anteroposterior and lateral radiographs showing stable dual-plate fixation of the distal humerus with associated plate fixation of the ipsilateral ulna.
Despite adherence to physiotherapy, the patient reported persistent pain and elbow stiffness at 6 months. She experienced difficulty performing daily activities, such as dressing and grooming.
Clinical examination and laboratory assessment
On presentation, there was localized tenderness over the medial and posterior aspects of the elbow, and the range of motion was limited to 40–70° in flexion–extension, with pain at extremes of movement. Forearm pronation and supination were preserved. The surgical scars were well-healed with no local inflammatory signs. Laboratory investigations, including white blood cell count, erythrocyte sedimentation rate, and C-reactive protein, were within normal limits. Serum calcium, phosphate, and vitamin D levels were normal. These findings excluded infection or metabolic bone disease.
Radiological evaluation
Serial radiographs showed satisfactory alignment and intact fixation of both the distal humerus and ulnar fractures. The ulnar fracture had united, while the distal humerus displayed persistent fracture lines and absence of callus formation consistent with non-union. At 6 months post-operatively, there was no bridging callus, and the fracture ends appeared atrophic (Fig. 3). Hardware remained intact, and no malalignment or implant failure was seen.
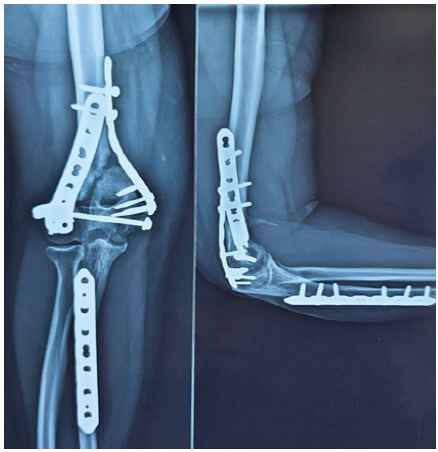
Figure 3: Six-month post-operative anteroposterior and lateral radiographs showing persistent distal humerus non-union with visible fracture lines and intact fixation hardware.
Since plain radiographs adequately defined the pathology, a CT scan was considered unnecessary. The diagnosis of aseptic atrophic non-union of the distal humerus was established.
Intervention
The patient was counseled regarding revision surgery with bone grafting, which remains the gold standard for humeral non-union. However, she declined further surgical intervention due to financial and personal concerns. After discussing the limited but emerging evidence for biological therapy, a conservative approach using autologous PRP was selected.
PRP preparation
PRP was prepared from peripheral venous blood using a standardized double-spin centrifugation protocol. The first spin was performed at 100 g for 15 min to separate red blood cells from plasma and buffy coat. The second spin at 1600 g for 20 min concentrated platelets within the lower third of the plasma volume. The resulting product was leukocyte-poor PRP, containing a high concentration of platelets and growth factors without the use of commercial kits or activation agents. This technique yielded approximately a six-fold increase in platelet concentration while preserving cell integrity, as described in recent literature.
Injection protocol
A total of seven injections of 4 mL each were administered over 4 consecutive days, targeting the peri-fracture region using anatomical landmarks under strict aseptic precautions. Image guidance was not used since the fracture site was superficial and easily palpable. The injections were performed in an outpatient setting, and the patient tolerated the procedure well with no local or systemic adverse effects (Fig. 4).
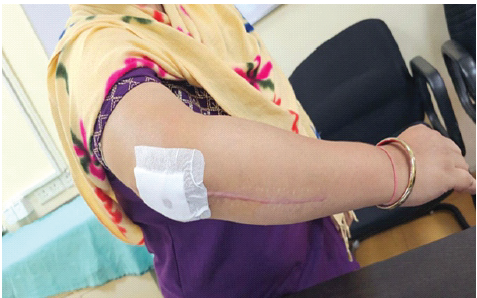
Figure 4: Clinical photograph showing post-procedure dressing following peri-fracture platelet-rich plasma injection administered under aseptic conditions.
Rehabilitation and follow-up
The patient continued her established upper-limb physiotherapy regimen, focusing on pain-free active and passive range of motion, scapulothoracic stability, and progressive strengthening. No additional immobilization or bracing was applied. Analgesia was limited to oral paracetamol (500 mg as needed, maximum 2 g/day), and no non-steroidal anti-inflammatory drugs or bone metabolism agents (such as bisphosphonates or teriparatide) were used. Follow-up evaluations were conducted monthly, including pain assessment, range of motion measurement, and serial radiographs to monitor healing progression.
The patient was followed over a 6 months period following completion of PRP therapy. She attended monthly outpatient visits where pain, range of motion, and radiographic healing were assessed. At 1 month post-treatment, she reported reduced pain and improved use of the arm, although imaging showed no significant change at the fracture site. Despite this, she resumed light household tasks and was able to use the arm for basic activities without major discomfort. By the 3rd month, she demonstrated progressive improvement in function. At 4 months, radiographs revealed visible bridging callus across the distal humerus (Fig. 5), and by 6 months, cortical continuity was seen, confirming radiological union (Fig. 6).
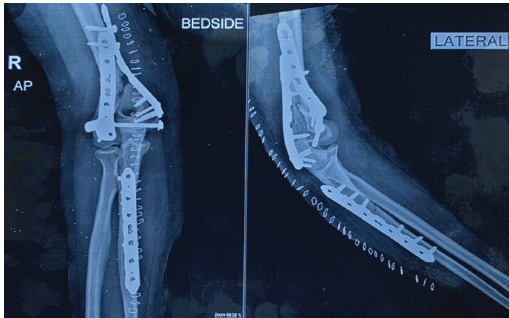
Figure 5: Four-month post-platelet-rich plasma anteroposterior and lateral radiographs showing early bridging callus formation at the distal humerus fracture site with stable fixation in situ.
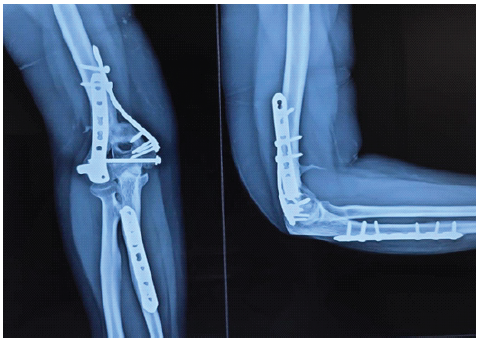
Figure 6: Six-month post-platelet-rich plasma anteroposterior and lateral radiographs demonstrating cortical continuity and radiological union of the distal humerus with stable fixation in situ.
By the end of follow-up, she had regained a flexion-extension arc of approximately 20–90°, with full pronation and supination (Fig. 7). While flexion remained limited, the elbow was stable and pain-free in the functional range. She was able to resume self-care and light household activities, which she had been unable to do previously.
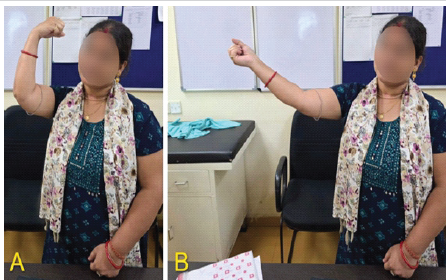
Figure 7: Clinical photographs at 6-month follow-up demonstrating active elbow range of motion showing (a) flexion and (b) extension.
The patient was discharged from routine follow-up with a personalized home-based physiotherapy plan. She was advised to return for re-evaluation if symptoms worsened or if further functional improvement was required. At the time of writing, she remains alive, independent, and clinically well.
Distal humerus fractures are technically demanding injuries due to their anatomical complexity, intra-articular involvement, and relatively thin cortical bone in the supracondylar region. In younger adults, they often result from high-energy trauma, leading to comminuted fractures with compromised vascular supply. Although internal fixation remains the standard approach, non-union may occur due to inadequate biological healing, mechanical instability, or patient-related factors, such as smoking, comorbidities, or early return to activity. In this case, the patient experienced non-union despite adequate implant positioning and maintained alignment, with no evidence of infection or systemic causes of delayed healing [5].
Distal humerus non-union remains a difficult clinical challenge despite modern fixation techniques. It may present as either a hypertrophic (biologically active) or atrophic (biologically inactive) non-union. In this case, the radiographic appearance suggested an atrophic non-union, characterized by an absence of callus and minimal biological activity at the fracture site. Non-union at the distal humerus can occur in either the extra-articular (supracondylar) or intra-articular regions (trochlea or capitellum). The supracondylar area is particularly prone to healing failure due to its limited blood supply and the mechanical forces that traverse the region. In normal biomechanics, loads from the elbow joint are transmitted through the supracondylar area into the diaphysis. However, in cases where fixation is inadequate or stability is compromised, movement at the elbow can result in repetitive micromotion at the fracture site. This so-called “windshield-wiper effect” leads to stress concentration on the hardware and further disrupts the healing process. Additional risk factors include severe soft tissue trauma, poor bone quality – particularly in osteoporotic patients – and patient-related factors, such as smoking or use of medications that impair bone regeneration. Joint stiffness, common after elbow injuries, can also contribute by shifting movement through the fracture site rather than the joint itself, increasing the mechanical strain on an already compromised union [6]. Although fixation appeared radiographically stable in this patient, biological healing had clearly failed. The patient declined revision surgery involving bone grafting and re-plating. Given this, a biologic strategy was pursued. Among available orthobiologics – such as bone morphogenetic proteins, mesenchymal stem cells, and PRP – the latter was selected due to its autologous nature, safety profile, accessibility, and lower regulatory barriers. PRP is an autologous concentration of platelets suspended in plasma, typically derived from peripheral blood through centrifugation. When activated, PRP releases a cascade of bioactive molecules – most notably platelet-derived growth factor, transforming growth factor-beta, vascular endothelial growth factor, and insulin-like growth factor – which collectively stimulate angiogenesis, fibroblast proliferation, osteoblastic differentiation, and extracellular matrix synthesis. These effects support its use in a range of musculoskeletal applications, including tendinopathies, osteoarthritis, and delayed bone healing [7,8]. While there is variability in PRP formulations, a leukocyte-poor PRP was used in this case, administered through multiple peri-fracture injections over a short interval. In our case, PRP was prepared using a standardized double-spin method, as supported by recent literature recommending it as an effective, low-cost protocol for clinical use. Whole blood was first centrifuged at 100 g for 15 min to separate red blood cells from plasma and buffy coat. A second centrifugation at 1600 g for 20 min was then performed to concentrate platelets at the bottom of the plasma column. The lower one-third of the resulting plasma – containing the highest platelet concentration – was collected for injection. This technique has demonstrated a high platelet yield (up to 99%), a 6-fold concentration over baseline, and maintains platelet membrane integrity without requiring activation agents. Such a protocol is particularly beneficial in low-resource settings where commercial kits may not be available, offering a reproducible and biologically effective PRP preparation for musculoskeletal applications [9]. By 4 months post-injection, radiographs showed early callus formation, with cortical continuity confirmed at 6 months. Notably, the patient reported reduced pain and improved mobility within the 1st month of therapy – clinical progress that preceded radiographic evidence of healing. This highlights the importance of functional monitoring in conservative fracture management. No adverse effects were observed during or after PRP administration. This case adds to the small but growing body of literature suggesting a role for PRP in the conservative treatment of long bone non-unions [10,11,12,13]. Unlike prior reports where PRP was used as an adjunct to revision surgery this case suggests a potential role for PRP as a standalone biological adjunct in carefully selected patients. While the outcome in this case is encouraging, the possibility of delayed natural healing cannot be excluded. The lack of a control group and a single-subject design limit generalizability. Moreover, the clinical improvement seen before radiological union raises the question of whether subjective recovery influenced perceived benefit.
Other conservative options for managing non-union include low-intensity pulsed ultrasound and electrical stimulation, both of which have shown promise in selected long bone non-unions. However, these require prolonged treatment durations and may be less accessible in low-resource settings compared to PRP, which can be administered with basic clinical infrastructure. In atrophic non-unions, the local biological milieu lacks sufficient osteogenic and angiogenic stimuli. PRP’s concentrated growth factors offer targeted biological stimulation, which may bridge the healing gap in such cases. This makes PRP an appealing adjunct, particularly when mechanical stability is already optimized, as in the present case. It is important to note that PRP is not yet universally accepted as standard care for fracture non-union. Guidelines are evolving, and clinical use requires clear patient counseling and ethical consideration regarding expectations and outcomes. Future prospective studies are warranted to define the role of PRP in fracture non-union, including optimal dosage, frequency, and patient selection criteria. Cost-effectiveness and long-term outcomes should also be evaluated.
Limitations
This report describes the outcome in a single patient, which limits generalizability. The absence of a comparator group prevents definitive attribution of fracture union solely to PRP. However, the documented clinical and radiological improvement following PRP therapy in the setting of previously failed healing suggests a possible biological contribution that merits further investigation. Radiological union was assessed using serial plain radiographs without computed tomography confirmation. Although CT could provide additional structural detail, radiographic union combined with functional recovery remains widely accepted in clinical practice. Standardized functional outcome scores were not formally recorded; however, meaningful improvement in pain, range of motion, and daily functional ability was documented. Follow-up was limited to 6 months after union, and longer surveillance would help assess durability of healing. PRP injections were administered using anatomical landmark guidance without image assistance, and platelet concentration or growth factor quantification was not performed. The injection protocol reflected an individualized biological treatment approach rather than a standardized regimen. While ongoing physiotherapy may have contributed to recovery, rehabilitation had been continued before PRP therapy without evidence of union, suggesting a potential adjunctive role of PRP.
• Distal humerus non-union remains a difficult complication after fixation
• PRP provides autologous growth factors that enhance angiogenesis and osteogenesis
• Biological stimulation is feasible when mechanical stability exists and infection is excluded
• Image-guided injection is ideal but may not be mandatory when the fracture site is superficial and palpable
• This case highlights PRP as a potential biological adjunct in selected patients who decline or are unfit for revision surgery, although larger studies are required to confirm efficacy.
Autologous PRP may provide a feasible biological adjunct in selected cases of aseptic distal humerus non-union with preserved mechanical stability. However, treatment decisions should be individualized, and further studies are required to establish standardized protocols.
References
- 1. De Crescenzo A, Garofalo R, Bartoli M, Pederzini LA, Celli A. Nonunion of distal humeral fractures: Current concepts. J ISAKOS 2024;9:100294. [Google Scholar] [PubMed]
- 2. Morrey ME, Morrey BF, Sanchez-Sotelo J, Barlow JD, O’Driscoll S. A review of the surgical management of distal humerus fractures and nonunions: From fixation to arthroplasty. J Clin Orthop Trauma 2021;20:101477. [Google Scholar] [PubMed]
- 3. Vauclair F, Goetti P, Nguyen NT, Sanchez-Sotelo J. Distal humerus nonunion: Evaluation and management. EFORT Open Rev 2020;5:289-98. [Google Scholar] [PubMed]
- 4. Andersen C, Wragg NM, Shariatzadeh M, Wilson SL. The use of platelet-rich plasma (prp) for the management of non-union fractures. Curr Osteoporos Rep 2021;19:1-14. [Google Scholar] [PubMed]
- 5. Yetter TR, Weatherby PJ, Somerson JS. Complications of articular distal humeral fracture fixation: A systematic review and meta-analysis. J Shoulder Elbow Surg 2021;30:1957-67. [Google Scholar] [PubMed]
- 6. Thakkar YN, Bava SS, Phunde R, Soni H, Haider H. Windshield wiper effect in human bone-peri-implant benign diaphyseal expansile lesion with humerus shaft fracture non-union with implant in situ – case report. J Orthop Case Rep 2023;13:32-7. [Google Scholar] [PubMed]
- 7. Collins T, Alexander D, Barkatali B. Platelet-rich plasma: A narrative review. EFORT Open Rev 2021;6:225-35. [Google Scholar] [PubMed]
- 8. Pretorius J, Habash M, Ghobrial B, Alnajjar R, Ellanti P. Current status and advancements in platelet-rich plasma therapy. Cureus 2023;15:e47176. [Google Scholar] [PubMed]
- 9. Muthu S, Krishnan A, Ramanathan KR. Standardization and validation of a conventional high yield platelet-rich plasma preparation protocol. Ann Med Surg (Lond) 2022;82:104593. [Google Scholar] [PubMed]
- 10. Ranjan R, Kumar R, Jeyaraman M, Arora A, Kumar S, Nallakumarasamy A. Autologous platelet-rich plasma in the delayed union of long bone fractures – a quasi experimental study. J Orthop 2022;36:76-81. [Google Scholar] [PubMed]
- 11. Li S, Xing F, Luo R, Liu M. Clinical effectiveness of platelet-rich plasma for long-bone delayed union and nonunion: A systematic review and meta-analysis. Front Med (Lausanne) 2022;8:771252. [Google Scholar] [PubMed]
- 12. Gómez-Barrena E, Rosset P, Lozano D, Stanovici J, Ermthaller C, Gerbhard F. Bone fracture healing: Cell therapy in delayed unions and nonunions. Bone 2015;70:93-101. [Google Scholar] [PubMed]
- 13. Hussain N, Johal H, Bhandari M. An evidence-based evaluation on the use of platelet rich plasma in orthopedics – a review of the literature. SICOT J 2017;3:57. [Google Scholar] [PubMed]











