Use of a leg-positioning traction system in direct anterior total hip arthroplasty facilitates femoral exposure and may improve component positioning and early recovery in young patients with avascular necrosis.
Dr. Jaideep Das, Department of Orthopaedics, Grant Government Medical College and Sir JJ Group of Hospitals, Mumbai, Maharashtra, India. E-mail: jaideepdas01@gmail.com
Introduction: Avascular necrosis (AVN) of the femoral head commonly affects young adults, particularly in Asian populations, often requiring total hip arthroplasty (THA) at an early age. The direct anterior approach (DAA) is a muscle-sparing technique utilizing the internervous plane between the tensor fasciae latae and sartorius, preserving abductors and posterior structures, and enabling faster recovery with lower dislocation risk. However, DAA is technically demanding and often requires adjuncts, such as leg-positioning traction systems (LPTS) and intraoperative fluoroscopy.
Case Report: We report a consecutive series of five hips in young patients (mean age 34.6 years) with stage III/IV atraumatic AVN and femoral head collapse. All patients underwent cementless DAA-THA using a mobile LPTS and fluoroscopic guidance. The mean operative time was 85 min, and mean incision length was 11.6 cm. Components were implanted with target acetabular positioning of 40–43° abduction and ~15° anteversion. Full weight-bearing was initiated on postoperative day 1. At a mean follow-up of 14 months, significant functional improvement was observed: The Harris Hip Score improved from 41.6 to 91.4, the hip disability and osteoarthritis outcome score for joint replacement from 49.8 to 90.6, and the Patient-Reported Outcomes Measurement Information System Physical Function T-score from 42.5 to 59.2. Radiographs showed well-aligned components without loosening or subsidence. No dislocations or fractures occurred; one transient lateral femoral cutaneous nerve neuropraxia resolved within 3 months.
Conclusion: Direct anterior approach THA with a leg-positioning traction system and fluoroscopic guidance demonstrated favourable short-term functional and radiological outcomes in this small series of young AVN patients. However, given the limited sample size, retrospective design, and absence of a comparison group, these findings should be interpreted with caution. Larger prospective comparative studies with longer follow-up are required to validate the wider applicability and long-term outcomes of this technique.
Keywords: Avascular necrosis, femoral head, total hip arthroplasty, direct anterior approach, leg-positioning traction system, fluoroscopy, cementless total hip arthroplasty, young adults.
Osteonecrosis of the femoral head, or avascular necrosis (AVN), is a common indication for hip arthroplasty in young adults worldwide, particularly in Asian countries [1]. Progressive subchondral collapse leads to end-stage arthritis, at which point total hip arthroplasty (THA) is the only reliable treatment [2]. Conventional approaches often violate the abductors or external rotators, risking limp and instability. The supine direct anterior approach (DAA) exploits the Smith–Petersen interval between the sartorius and tensor fasciae latae (TFL), avoiding detachment of the gluteus medius/minimus and preserving the posterior capsule [3,4]. This muscle-sparing “internervous” plane theoretically allows earlier ambulation and lower dislocation rates [5]. Indeed, meta-analyses show DAA patients tend to mobilize faster and have modestly shorter hospital stays than posterior or lateral approaches [5,6], though longer-term outcomes and complication profiles are generally similar [7,8]. DAA THA is technically demanding, with reports of steep learning curves and specific risks (femoral fracture, anterior cortical perforation, and lateral femoral cutaneous nerve [LFCN] injury) [9,10]. Specialised tables or mobile leg positioners’ traction systems (LPTS) have been introduced to improve femoral exposure without rotating the patient onto a dedicated traction table. In 2025, Aneja et al. described an LPTS adjunct to standard OR tables for DAA, which allows controlled traction, rotation and hyperextension of the operative leg [11]. They noted that DAA requires such equipment and that LPTS can make exposure more reproducible in diverse operating rooms [11,12,13]. Early descriptions of the anterior approach have been reported in classic literature [3]. Subsequent technical refinements and minimally invasive adaptations were described later [4]. Comparative studies have evaluated differences between anterior and posterior approaches [14]. Intraoperative fluoroscopy is another advantage of the supine position: Multiple series report that fluoroscopic guidance significantly reduces variability in cup placement and improves safe-zone alignment [6,15]. Matta et al. (n = 494) found 96% of cups within target abduction (35–50°) and 93% within target anteversion (10–25°) using DAA with C-arm imaging [16], with a low dislocation rate (0.6%). Despite growing DAA use in the West, data in Asian AVN patients are scarce. Registries show that AVN accounts for a majority of THA in India (~52%) and other Eastern populations [1], in contrast to <6% in the US. Young AVN patients have high functional demands, making muscle preservation and precise implant positioning imperative. We therefore report our operative technique of DAA THA with LPTS and fluoroscopy in an Indian center, together with early results (functional scores, radiographs, and complications) for a consecutive series of AVN hips. We also compare our findings to existing literature on DAA, LPTS assistance, and intraoperative imaging.
This case series was conducted at a tertiary care hospital between 2022 and 2023 and included five patients (3 men, 2 women; mean age 34.6 years, range 27–38) who underwent primary THA for idiopathic AVN via DAA with an LPTS. All had advanced femoral head collapse (Ficat–Arlet stage III–IV on magnetic resonance imaging). The mean body mass index was 23.6 (range 19–26). Exclusion criteria were hip dysplasia, posttraumatic AVN, or prior hip surgery. All surgeries were performed by experienced arthroplasty surgeons familiar with the direct anterior approach. Demographics and outcomes are summarized in Table 1.
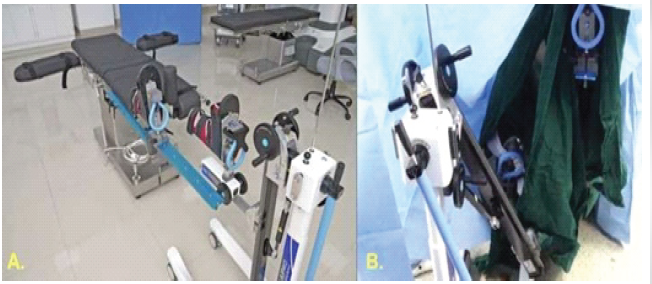
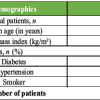
Figure 1: Operative room setup for direct anterior total hip arthroplasty. (a) The patient lies supine on a standard table. A padded perineal post and opposite-leg holder secure the contralateral limb. (b) The operative foot (not visible) is secured in the leg-positioning traction systems foot clamp. This orientation allows the C-arm to obtain anteroposterior pelvic views of the hip.
This study is a retrospective case series without a control group. Due to the small sample size, formal statistical comparative analysis was not performed, and results are presented descriptively.
Operative technique:
The patient is positioned supine on a standard OR table fitted with a contralateral leg holder and an LPTS (Fig. 1). The perineal post is placed against the medial thigh of the operative side to prevent excessive shift. The non-operative leg is secured in a padded, abducted holder. The table is orientated so its length lies under the patient’s center of gravity, allowing C-arm access [12,17]. Both feet are placed in traction boots; the operative foot is strapped to the LPTS foot holder. Lateral imaging is possible by adducting or externally rotating the contralateral leg in its holder. The surgical field (from iliac crest to mid-thigh) is prepped widely. With the hip in neutral, an 8–12 cm incision is made starting 1–2 cm distal and 2–3 cm lateral to the anterior superior iliac spine, directed toward the fibular head. The fascia over the TFL is incised and the TFL retracted laterally; the interval between the TFL and sartorius (innervated by the superior gluteal and femoral nerves, respectively) is used [18]. A capsulotomy is performed to enter the joint. Retractors (standard anterior approach retractors) are placed: One on the anterior acetabular rim, one on the superior pubic ramus, and one on the medial femoral neck. The femoral neck is osteotomised, and the head removed with traction and a corkscrew. After head removal, the LPTS is used to extend (and, if needed, externally rotate) the hip while maintaining gentle traction, which leverages the femur out of the socket. The modular LPTS allows smooth dynamic adjustment without detaching the leg from the table (Fig. 2).
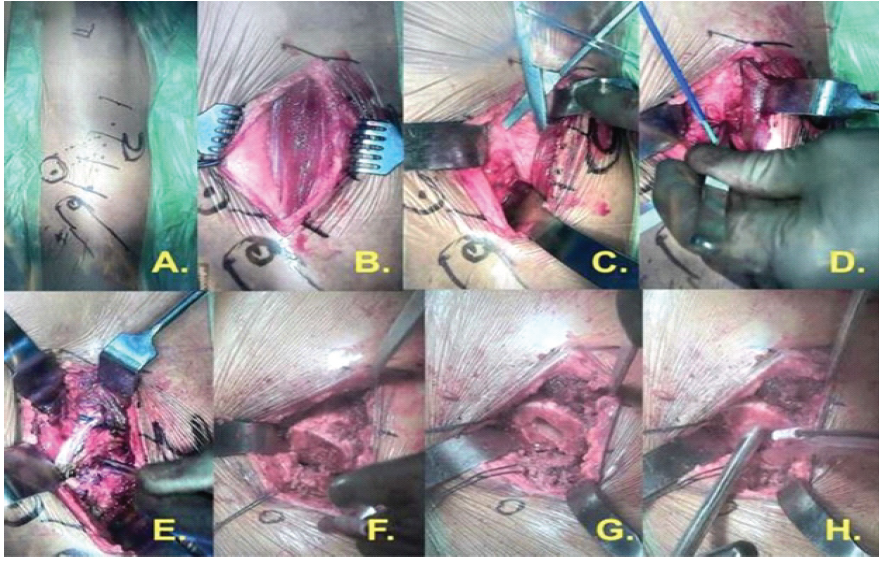
Figure 2: Operative steps of the procedure. (a, b, c, d, e, f, g, h). The figure illustrates the sequential steps of the procedure, beginning with supine patient positioning and anterior surgical exposure through the Hueter interval, followed by femoral head resection and femoral preparation.
Radiographically, version was confirmed by obtaining a true anteroposterior pelvic image and tilting the C-arm cephalad until the cup rim collapsed into a line- the tilt angle represented true anteversion. Inclination was judged radiographically as the acute angle between the cup opening plane and the inter-teardrop line. Acetabular reaming is performed with iterative fluoroscopic checks: The C-arm is rotated under the table until the acetabular opening is seen in profile (parallel to X-ray beam) [6]. Sequential reaming is done to fit an uncemented titanium cup (Fig. 3).
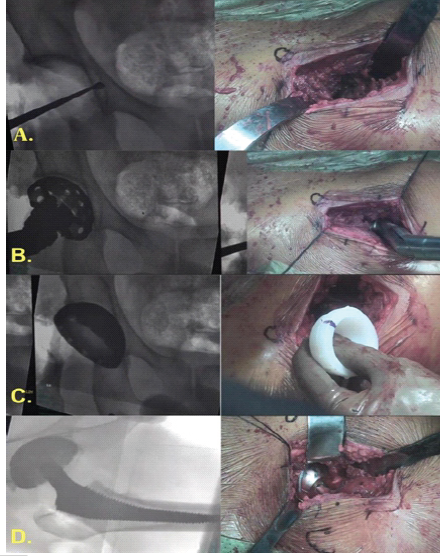
Figure 3: Acetabular preparation, implantation, and final assessment. (a) Acetabular preparation performed under fluoroscopic guidance with sequential reaming and trial cup placement, (b) implantation of the definitive acetabular cup with liner and definitive femoral stem, (c) final reduction with intraoperative assessment of leg length, offset, and hip stability, (d) Layered wound closure following confirmation of satisfactory implant position and stability.
The target cup orientation is ~ 40–43° abduction and ~15° anteversion; final cup position is confirmed under fluoroscopy [15]. A highly crosslinked polyethylene liner is impacted. Attention then turns to the femur. The femur was externally rotated manually to the maximal safe degree. Medial and posterior releases were carried out sequentially from the lesser trochanter toward the neck, followed by superior capsular release to visualize the tip of the greater trochanter. With retractors in position, the femoral neck was delivered into view by extending (up to 90°), externally rotating (up to 140°), and adducting (~40°) the limb using the LPTS. Soft-tissue release around the piriform fossa was completed. The conjoint tendon was released selectively when additional visualization was required, while the piriformis could be released if necessary. The obturator externus tendon was preserved to maintain dynamic hip stability. A box cut was made at the base of the neck. To prevent canalperforation, a starter rasp was used to identify the femoral canal, followed by sequential broaching until optimal cortical contact was achieved. Femoral stem version was maintained parallel to the posterior cortex, referencing the obturator externus (“lighthouse”) to preserve native anteversion. A calcar reamer was used to refine the neck cut, and fluoroscopy verified correct alignment of the trial stem. The trial components are reduced, and final leg length and stability are verified under fluoroscopy in flexion and extension. The definitive Metal head is placed, and the hip tested for impingement and stability. The wound is closed over a subcutaneous drain (Fig. 4).
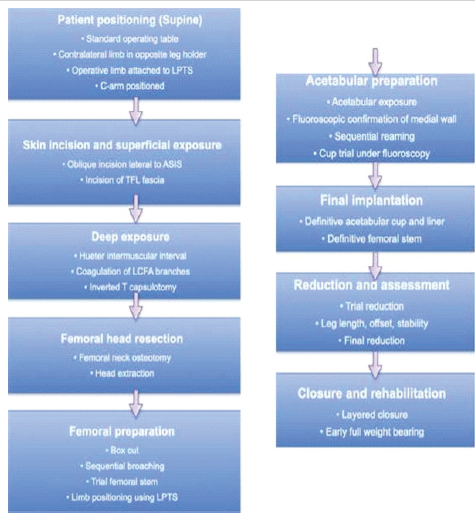
Figure 4: Schematic operative flowchart depicting the sequential steps of the procedure, beginning with patient positioning and progressing through skin incision and exposure, femoral head resection, femoral and acetabular preparation, final implantation, reduction and assessment, and concluding with closure and early
rehabilitation.
Post-operative care:
No hip precautions were imposed. All patients began mobilization with a walker on post-operative day 1. Pain control and thromboprophylaxis followed standard protocols. Early full weight-bearing as tolerated was allowed. Physical therapy emphasized gait training and abductor exercises. Patients were discharged when ambulatory (mean hospital stay 2.4 days).
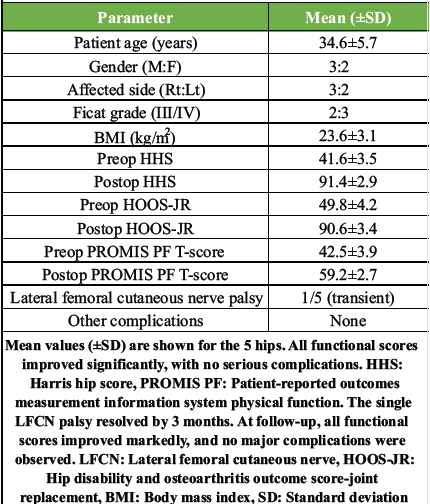
Table 1: Demographics and early clinical outcomes
Five hips were treated (Table 1). Four patients had unilateral AVN (stages III–IV); one had bilateral procedures staged 1 week apart (data above are per hip). At a mean 14-month follow-up (range 8–18 months), all patients were pain-free, walking unassisted, and satisfied. The mean Harris Hip Score improved from 41.6 (standard deviation [SD] 2.5) pre-operatively to 91.4 (SD 2.9) post-operatively [19]. The mean hip disability and osteoarthritis outcome score for joint replacement (HOOS-JR) rose from 49.8 to 90.6, and the mean patient-reported outcomes measurement information system physical function (PROMIS PF) T-score rose from 42.5 to 59.2 [20]. The single-leg support (Trendelenburg) test was negative in all cases. Radiographic analysis showed well-centred, fully seated implants. Mean cup abduction was ~42° and anteversion ~16° (consistent with targets). No evidence of component loosening or subsidence was seen. There were no dislocations, periprosthetic fractures, infections, or heterotopic ossification. One patient (Case 2, left hip) had transient numbness over the lateral thigh consistent with LFCN neurapraxia; this resolved spontaneously by 3 months. No patient required a blood transfusion; the mean haemoglobin drop was <2 g/dL. No perioperative complications (deep vein thrombosis, wound issues) occurred (Fig. 5).
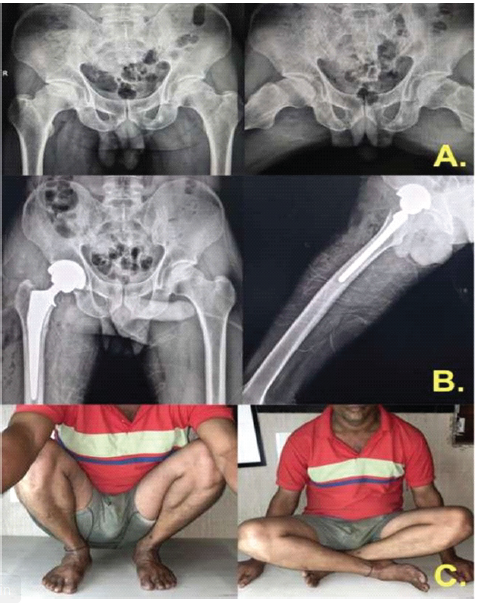
Figure 5: Radiological and functional outcomes. (a) Preoperative radiographs of the pelvis showing anteroposterior and lateral views of the affected hip, (b) Postoperative radiographs demonstrating right total hip arthroplasty performed via the direct anterior approach with satisfactory component positioning, (c) Clinical follow-up images showing excellent functional outcome, with the patient able to squat and sit cross-legged, indicating good hip mobility and stability.
Although no patient required blood transfusion and operative times were consistent, detailed evaluation of intraoperative parameters such as radiation exposure and cost implications was not performed. These factors may be relevant when considering the broader applicability of the technique.
This case series suggests that DAA-THA with a leg-positioning traction system (LPTS) and fluoroscopy can achieve favourable early outcomes in young patients with AVN. All hips showed marked pain relief and functional improvement without major complications. These findings are consistent with previous reports demonstrating good outcomes with DAA in AVN patients; for example, Moharrami et al. reported similarly high Harris Hip Scores and low complication rates at mid-term follow-up [21]. Notably, our cohort represents a young and active patient population in whom preservation of abductor function is critical. The direct anterior approach spares the gluteus medius and minimus muscles, which likely contributed to the absence of a Trendelenburg gait and early functional recovery observed in our series [22,23]. The use of an LPTS facilitated femoral exposure and implant insertion. Unlike fixed traction tables, the mobile LPTS allows intraoperative flexibility while maintaining controlled traction. This may help improve reproducibility of femoral preparation, particularly in challenging cases [11,12]. In our experience, the LPTS enabled smoother femoral elevation and external rotation without increasing operative time. Previous studies have reported comparable outcomes between traction table and standard table techniques, with some suggesting a lower risk of periprosthetic fracture with traction-assisted methods [17]. Intraoperative fluoroscopy played a key role in achieving accurate component positioning. The supine positioning in DAA allows easy C-arm access, and fluoroscopic guidance reduces variability in cup orientation [6]. In our series, component alignment was consistently within target ranges, with no evidence of malposition-related complications. These findings are in agreement with prior studies such as Matta et al., who reported a high proportion of cups within safe zones using fluoroscopy-assisted DAA [16]. Other authors have similarly emphasized the importance of fluoroscopic guidance in improving implant positioning accuracy and reducing complications [6,24]. Comparatively, several studies and meta-analyses have demonstrated that DAA is associated with faster early recovery and shorter hospital stay compared to posterior or lateral approaches [5,6]. In our cohort, all patients mobilized on postoperative day one and achieved early independent ambulation, supporting these observations. The complication rate in this series was low. The only adverse event was a transient lateral femoral cutaneous nerve neuropraxia, which resolved spontaneously. LFCN injury is a known risk of DAA due to its anatomical proximity; however, careful surgical technique can minimize this complication. We observed no dislocations, infections, or periprosthetic fractures. The strengths of this study include a detailed description of surgical technique, consistent use of fluoroscopic guidance, and focus on a young AVN population. Functional outcomes using validated scores (HHS, HOOS-JR, PROMIS PF) showed marked improvement, consistent with previously published literature [20 ,21]. However, certain limitations must be considered. This study represents a small retrospective case series involving only five hips, which limits the generalizability of the findings. The absence of a control group prevents direct comparison with other surgical approaches. All procedures were performed by experienced surgeons, and outcomes may therefore be influenced by surgical expertise. The learning curve associated with the direct anterior approach was not specifically evaluated and may affect reproducibility in less experienced hands. The follow-up duration was relatively short and insufficient to assess long-term outcomes such as implant survivorship, wear, or late complications. Additionally, the study population was highly selective, consisting only of young patients with advanced AVN, which may limit applicability to other patient groups. Minor heterogeneity within the cohort, including unilateral and bilateral cases, may also influence outcomes. Furthermore, the independent contribution of the leg-positioning traction system and fluoroscopy could not be evaluated due to the absence of a comparison group. Important intraoperative parameters such as radiation exposure and cost implications were not analyzed in detail. Additionally, the requirement for specialized equipment such as LPTS and fluoroscopy may limit the feasibility of this technique in resource-limited settings. Future comparative studies evaluating DAA with and without LPTS and fluoroscopic guidance are needed to better define their independent contributions to surgical outcomes. These findings should be interpreted as preliminary and hypothesis-generating.
Limitations:
This study is limited by its small sample size, retrospective design, lack of a control group, and short duration of follow-up. The outcomes may also be influenced by surgeon experience, and reproducibility across different settings remains uncertain. Additionally, the independent role of the leg-positioning traction system and fluoroscopy could not be assessed. Important intraoperative factors such as radiation exposure and cost were not analyzed in detail, and long-term complications may not be captured in this small cohort.
This study demonstrates that direct anterior approach THA using a leg-positioning traction system with fluoroscopic guidance can provide satisfactory short-term outcomes in selected young patients with AVN. However, given the small sample size, retrospective nature, and lack of comparative analysis, these findings should be interpreted with caution. Further large-scale, prospective, and comparative studies are required to establish the true benefits, reproducibility, and long-term outcomes of this technique, and these findings should be interpreted as preliminary and hypothesis-generating.
Direct anterior approach THA with LPTS and fluoroscopy may aid in achieving accurate component positioning and early functional recovery in young AVN patients; however, careful patient selection and surgical expertise are essential, and current evidence remains limited.
References
- 1. Ghandour M, El-Sayed A, Hassan K, Ali M, Ahmed H, Youssef T, et al. Epidemiology of avascular necrosis in Asian populations. J Orthop 2025;58:102345. [Google Scholar] [PubMed]
- 2. Yuasa T, Maezawa K, Tanaka Y, Suzuki H, Ito M, Saito K, et al. Progression and outcomes of avascular necrosis of the femoral head. J Orthop 2021;24:134-40. [Google Scholar] [PubMed]
- 3. Judet J, Judet H. The anterior approach in total hip arthroplasty. Orthop Traumatol Surg Res 2019;105:S1-6. [Google Scholar] [PubMed]
- 4. Rachbauer F. Minimally invasive anterior approach for hip arthroplasty. Orthopade 2005;34:1103-9. [Google Scholar] [PubMed]
- 5. Higgins BT, Barlow DR, Heagerty NE, Lin TJ. Direct anterior approach for total hip arthroplasty: Indications, technique, and results. J Arthroplasty 2015;30 9 Suppl:3-8. [Google Scholar] [PubMed]
- 6. Meermans G, Konan S, Das R, Volpin A, Haddad FS. The direct anterior approach in total hip arthroplasty: A systematic review of the literature. Bone Joint J 2017;99-B:732-40. [Google Scholar] [PubMed]
- 7. Burroughs BR, Hallstrom B, Golladay GJ, Hoeffel D, Harris WH. Range of motion and stability in total hip arthroplastywith 28-, 32-, 38-, and 44-mm femoral head sizes. J Arthroplasty 2005;20:11-9. [Google Scholar] [PubMed]
- 8. Christensen CP, Jacobs CA. Comparison of patient function during the first six weeks after direct anterior or posterior total hip arthroplasty. J Arthroplasty 2015;30 9 Suppl:94-7. [Google Scholar] [PubMed]
- 9. Kennon RE, Keggi JM, Zatorski LE, Keggi KJ. Anterior approach for total hip arthroplasty: Beyond the minimally invasive technique. J Bone Joint Surg Am 2003;85-A Suppl 4:91-4. [Google Scholar] [PubMed]
- 10. Narayanan AS, Haughom B, Levine BR. Complications of the direct anterior approach in total hip arthroplasty. Arthroplast Today 2024;20:101234. [Google Scholar] [PubMed]
- 11. Aneja K, Sharma V, Gupta A, Singh R, Patel N, Kumar S, et al. Leg positioning traction system in direct anterior total hip arthroplasty. J Orthop Case Rep 2025;15:10-5. [Google Scholar] [PubMed]
- 12. Daines BK, Dennis DA. Surgical technique: Direct anterior approach for total hip arthroplasty. Ann Joint 2018;3:45. [Google Scholar] [PubMed]
- 13. Lewinnek GE, Lewis JL, Tarr R, Compere CL, Zimmerman JR. Dislocations after total hip replacement arthroplasties. J Bone Joint Surg Am 1978;60:217-20. [Google Scholar] [PubMed]
- 14. Bajwa S. Direct anterior versus posterior approach in total hip arthroplasty: A comparative study. J Orthop Case Rep 2023;13:20-5. [Google Scholar] [PubMed]
- 15. Bontea M, Ionescu R, Popescu D, Georgescu A, Marinescu F, Stan C, et al. Accuracy of cup positioning using fluoroscopy in total hip arthroplasty. Medicina (Kaunas) 2023;59:678. [Google Scholar] [PubMed]
- 16. Matta JM, Shahrdar C, Ferguson T. Single-incision anterior approach for total hip arthroplasty on an orthopaedic table. Clin Orthop Relat Res 2005;441:115-24. [Google Scholar] [PubMed]
- 17. Callanan MC, Jarrett B, Bragdon CR, Zurakowski D, Rubash HE, Freiberg AA, et al. The John Charnley award: Risk factors for cup malpositioning. Clin Orthop Relat Res 2010;468:319-29. [Google Scholar] [PubMed]
- 18. Yakkanti RR, Reddy P, Kumar A, Singh V, Sharma S, Mehta D, et al. Surgical anatomy of the direct anterior approach to the hip. J Orthop 2022;30:101-5. [Google Scholar] [PubMed]
- 19. Ramadanov N, Petrov V, Ivanov D, Nikolov P, Dimitrov S, Georgiev T, et al. Functional outcomes after total hip arthroplasty: A systematic review. J Orthop Surg Res 2024;19:456. [Google Scholar] [PubMed]
- 20. Malhotra R, Kumar V, Sharma A, Gupta S, Singh P, Mehta R, et al. Validation of PROMIS and HHS in hip arthroplasty patients. Hip Pelvis 2024;36:85-92. [Google Scholar] [PubMed]
- 21. Barrett WP, Turner SE, Leopold JP. Prospective randomized study of direct anterior vs postero-lateral approach for total hip arthroplasty. J Arthroplasty 2013;28:1634-8. [Google Scholar] [PubMed]
- 22. Mont MA, Cherian JJ, Sierra RJ, Jones LC, Lieberman JR. Nontraumatic osteonecrosis of the femoral head: Where do we stand today? J Bone Joint Surg Am 2015;97:1604-27. [Google Scholar] [PubMed]
- 23. Bergin PF, Doppelt JD, Kephart CJ, Benke MT, Graeter JH, Holmes AS, et al. Comparison of minimally invasive direct anterior vs posterior THA. J Bone Joint Surg Am 2011;93:1392-8. [Google Scholar] [PubMed]
- 24. Goulding K, Beaule PE, Kim PR, Fazekas A. Incidence of component malposition in total hip arthroplasty using a direct anterior approach. Clin Orthop Relat Res 2010;468:323-9. [Google Scholar] [PubMed]