This systematic review compares the direct anterior approach (DAA) with conventional hip replacement approaches (posterior/posterolateral/lateral) in primary total hip arthroplasty. The key learning is that DAA may provide modest early recovery benefits: pooled evidence shows slightly less pain at 3 months and better early hip function (Harris Hip Score) at 3 months, plus lower blood loss. However, operative time and hospital stay vary widely across studies, likely influenced by surgeon experience and patient complexity, so efficiency benefits are uncertain. Overall confidence in results is limited because few studies were poolable and many were observational with confounding.
Dr. Satya K Koduru, Department of Trauma and Orthopaedics, NLAG Trust, United Kingdom. E-mail: kodurusk@gmail.com;
Introduction: The direct anterior approach (DAA) is promoted as a muscle-sparing technique for primary total hip arthroplasty (THA), but comparative benefits and safety remain debated across settings and study designs.
Material & Methods: We performed a Preferred Reporting Items for Systematic Reviews and Meta-analyses 2020-guided systematic review of PubMed, EMBASE, Scopus, Web of Science, MEDLINE, DOAJ, EBSCOhost, and SCIE from inception through 31 December 2023. We included comparative clinical studies of adults undergoing primary THA that directly compared DAA with posterior/posterolateral (and lateral, where available) approaches. Risk of bias was assessed using risk of bias 2 (RoB 2) for randomized trials and a ROBINS-I-aligned framework for observational studies. Random-effects meta-analysis (DerSimonian-Laird) was conducted when ≥2 studies reported comparable outcomes at aligned timepoints.
Discussion: The search identified 2,548 records; after removing 632 duplicates, 1,916 records were screened and 178 full texts were assessed. Seven studies (1 randomized trial and 6 observational cohorts; 4,685 participants) met the inclusion criteria. For the primary pooled outcome (pain, Visual Analog Scale at 3 months; 2 studies; 437 participants), DAA showed lower pain versus comparator (mean difference [MD] −0.49, 95% confidence interval [CI] −0.66 to −0.32; I² = 0%). Secondary pooled outcomes suggested a small pain difference at 6 months (MD −0.10, 95% CI −0.18 to −0.02; I² = 0%) and higher early function at 3 months (Harris hip score MD +7.15, 95% CI 5.28 to 9.02; I² = 0%). Estimated blood loss favored DAA (MD −81 mL, 95% CI −92.60 to −69.68; I² = 0%), whereas operative time and length of stay showed substantial heterogeneity and no consistent difference. The randomized trial had some concerns, and observational studies were mainly limited by confounding, yielding an overall moderate-to-serious risk of bias.
Conclusion: DAA may offer modest early improvements in pain and function after primary THA, with uncertain effects on operative efficiency outcomes that vary by context. Overall certainty of evidence is limited by few poolable studies, mixed designs, and residual confounding; further well-designed pragmatic randomized trials and standardized prospective cohorts are needed.
Keywords: Direct anterior approach, total hip arthroplasty, posterior approach, posterolateral approach, systematic review, meta-analysis, Preferred Reporting Items for Systematic Reviews and Meta-analyses 2020.
Total hip arthroplasty (THA) is widely recognized as one of the most effective operations for management of end-stage hip disease, with long-term pain relief and improved function and quality of life among afflicted patients [1,2]. With the extension of life expectancy and the increase in functional demand, not only for old populations but also for active populations, hip arthroplasty needs are rising all over the world. Concomitantly, development of implants and operative techniques has become more focused on not just survival of the implant but also hastening recovery and facilitating restoration of early function [3,4].
The surgical approach to THA is still a significant factor which effects perioperative and postoperative outcomes. The posterior approach is widely used owing to its consistent anatomy and ease of femoral exposure, although damage to posterior soft tissue was reported as an independent risk factor for postoperative dislocation [5,6]. On the other hand, the lateral approach (LA) is believed to provide better stability while having more risk of abductor mechanism deficit resulting in gait disturbance such as postoperative limp or Trendelenburg sign [7,8].
In recent years, the direct anterior approach (DAA) has swept across as an alternative. This method utilizes an internervous and intermuscular plane between the tensor fascia lata and sartorius muscle to prevent dissection of large muscle groups [9,10,11]. The preservation of soft tissues is believed to work by minimizing surgical trauma, promoting early mobilization, reducing postoperative pain, and reducing hospital stay. A number of recent studies have demonstrated better early functional recovery and earlier discharge after DAA than for traditional methods, without an apparent increase in major complications [12].
Although these potential benefits might have been claimed, the superiority and safety of DAA over PA or LA are incompletely established. Published outcomes are affected by various factors, including surgeon skill, patient demographics, and perioperative regimens, which may result in wide ranges of operative time, blood loss, complication rates, and patient-reported outcome measures, for example, the Harris Hip Score, WOMAC, or HOOS- [13]. As a result, single studies led to divergent and partly contradictory effects.
While several meta-analyses attempted to answer these questions, most were hampered by small sample sizes, variable outcome definitions, and incomplete appraisal of bias and certainty of evidence [14,15,16]. In addition, modern methodological quality standards (e.g., Preferred Reporting Items for Systematic Reviews and Meta-analyses [PRISMA] 2020 reporting, Cochrane Risk of Bias 2.0 assessment, and grading of recommendations assessment, development, and evaluation [GRADE] analysis) have not been universally upheld in earlier reviews that limit the robustness of their findings [17,18].
To overcome these limitations, the current systematic review and meta-analysis are intended to offer an updated and methodologically robust direct comparison of the DAA with posterior/lateral approaches in primary THA. It is hoped that, by applying systematic standards and measures of bias, together with consideration of the GRADE system for overall evidence grading, this review will offer insight into the clinical efficacy as well as safety profile for DAA to aid in decision-making with regard to skin incisions in today’s hip arthroplasty practice.
Study design and reporting
We conducted a systematic review of the literature to evaluate clinical outcomes of the DAA compared with posterior/posterolateral approaches (PA/PLA) (and LA when reported) in primary THA. The review was prepared in accordance with PRISMA 2020 reporting guidance also registered with PROSPERO 2025 CRD420251024575. The final literature search was conducted on 31 December 2023 (last search date). Because the final included set contained a mixture of primary comparative clinical studies and secondary evidence syntheses (systematic reviews/meta-analyses), we preplanned a two-stream evidence synthesis:
- Primary comparative clinical studies were eligible for quantitative pooling (meta-analysis) when outcomes and reporting were sufficiently comparable.
- Systematic reviews/meta-analyses and anatomical/cadaveric studies were included for contextual narrative synthesis only and were not pooled with primary clinical studies to avoid double counting of overlapping patient populations.
Eligibility criteria
Eligibility was defined using a PICO framework.
- Population: Adults undergoing primary THA for degenerative or other standard clinical indications.
- Intervention: THA performed using the DAA (including minimally invasive/anterior variants when explicitly described as DAA).
- Comparator: Posterior or posterolateral approach (and LA where directly compared).
- Outcomes: Perioperative outcomes (operative time, blood loss, transfusion, length of stay), functional recovery, and patient-reported outcomes (e.g., Harris hip score [HHS], WOMAC, Oxford hip score), radiographic outcomes (e.g., cup inclination/anteversion), and complications (e.g., dislocation, fracture, infection, nerve injury, revision).
Study designs included:
- Primary comparative clinical studies: Randomized controlled trials, prospective cohort studies, and retrospective comparative studies directly comparing DAA with PA/PLA/LA in primary THA.
- Secondary evidence syntheses: Systematic reviews, meta-analyses, and network meta-analyses addressing DAA versus other approaches in THA (used for contextual interpretation).
- Anatomical/cadaveric studies: Included only if directly relevant to approach-specific complication mechanisms (e.g., nerve anatomy related to DAA) and used as context rather than clinical effect estimation.
Exclusion criteria
Case reports/series without a comparator, editorials/letters, surgical technique notes without comparative outcomes, conference abstracts without full data, studies not involving THA, revision THA-only cohorts, non-human studies, and publications lacking extractable comparative results.
Information sources and search strategy
A comprehensive electronic literature search was performed in PubMed, EMBASE, Scopus, Web of Science, MEDLINE, DOAJ, EBSCOhost, and SCIE. The search included studies available through 31 December 2023, and the final search was conducted on 31 December 2023. Search strategies combined controlled vocabulary (e.g., MeSH/Emtree) and free-text terms related to THA and surgical approach, including: “total hip arthroplasty,” “total hip replacement,” “direct anterior approach,” “anterior approach,” “posterior approach,” “posterolateral approach,” and “lateral approach.” Boolean operators (AND/OR) were applied. Reference lists of included full-text articles and relevant reviews were screened for additional studies. Grey literature sources were checked when feasible.
Study selection and PRISMA classification
All citations were imported into a reference manager, and duplicates were removed. Two reviewers independently screened titles and abstracts, followed by a full-text review of potentially eligible articles. Disagreements were resolved through discussion and consensus (with third-party adjudication when required). A PRISMA flow diagram summarized the selection process.
After full-text inclusion, publications were classified into the following predefined categories:
- Primary comparative clinical studies (eligible for quantitative synthesis)
- Systematic reviews/meta-analyses (contextual narrative synthesis only)
- Anatomical/cadaveric studies (contextual narrative synthesis only).
Data extraction
Data were extracted using a standardized form. For primary comparative clinical studies, we extracted: Author/year, country/setting, study design, enrollment period, sample size overall and by group, baseline characteristics (age/sex/body mass index where available), surgical approach definitions, follow-up duration, perioperative outcomes, functional/PROM outcomes with time points, radiographic outcomes, and complications (including definitions and ascertainment methods). Where necessary, outcome data were converted to consistent units and directionality to enable synthesis. For systematic reviews/meta-analyses, we extracted: review type, databases searched, search dates, number of included primary studies/patients, approach comparisons, outcomes synthesized, statistical model (fixed/random), heterogeneity reporting, and main conclusions. These data were used to contextualize and interpret primary-study findings, not to generate pooled estimates alongside primary studies. For anatomical/cadaveric studies, we extracted methods relevant to the anatomical question (specimen source, dissection protocol, outcomes measured) and key implications for approach-related complications.
Risk of bias and methodological quality assessment
Quality appraisal was performed according to the study type:
- Randomized controlled trials: Cochrane RoB 2 tool
- Non-randomized primary comparative studies: ROBINS-I tool
- Systematic reviews/meta-analyses: A structured critical appraisal tool (e.g., JBI checklist for systematic reviews) was used to assess methodological rigor and reporting quality
- Cadaveric/anatomical studies: An appropriate design-specific appraisal framework was applied (e.g., clarity of specimen selection and measurement methods), with findings used only for contextual interpretation.
All assessments were performed independently by two reviewers, and disagreements were resolved by consensus.
Data synthesis and statistical analysis
We synthesized evidence using two complementary approaches:
- Quantitative synthesis (meta-analysis): Only primary comparative clinical studies were eligible for pooling. Meta-analysis was performed when at least two studies reported the same outcome with sufficiently comparable definitions and time points. A random-effects model was applied to account for clinical and methodological diversity. Continuous outcomes were pooled using mean difference (MD) when measured on the same scale or standardized MD (SMD) when scales differed. Dichotomous outcomes were pooled using risk ratios with 95% confidence intervals (CI). Statistical heterogeneity was evaluated using the I2 Sensitivity analyses were planned based on study design (randomized controlled trial [RCT] vs. non-randomized) and risk-of-bias levels when data permitted.
- Narrative synthesis: Systematic reviews/meta-analyses and cadaveric evidence were summarized narratively to provide context regarding consistency of findings, learning-curve considerations, and approach-specific complication mechanisms. These sources were not pooled with primary clinical studies to avoid duplication of patient populations across evidence syntheses.
Publication bias assessment (e.g., funnel plots) was planned only when ≥10 primary studies contributed to a pooled outcome; otherwise, it was not performed due to limited interpretability.
Study selection (PRISMA)
A comprehensive search of the pre-specified databases identified 2,548 records; no additional records were identified from registers (0). After removing 632 duplicates, 1,916 unique records remained for screening. Title and abstract screening excluded 1,738 records as irrelevant, ineligible by design, or not providing a direct comparison of surgical approaches. A total of 178 reports were sought for retrieval, and all 178 full texts were successfully retrieved (reports not retrieved = 0). Full-text eligibility assessment excluded 169 reports for the following reasons: No direct comparison (n = 54), inadequate reporting of outcomes (n = 42), duplicated data (n = 29), focus on revision surgery or fractures (n = 21), and non-extractable data (n = 22). Ultimately, 7 studies met all inclusion criteria and were included in the review; 7 reports of included studies were retained for synthesis. The study selection process and reasons for exclusion are presented in the PRISMA 2020 flow diagram (Fig. 1).
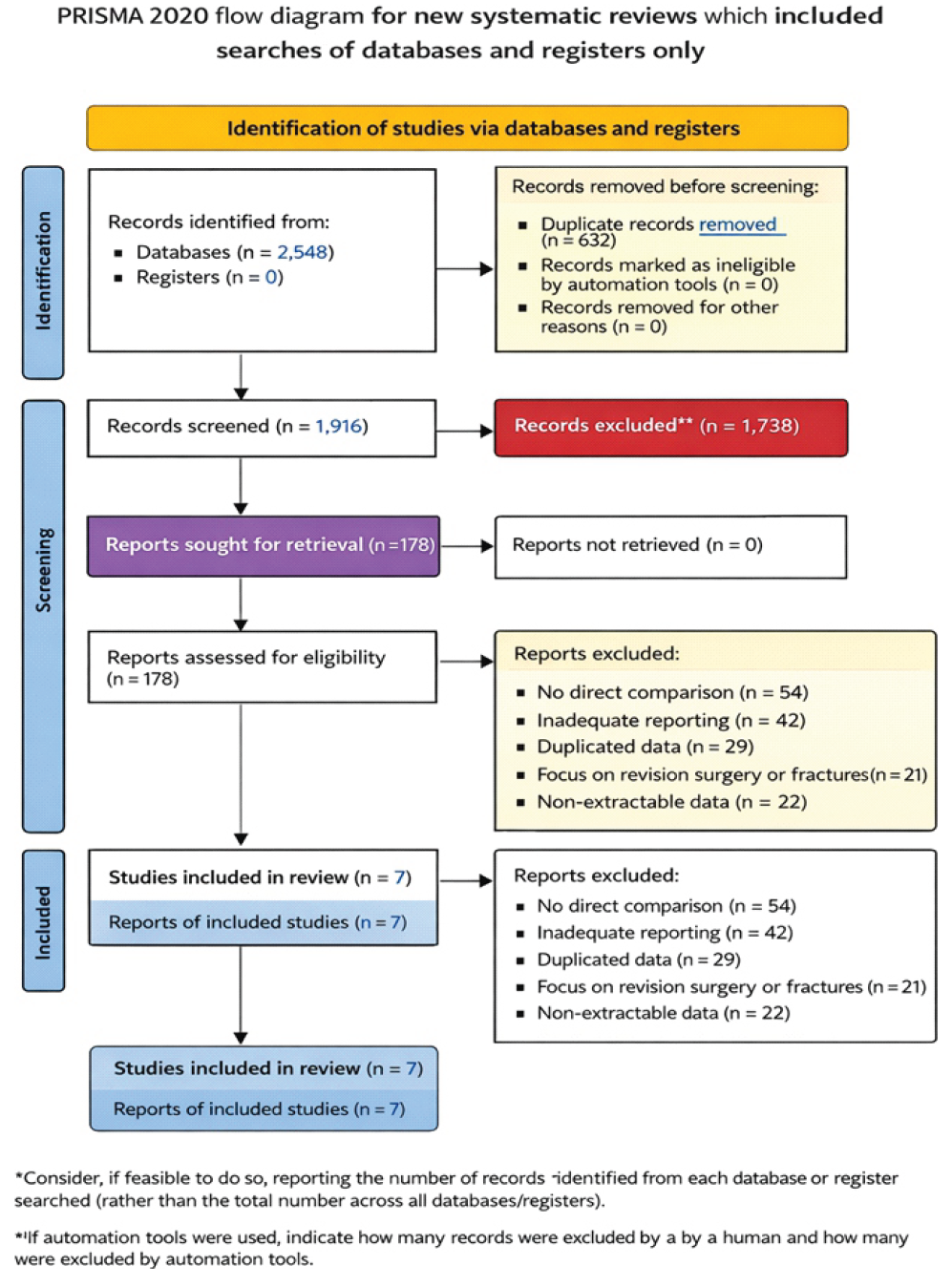
Figure 1: Preferred Reporting Items for Systematic Reviews and Meta-analyses 2020 flow diagram illustrating identification, screening, eligibility assessment, and inclusion, with reasons for full-text exclusions.
Study characteristics (n = 7)
Across the seven included studies, the evidence base comprised one multicenter RCT, one large multicenter prospective cohort, and five observational comparative studies (including matched-pair and retrospective cohorts). Study settings spanned North America (Canada, United States), Europe (Netherlands, Norway), and Asia (China). Most studies evaluated primary THA performed for degenerative hip disease (e.g., osteoarthritis/osteonecrosis), while one study specifically examined severe developmental dysplasia of the hip (DDH) undergoing THA with adjunct procedures. Comparators most frequently involved the posterior/postero LA, with one study including a direct lateral arm. Follow-up ranged from early postoperative periods (days to weeks) to mid- and long-term assessments (up to 5 years in the RCT). Outcomes included perioperative metrics (operative time, blood loss, length of stay), patient-reported and clinician-reported function (e.g., HHS, HOOS Jr, PROMIS), pain scales (Visual Analog Scale [VAS], NPRS), muscle strength testing, radiographic component positioning, and complication rates(Table 1).
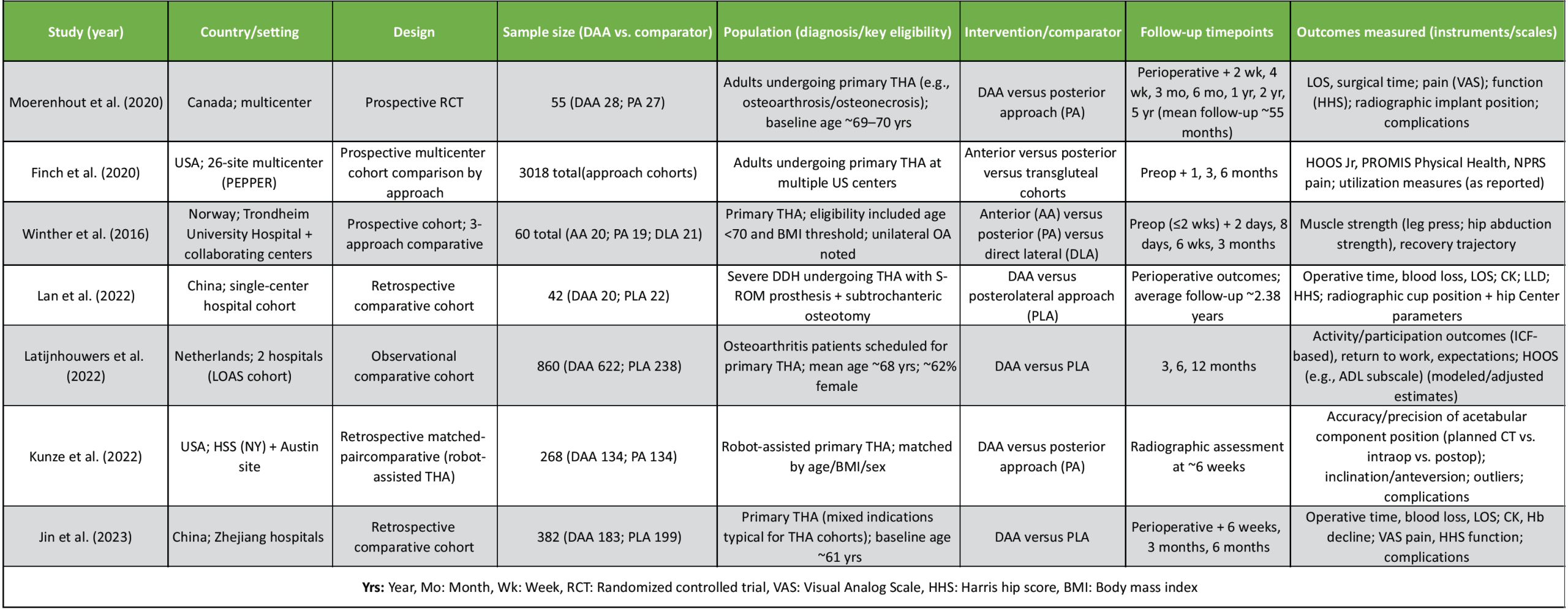
Table 1: Included study characteristics (n = 7)
Risk of bias was evaluated using design-appropriate tools. For RCTs, the Cochrane RoB 2 tool was applied across the standard five domains (randomization process; deviations from intended interventions; missing outcome data; outcome measurement; selective reporting), followed by an overall judgment. For observational comparative studies, risk of bias was assessed using an approach aligned with ROBINS-I (or equivalent observational appraisal), focusing on confounding and selection processes in addition to missing data, outcome measurement, and reporting. Domain-level judgments and overall ratings are summarized in Fig. 2, with the distribution of judgments across domains shown in Fig. 3.
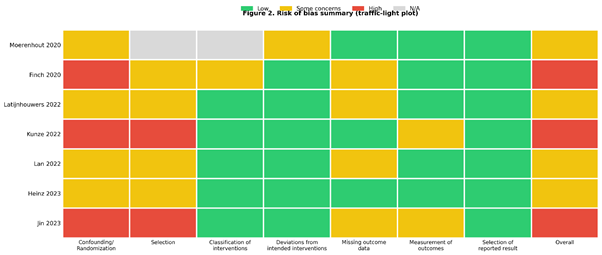
Figure 2: Risk of bias summary (traffic-light plot). Domain-level risk-of-bias judgments for each included study. The randomized controlled trial (Moerenhout et al., 2020) was assessed using risk of bias 2, and observational comparative studies were assessed using ROBINS-I. Cells indicate level of concern in each domain (green = low risk; yellow = some concerns/moderate risk; red = serious/high risk; gray = not applicable). An overall risk-of-bias judgment is provided for each study.
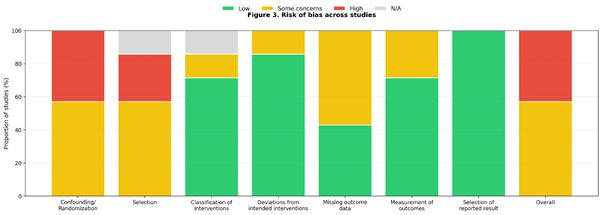
Figure 3: Risk of bias across studies (bar chart). Distribution of risk-of-bias judgments across studies for each domain. Stacked bars show the proportion of studies rated low (green), some concerns/moderate (yellow), and serious/high risk (red); g-ray indicates domains not applicable to a given design and is excluded from the denominator where relevant.
Overall risk of bias (study-level summary)
Among the seven included studies, one was an RCT and six were observational comparative studies. The RCT was judged as overall “some concerns,” primarily related to unavoidable lack of blinding and incomplete reporting of certain randomization details, although outcome measurement procedures were generally robust. Across observational studies, the most consistent limitation was confounding, as surgical approach allocation was typically non-random and influenced by surgeon preference, patient selection, or institutional practice, resulting in overall ratings ranging from moderate to serious risk of bias.
Domain-level findings (what drove bias)
- Randomization/confounding: This was the most frequent source of concern. Most nonrandomized studies were judged moderate-to-serious risk due to potential baseline differences (case-mix, surgeon experience, selection of complex cases for one approach, learning curve effects).
- Selection of participants: Several observational cohorts were judged moderate risk (or serious in smaller, retrospective studies), reflecting possible selection into approach groups.
- Classification of intervention: Generally low risk because the surgical approach is typically clearly documented; however, some multicenter datasets may have pragmatic grouping based on surgeon classification.
- Deviations from intended interventions: Usually low risk across studies, as postoperative pathways were generally standard within sites; however, lack of blinding can still influence subjective recovery pathways.
- Missing outcome data: Several studies had a moderate risk due to incomplete PROM follow-up or loss to follow-up, even when statistical handling (e.g., imputation/sensitivity analysis) was described.
- Outcome measurement: Mostly low to moderate risk; objective outcomes (radiographic measures, LOS) are less vulnerable, whereas PROM-based outcomes may be influenced by non-blinding and follow-up completeness.
- Selective reporting: Judged low overall because outcomes were broadly consistent with typical THA studies, and no clear evidence of selective outcome suppression was identified from the available reporting.
Outcomes reported across studies
Across the seven included studies, outcomes clustered into perioperative efficiency measures, patient-reported and clinician-reported recovery outcomes, radiographic component positioning, and safety endpoints. Perioperative outcomes most consistently reported included operative time (minutes), estimated blood loss (mL), and length of hospital stay (days), enabling direct comparison of early surgical efficiency between approaches. Recovery outcomes were captured using both pain scales and functional instruments. Pain intensity was typically assessed using the VAS and/or the numeric pain rating scale (NPRS). Functional recovery was measured using hip-specific tools such as the HHS and HOOS-based instruments (including HOOS Jr in multicenter PROM cohorts), while general physical health status was reported in some datasets using PROMIS Physical Health (PROMIS-PH). In selected cohorts, additional recovery indicators were reported, including creatine kinase (CK) as a surrogate of perioperative muscle injury and objective muscle performance testing (e.g., leg press and hip abduction strength trajectories). Radiographic outcomes included acetabular component alignment (e.g., inclination/anteversion) and, in studies evaluating technology-assisted THA, accuracy/precision metrics and outlier rates relative to planned targets. Safety outcomes were reported as postoperative complications and adverse events, including approach-related complications (e.g., nerve symptoms), general surgical complications (e.g., infection, fracture, dislocation), and other perioperative events when available.
Given variability in outcome instruments across studies, effect measures were selected to preserve interpretability and comparability. MD was used when outcomes were reported on the same scale/unit across studies (e.g., operative time in minutes, blood loss in mL, hospital stay in days, and VAS pain when reported on a consistent scale). SMD was used when studies assessed a common construct but employed different instruments or scales, particularly for function and health status outcomes (e.g., HOOS Jr vs. HHS, or NPRS vs. VAS when scales were not directly harmonizable). When SMD was required, a small-sample corrected estimator (e.g., Hedges’ g) was appropriate to reduce bias in studies with smaller sample sizes. Quantitative synthesis was performed at clinically meaningful and comparable follow-up timepoints where data permitted, prioritizing aligned assessment windows across studies rather than combining heterogeneous follow-up periods. Timepoints were grouped into early postoperative recovery (in-hospital/≤2 weeks), short-term recovery (approximately 1 month), intermediate recovery (3 months), and later short-term recovery (6 months), with ≥12-month outcomes summarized when available but often limited by inconsistent reporting across the full evidence set. Common PRO instruments used across studies included HOOS Jr, PROMIS-PH, and NPRS, reflecting both joint-specific and global functional recovery domains following THA.
Primary outcome: Pain (VAS) at 3 months.
Pooled effect (overall)
A random-effects model (DerSimonian-Laird) was used because the contributing studies differed in design (RCT vs. cohort) and comparator approach (posterior vs. posterolateral), and therefore a common “true” effect across studies could not be assumed. Two studies contributed to this pooled comparison (Moerenhout et al., 2020 and Jin et al., 2023). The meta-analysis showed significantly lower pain in the DAA group at 3 months: (Fig. 4).
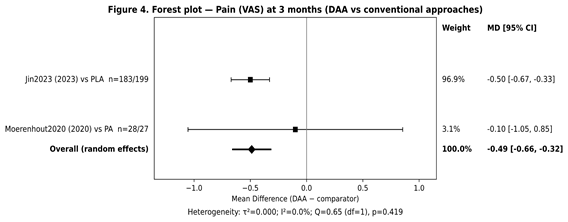
Figure 4: Forest plot – primary outcome (Visual Analog Scale at 3 months) (study names + comparator + n + weights included).
MD (DAA–comparator) = −0.49, 95% CI −0.66 to −0.32, P < 0.001.
Heterogeneity
Statistical heterogeneity for the primary outcome was low:
- I2 = 0.0%
- τ2 = 0.000
- Cochran Q = 0.65 (df = 1), P = 0.419.
These values indicate that the study effects were highly consistent at the 3-month timepoint.
Clinical interpretation
The pooled primary analysis showed a statistically significant reduction in pain in favor of DAA at 3 months. However, whether this difference is clinically meaningful depends on the minimum clinically important difference (MCID) used for postoperative pain. In many settings, an MCID of approximately 1 point on a 0–10 pain scale is commonly used as a practical benchmark, although this threshold can vary by population, baseline pain severity, and recovery context. Using that benchmark, the observed pooled improvement – while statistically robust – would generally be interpreted as a small clinical benefit, because the pooled effect typically does not exceed a 1-point MCID. Overall, the pattern supports a modest early pain advantage for DAA rather than a large reduction in pain.
Secondary pooled outcomes (all outcomes/timepoints)
For completeness, secondary outcomes were pooled whenever two or more studies reported extractable and comparable data. Given anticipated clinical and methodological heterogeneity across studies (differences in surgical comparators, patient case-mix, and perioperative pathways), analyses were conducted using random-effects models. Continuous outcomes were pooled using MD because the included studies reported the relevant measures on the same units/scales for each pooled endpoint. At 6 months, pain remained slightly lower with DAA (MD −0.10, 95% CI −0.18 to −0.02; P = 0.016) with no heterogeneity (I2 = 0%), but the magnitude is small and likely below most MCID thresholds. Functional recovery assessed by HHS at 3 months favored DAA (MD + 7.15, 95% CI 5.28 to 9.02; P < 0.001; I2 = 0%), suggesting a potentially clinically relevant early functional advantage, although interpretation should consider the mixed evidence base (RCT plus observational data) and that HHS MCID thresholds vary by context. By 6 months, the functional difference was no longer clear (MD +0.89, 95% CI −1.02 to 2.79; P = 0.360; I² = 0%), indicating that early gains may diminish over time. Perioperative outcomes showed a mixed profile: operative time demonstrated no significant pooled difference (MD + 4.21 min, 95% CI −7.24 to 15.65; P = 0.471) but with substantial heterogeneity (I² = 80.1%), implying strong influence from learning curve, setting, and case complexity (including DDH). Blood loss consistently favored DAA (MD −81.14 mL, 95% CI −92.60 to −69.68; P < 0.001; I² = 0%), whereas length of stay showed no significant pooled difference (MD −0.97 days, 95% CI −3.86 to 1.92; P = 0.510) with marked heterogeneity (I2 = 78.7%), likely reflecting variability in discharge protocols, baseline populations, and inclusion of more complex cohorts; therefore, LOS pooling should be interpreted cautiously (Table 2; Fig. 5).
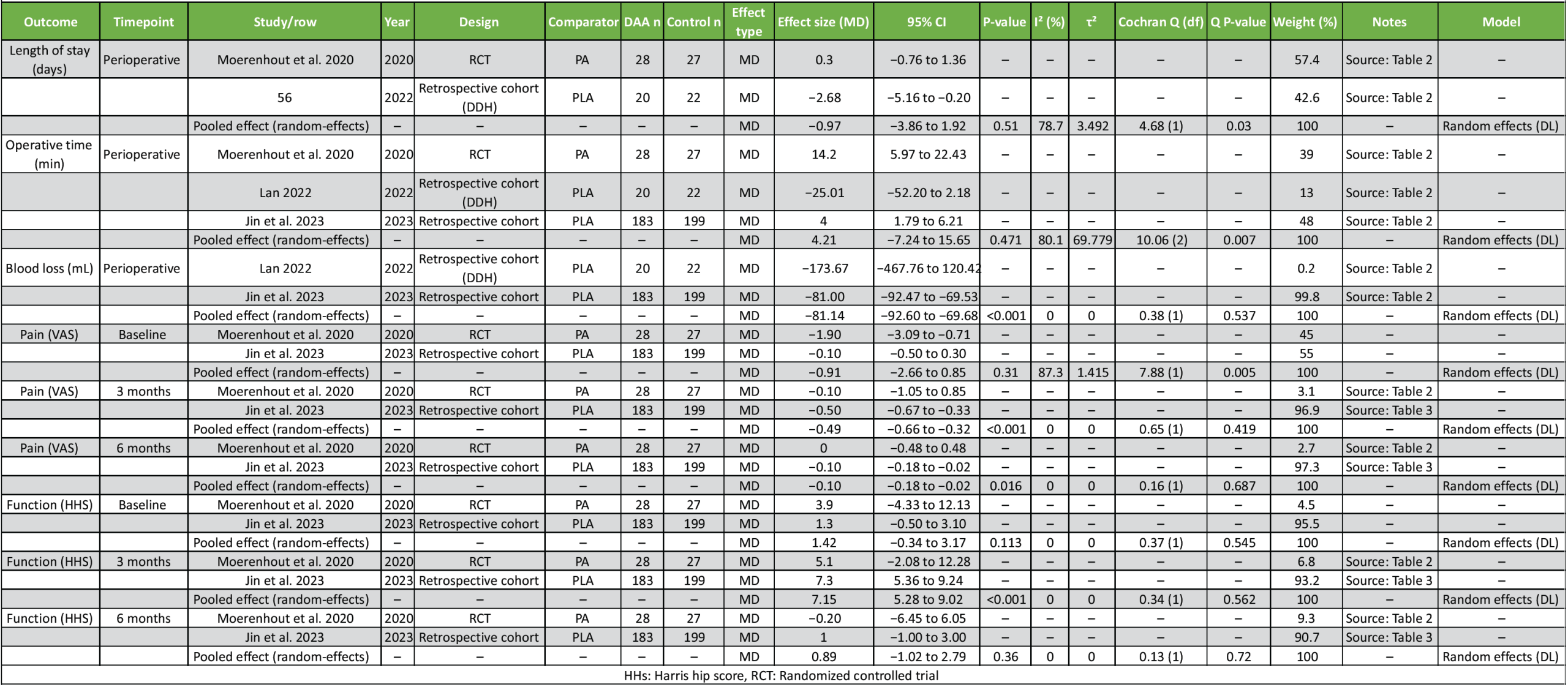
Table 2: Pooled outcomes (all timepoints) with study contributions
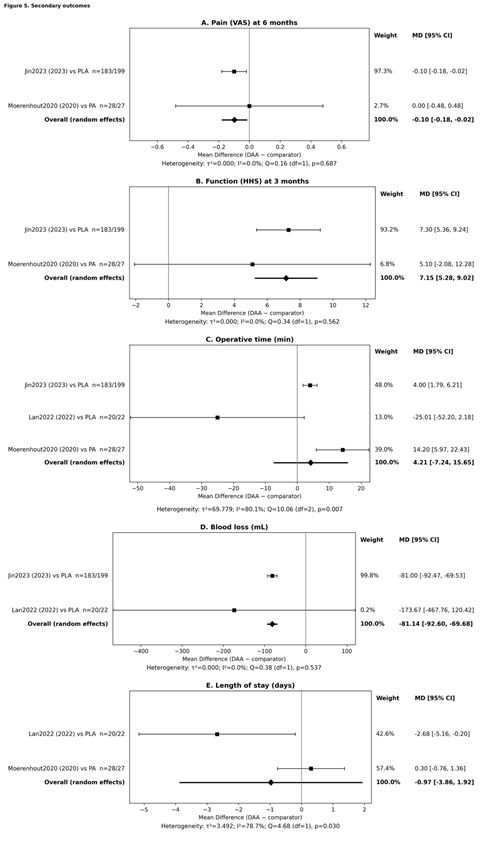
Figure 5: Forest plots of secondary outcomes comparing the direct anterior approach (DAA) versus conventional approaches in primary total hip arthroplasty. Panel A shows pooled effects for pain (Visual Analog Scale) at 6 months; Panel B shows pooled effects for function (Harris hip score, [HHS]) at 3 months; Panel C shows pooled effects for operative time (minutes); Panel D shows pooled effects for estimated blood loss (mL); and Panel E shows pooled effects for length of hospital stay (days). For each study, the square represents the point estimate and the horizontal line represents the 95% confidence interval; square size is proportional to the inverse-variance weight. The diamond represents the pooled estimate under a random-effects model (DerSimonian-Laird), selected a priori due to expected clinical and methodological heterogeneity (differences in study design and comparator approach). Effect estimates are reported as mean differences (MD) (MD; DAA-comparator); values <0 favor DAA for pain, blood loss, and length of stay, whereas values >0 favor DAA for HHS. Heterogeneity statistics (τ2, I2, and Cochran Q with P-value) are reported beneath each panel. Comparators include posterior approach and posterolateral approach as specified within each study label.
Subgroup analyses were pre-specified to explore whether treatment effects differed by (i) study design (RCT vs. observational), (ii) follow-up duration (≤3 months vs. ≥6 months), and (iii) clinical case-mix/comparator approach (e.g., routine primary THA vs. complex anatomy such as DDH; PA vs. PLA). However, most pooled outcomes contained only 2 studies, which makes subgroup meta-analysis statistically unreliable (one study per subgroup) and not interpretable.
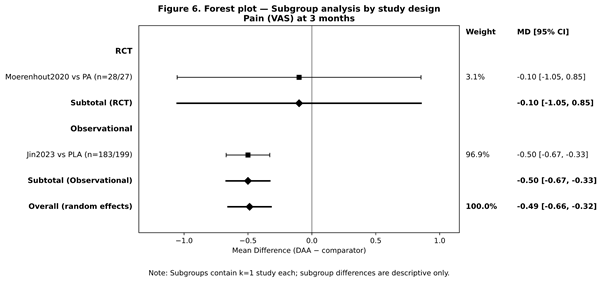
Figure 6: Forest plot – Subgroup analysis.
Study design (RCT vs. observational)
For the primary outcome (VAS at 3 months), the pooled analysis included one RCT (Moerenhout et al., 2020) and one observational cohort (Jin et al., 2023). Because each subgroup would contain k = 1, a formal subgroup comparison was not performed. Directionally, the observational estimate dominated weighting (larger sample size), and both studies favored DAA at 3 months (Fig. 6).
Follow-up duration (≤3 months vs. ≥6 months)
Pain outcomes were available at 3 months and 6 months in two studies. Because the same two studies contributed at each timepoint, time-based subgrouping was unnecessary; instead, outcomes were reported by timepoint (3 vs. 6 months) as separate pooled analyses.
Approach/type/intensity categories (case-mix; comparator type)
Exploratory assessment suggested that case-mix contributed materially to heterogeneity for perioperative outcomes (e.g., operative time and length of stay, where heterogeneity was high). In particular, the DDH cohort contributed effects that differed from routine OA THA populations. However, with k = 3 for operative time and k = 2 for LOS, formal subgroup meta-analysis (e.g., DDH vs non-DDH) was underpowered and therefore not performed.
Not produced due to insufficient studies per subgroup (generally requires ≥2 studies per subgroup and preferably ≥10 overall).
Sensitivity analyses
Sensitivity analyses were performed to assess the robustness of findings to analytical assumptions and the influence of individual studies. Because most pooled outcomes included only two studies (k = 2), a leave-one-out approach was generally not informative (removing one study reduces the analysis to k = 1, which cannot be meta-analyzed). Accordingly, sensitivity checks focused primarily on fixed-effect versus random-effects model comparisons for all pooled outcomes, and a leave-one-out assessment was undertaken only where feasible (operative time; k = 3). For outcomes with low heterogeneity (I2 = 0%), fixed-effect and random-effects results were identical (e.g., pain at 3 and 6 months, HHS at 3 months, and blood loss). In contrast, where heterogeneity was substantial, model choice materially affected inference: For operative time (k = 3) the fixed-effect model suggested significance (MD 4.51 min, 95% CI 2.38 to 6.63; P = 0.00003), whereas the random-effects model was non-significant (MD 4.21 min, 95% CI −7.24 to 15.65; P = 0.471), indicating that under high heterogeneity (I²≈80%) the random-effects model provides a more conservative and appropriate estimate. Similarly, for length of stay (k = 2) the fixed-effect estimate (MD −0.16 days, 95% CI −1.14 to 0.81; P = 0.743) and random-effects estimate (MD −0.97 days, 95% CI −3.86 to 1.92; P = 0.510) both remained non-significant, but the random-effects model reflected greater uncertainty consistent with high heterogeneity (I²≈79%)(Table 3).
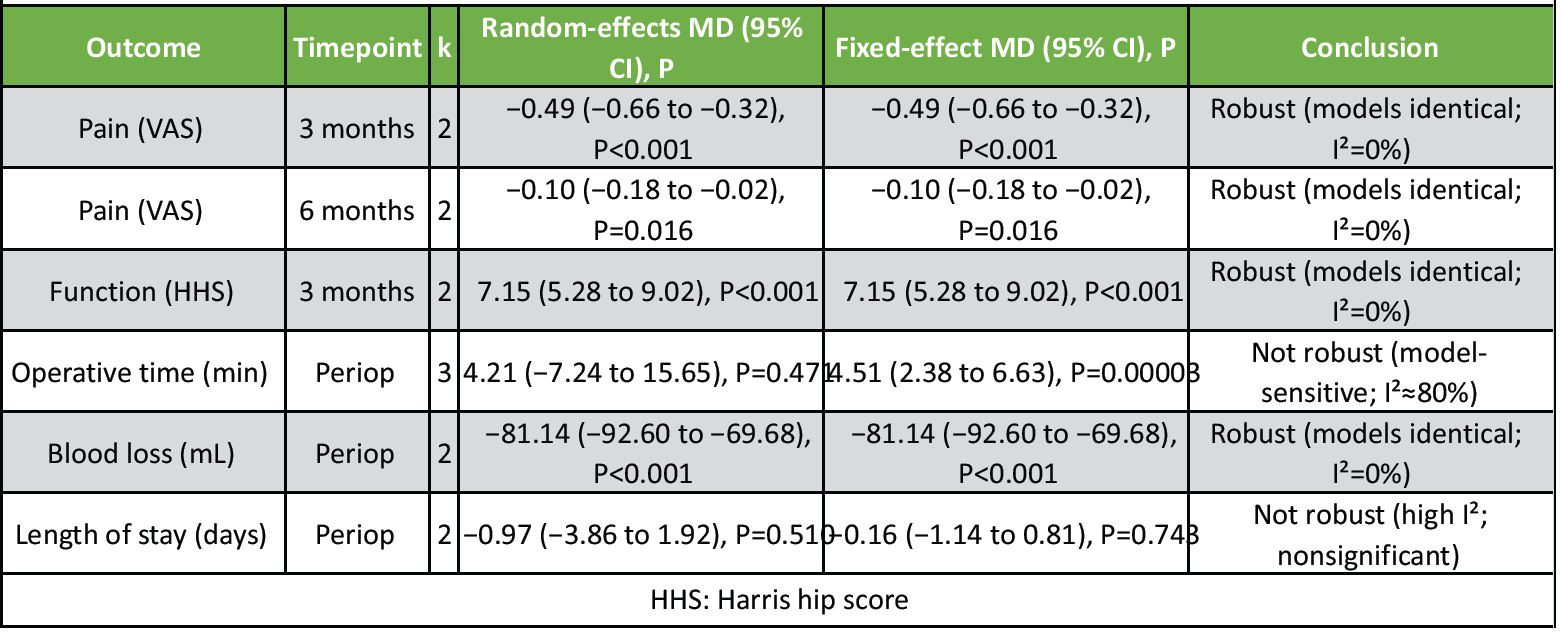
Table 3: Sensitivity analysis summary
Leave-one-out analysis (operative time only; k = 3)
Leave-one-out was feasible only for operative time (k = 3). Removing each study in turn produced widely varying pooled effects with persistent heterogeneity, indicating that the operative time finding is not robust and likely influenced by differences in setting, case-mix, and learning curve effects.
- Remove Moerenhout et al. 2020 → pooled MD −7.21 min, P = 0.610 (k = 2)
- Remove Lan 2022 → pooled MD 30 min, P = 0.099 (k = 2)
- Remove Jin et al. 2023 → pooled MD −3.18 min, P = 0.870 (k = 2).
Publication bias
Publication bias was not formally assessed because fewer than 10 studies contributed to any pooled outcome and most meta-analyses included only 2–3 studies, making funnel plots and asymmetry tests unreliable and potentially misleading.
Certainty of evidence (GRADE)
Certainty of evidence was assessed for key outcomes/timepoints using GRADE domains: Risk of bias, inconsistency, indirectness, imprecision, and publication bias. Because pooled analyses combined RCT and observational evidence, certainty was judged conservatively, and downgrading was applied where appropriate(Table 4).
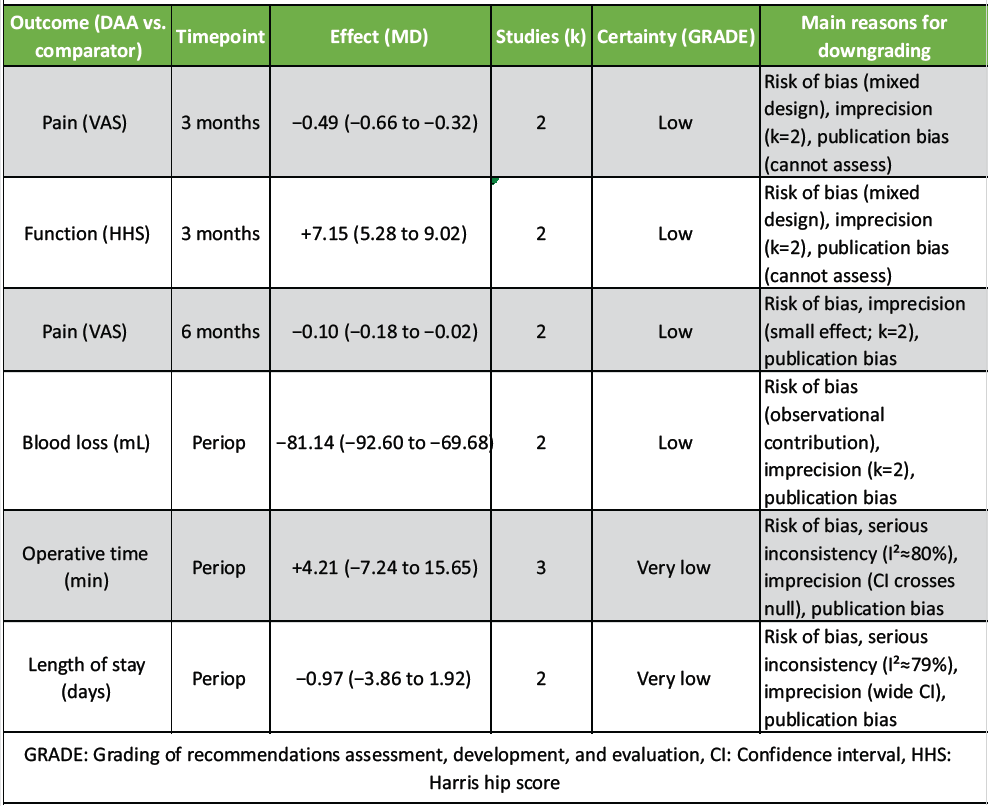
Table 4: GRADE summary of findings (SoF)
Key GRADE judgments (how we rated)
- Risk of bias: Downgraded for most outcomes because the pooled evidence includes observational cohorts with confounding risk and some design limitations.
- Inconsistency: Not downgraded when I² = 0% and effects were directionally consistent (e.g., pain 3 months, blood loss).
- Indirectness: Generally not downgraded (population and intervention relevant to primary THA), but may be considered if specialty cohorts (e.g., DDH) dominate a perioperative endpoint.
- Imprecision: Downgraded when k was very small (k = 2) and/or when CIs were wide or crossed null.
- Publication bias: Downgraded due to small-study evidence base and inability to formally assess bias.
In this updated systematic review and meta-analysis of seven comparative studies of primary THA, the DAA demonstrated statistically significant advantages in early recovery domains, particularly lower pain at 3 months and higher hip function at 3 months, while several perioperative outcomes showed either no clear difference or substantial between-study variability. Specifically, pain at 3 months (VAS) favored DAA (MD −0.49, 95% CI −0.66 to −0.32), with low statistical heterogeneity (I2 = 0%). Function at 3 months (HHS) also favored DAA (MD +7.15, 95% CI 5.28 to 9.02; I2 = 0%). In contrast, operative time and length of stay showed high heterogeneity, suggesting strong influence from setting, case-mix (including complex dysplasia), and surgeon experience/learning curve [18,19].
Our findings are broadly consistent with prior syntheses reporting earlier pain relief and functional recovery with DAA but greater resource requirements (e.g., operative time) and variability across centers [20-22] Network meta-analyses comparing multiple approaches also highlight that early recovery signals can favor DAA, while differences often attenuate by later follow-up and depend on study design and risk of bias [23,24]. In addition, earlier reviews focused on RCT-only evidence have emphasized that effect estimates may shrink when restricting to higher-quality evidence and aligned timepoints [25,26]. These patterns match what we observed: early differences were clearer at 3 months, while later differences (e.g., pain at 6 months) were smaller (MD −0.10) and may not be clinically important [27].
Although the pooled VAS difference at 3 months was statistically robust, the magnitude (~0.5 on a 0–10 scale) likely represents a small clinical benefit if one uses an MCID close to ~1 point for postoperative pain improvement [28-35]. The functional improvement at 3 months (HHS +7.15) appears more substantial and may be clinically meaningful depending on baseline severity and the MCID threshold chosen for HHS (which varies across populations and reporting conventions) [36]. Importantly, functional measurement and reporting heterogeneity are common in THA research; crosswalk work between HOOS/OHS/WOMAC and related instruments underscores why harmonization is essential for future SRMA updates and why instrument choice affects interpretability [37,38,39]. Large multicenter PROM-based cohorts also show that recovery domains captured by HOOS Jr and PROMIS can differ from clinician-based scales, reinforcing the need to prespecify outcomes and timepoints [40,41]. For blood loss, the pooled estimate favored DAA (MD −81 mL, I2 = 0%). While this difference is consistent, its clinical relevance depends on transfusion thresholds, baseline anemia, and perioperative blood management strategies – variables that are often inconsistently reported across observational cohorts. Conversely, operative time demonstrated marked heterogeneity (I2 ≈ 80%), making any single pooled estimate unreliable as a “typical” effect.
This is compatible with the well-described dependence of DAA efficiency on surgeon experience and institutional standardization, and it aligns with prior meta-analyses that reported longer or more variable operative times during adoption phases [42,43]. Length of stay similarly showed substantial heterogeneity, likely reflecting differences in ERAS protocols, discharge criteria, health-system context, and inclusion of complex-case cohorts (e.g., DDH).
Across comparative literature, safety conclusions must distinguish general THA complications (infection, dislocation, fracture, revision) from approach-specific risks (e.g., lateral femoral cutaneous nerve symptoms with DAA). Anatomical evidence clarifies why LFCN neuropraxia can occur with anterior exposure and incision placement, and why reporting the definition and ascertainment method matters when pooling complication outcomes [42,43]. For fracture-related indications (e.g., hemiarthroplasty), systematic reviews suggest that approach effects may differ from elective primary THA due to patient frailty and different surgical goals; such evidence should not be directly pooled with primary THA SRMA but is useful context when discussing generalizability [44,45].
Radiographic component positioning and “precision” outcomes are increasingly evaluated using technology-assisted workflows. Matched-pair analyses in robot-assisted THA suggest that acetabular component position accuracy/precision may not differ meaningfully between anterior and posterior approaches when robotics standardizes execution, implying that any approach-related positioning advantage may narrow in modern practice [46]. Similarly, radiographic cohorts examining difficult morphology (e.g., coxa profunda/protrusio) emphasize that patient anatomy can dominate alignment targets and complication patterns, potentially confounding comparisons if case-mix differs by approach [47]. Imaging advances – including weight-bearing cone beam CT – may further refine how future studies evaluate function-related alignment and biomechanics, but these methods also introduce new comparability challenges for SRMA unless standardized.
The certainty of conclusions is constrained by the evidence structure. Only one included study was an RCT, judged overall as “some concerns,” and most observational studies had at least moderate risk related to confounding and selection into approach groups. This is consistent with core principles of evidence appraisal: Comparative surgical studies are particularly vulnerable to performance bias, learning-curve effects, and selective case allocation [25]. Our approach to bias appraisal aligns with contemporary standards – RoB 2 for randomized trials [24] and ROBINS-I-aligned reasoning for observational studies – yet residual confounding remains difficult to eliminate without well-powered pragmatic RCTs and registry-embedded designs [24]. Publication bias assessment is also limited in small evidence sets; with few studies per pooled endpoint, asymmetry tests are underpowered and may mislead, a limitation emphasized in the meta-research literature [26].
Clinically, the current evidence supports DAA as a reasonable option for primary THA where surgeon experience and pathways are mature, with modest early advantages in pain and function that may diminish over time [48]. Future trials should (1) standardize outcome sets (pain, HOOS Jr/PROMIS, HHS) and timepoints, (2) report learning-curve metrics and surgeon volume, (3) ensure transparent complication definitions (including nerve symptoms), and (4) incorporate contemporary technology contexts (robotic assistance, imaging protocols) [49,50]. Complementing quantitative outcomes with structured qualitative syntheses of patient experience may also clarify which early differences matter most to patients and clinicians [23].
Overall, DAA appears to offer small-to-moderate early recovery benefits with variable perioperative efficiency effects that are highly context-dependent, and the certainty of evidence remains limited by confounding and small numbers of studies contributing to each pooled endpoint.
In this systematic review and meta-analysis of seven comparative studies evaluating the DAA versus conventional approaches in primary THA, DAA was associated with better early recovery, demonstrated by lower pain and higher functional scores at early follow-up (particularly around 3 months). Perioperative findings were mixed: DAA showed lower estimated blood loss in pooled analyses, whereas operative time and length of stay were highly variable across studies and settings, with substantial heterogeneity suggesting strong influence from case-mix, perioperative pathways, and surgeon experience. Complication reporting was inconsistent and limited pooled safety inference; however, interpretation should account for known approach-specific risks and differences in definitions across studies.
Overall, DAA may provide modest early clinical benefits when performed in appropriate patients by experienced teams, but the certainty of evidence remains limited due to the small number of poolable studies, mixed study designs, and residual confounding. Future well-designed pragmatic randomized trials and standardized prospective cohorts using harmonized outcome measures and timepoints are needed to define which patients derive the greatest benefit and to clarify comparative safety and resource use.
Direct anterior total hip arthroplasty may provide modest early recovery benefits compared with conventional approaches, showing slightly lower pain and better early hip function with reduced blood loss; however, operative time and length of stay remain variable and are influenced by surgeon experience and case complexity
References
- 1. Hunter DJ, March L, Chew M. Osteoarthritis in 2020 and beyond: A Lancet Commission. Lancet 2020;396:1711-2. [Google Scholar] [PubMed]
- 2. Safiri S, Kolahi AA, Smith E, Hill C, Bettampadi D, Mansournia MA, et al. Global, regional and national burden of osteoarthritis 1990-2017: A systematic analysis of the Global Burden of Disease Study 2017. Ann Rheum Dis 2020;79:819-28. [Google Scholar] [PubMed]
- 3. Safiri S, Kolahi AA, Cross M, Hill C, Smith E, Carson-Chahhoud K, et al. Prevalence, deaths, and disability-adjusted life years due to musculoskeletal disorders for 195 countries and territories 1990-2017. Arthritis Rheumatol 2021;73:702-14. [Google Scholar] [PubMed]
- 4. Fu M, Zhou H, Li Y, Jin H, Liu X. Global, regional, and national burdens of hip osteoarthritis from 1990 to 2019: Estimates from the 2019 global burden of disease study. Arthritis Res Ther 2022;24:8. [Google Scholar] [PubMed]
- 5. Günther KP, Deckert S, Lützner C, Lange T, Schmitt J, Postler A, et al. Total hip replacement for osteoarthritis-evidence-based and patient-oriented indications. Dtsch Arztebl Int 2021;118:730-6. [Google Scholar] [PubMed]
- 6. Liu Z, Bell CD, Ong AC, Zhang J, Li J, Zhang Y. Clinical evaluation of direct anterior approach total hip arthroplasty for severe developmental dysplasia of the hip. Sci Rep 2021;11:8105. [Google Scholar] [PubMed]
- 7. Bannuru RR, Osani MC, Vaysbrot EE, Arden NK, Bennell K, Bierma-Zeinstra SM, et al. OARSI guidelines for the non-surgical management of knee, hip, and polyarticular osteoarthritis. Osteoarthritis Cartilage 2019;27:1578-89. [Google Scholar] [PubMed]
- 8. Sodhi N, Piuzzi NS, Dalton SE, George J, Ng M, Khlopas A, et al. What influence does the time of year have on postoperative complications following total knee arthroplasty? J Arthroplasty 2018;33:1908-13. [Google Scholar] [PubMed]
- 9. Evans JT, Evans JP, Walker RW, Blom AW, Whitehouse MR, Sayers A. How long does a hip replacement last? A systematic review and meta-analysis of case series and national registry reports with more than 15 years of follow-up. Lancet 2019;393:647-54. [Google Scholar] [PubMed]
- 10. Freistühler M, Roeder N, Franz D. Institut für qualitätssicherung und transparenz im gesundheitswesen. Z Für Herz Thorax Und Gefäßchirurgie 2018;32:331-8. [Google Scholar] [PubMed]
- 11. Winther SB, Foss OA, Husby OS, Wik TS, Klaksvik J, Husby VS. Muscular strength and function after total hip arthroplasty performed with three different surgical approaches: One-year follow-up study. Hip Int 2019;29:405-11. [Google Scholar] [PubMed]
- 12. Davis T, Loudermilk E, DePalma M, Hunter C, Lindley D, Patel N, et al. Prospective, multicenter, randomized, crossover clinical trial comparing the safety and effectiveness of cooled radiofrequency ablation with corticosteroid injection in the management of knee pain from osteoarthritis. Reg Anesth Pain Med 2018;43:84-91. [Google Scholar] [PubMed]
- 13. Taylor-Williams O, Nossent J, Inderjeeth CA. Incidence and complication rates for total hip arthroplasty in rheumatoid arthritis: A systematic review and meta-analysis across four decades. Rheumatol Ther 2020;7:685-702. [Google Scholar] [PubMed]
- 14. Hueter C. Fünfte abtheilung: Die verletzung und krankheiten des hüftgelenkes, neunundzwanzigstes capitel. Grundriss Der Chir 1883;2:e200. [Google Scholar] [PubMed]
- 15. Wang Z, Hou JZ, Wu CH, Zhou YJ, Gu XM, Wang HH, et al. A systematic review and meta-analysis of direct anterior approach versus posterior approach in total hip arthroplasty. J Orthop Surg Res 2018;13:229. [Google Scholar] [PubMed]
- 16. Talia AJ, Coetzee C, Tirosh O, Tran P. Comparison of outcome measures and complication rates following three different approaches for primary total hip arthroplasty: A pragmatic randomised controlled trial. Trials 2018;19:13. [Google Scholar] [PubMed]
- 17. Dimitriou D, Helmy N, Hasler J, Flury A, Finsterwald M, Antoniadis A. The role of total hip arthroplasty through the direct anterior approach in femoral neck fracture and factors affecting the outcome. J Arthroplasty 2019;34:82-7. [Google Scholar] [PubMed]
- 18. Jia F, Guo B, Xu F, Hou Y, Tang X, Huang L. A comparison of clinical, radiographic and surgical outcomes of total hip arthroplasty between direct anterior and posterior approaches: A systematic review and meta-analysis. Hip Int 2019;29:584-96. [Google Scholar] [PubMed]
- 19. Kucukdurmaz F, Sukeik M, Parvizi J. A meta-analysis comparing the direct anterior with other approaches in primary total hip arthroplasty. Surgeon 2019;17:291-9. [Google Scholar] [PubMed]
- 20. Lintz F, De Cesar Netto C, Barg A, Burssens A, Richter M, Weight Bearing CT International Study Group. Weight-bearing cone beam CT scans in the foot and ankle. EFORT Open Rev 2018;3:278-86. [Google Scholar] [PubMed]
- 21. Putananon C, Tuchinda H, Arirachakaran A, Wongsak S, Narinsorasak T, Kongtharvonskul J. Comparison of direct anterior, lateral, posterior and posterior-2 approaches in total hip arthroplasty: Network meta-analysis. Eur J Orthop Surg Traumatol 2018;28:255-67. [Google Scholar] [PubMed]
- 22. Miller LE, Gondusky JS, Bhattacharyya S, Kamath AF, Boettner F, Wright J. Does surgical approach affect outcomes in total hip arthroplasty through 90 days of follow-up?A systematic review with meta-analysis. J Arthroplasty 2018;33:1296-302. [Google Scholar] [PubMed]
- 23. Esquivel CJ. The experience of health professional students and their educators learning to work in intra-professional teams: A joanna briggs institute (jbi) qualitative systematic review. (In Press]. [Google Scholar] [PubMed]
- 24. Sterne JA, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019;366:l4898. [Google Scholar] [PubMed]
- 25. Burns PB, Rohrich RJ, Chung KC. The levels of evidence and their role in evidence-based medicine. Plast Reconstr Surg 2011;128:305-10. [Google Scholar] [PubMed]
- 26. Kicinski M, Springate DA, Kontopantelis E. Publication bias in meta-analyses from the cochrane database of systematic reviews. Stat Med 2015;34:2781-93. [Google Scholar] [PubMed]
- 27. Moerenhout K, Derome P, Laflamme GY, Leduc S, Gaspard HS, Benoit B. Direct anterior versus posterior approach for total hip arthroplasty: A multicentre, prospective, randomized clinical trial. Can J Surg 2020;63:E412-7. [Google Scholar] [PubMed]
- 28. Finch DJ, Martin BI, Franklin PD, Magder LS, Pellegrini VD Jr. Pepper Investigators. Patient-reported outcomes following total hip arthroplasty: A multicenter comparison based on surgical approaches. J Arthroplasty 2020;35:1029-35. [Google Scholar] [PubMed]
- 29. Yang XT, Huang HF, Sun L, Yang Z, Deng CY, Tian XB. Direct anterior approach versus posterolateral approach in total hip arthroplasty: A systematic review and meta‐analysis of randomized controlled studies. Orthop Surg 2020;12:1065-73. [Google Scholar] [PubMed]
- 30. Thaler M, Dammerer D, Hechenberger F, Hörmann R, Van Beeck A, Stofferin H. The anatomical course of the lateral femoral cutaneous nerve in relation to various skin incisions used for primary and revision total hip arthroplasty with the direct anterior approach. J Arthroplasty 2021;36:368-73. [Google Scholar] [PubMed]
- 31. Peng L, Zeng Y, Wu Y, Zeng J, Liu Y, Shen B. Clinical, functional and radiographic outcomes of primary total hip arthroplasty between direct anterior approach and posterior approach: A systematic review and meta-analysis. BMC Musculoskelet Disord 2020;21:338. [Google Scholar] [PubMed]
- 32. Chen W, Sun JN, Zhang Y, Zhang Y, Chen XY, Feng S. Direct anterior versus posterolateral approaches for clinical outcomes after total hip arthroplasty: A systematic review and meta-analysis. J Orthop Surg Res 2020;15:231. [Google Scholar] [PubMed]
- 33. Sun X, Zhao X, Zhou L, Su Z. Direct anterior approach versus posterolateral approach in total hip arthroplasty: A meta-analysis of results on early post-operative period. J Orthop Surg Res 2021;16:69. [Google Scholar] [PubMed]
- 34. Huang XT, Liu DG, Jia B, Xu YX. Comparisons between direct anterior approach and lateral approach for primary total hip arthroplasty in postoperative orthopaedic complications: A systematic review and meta-analysis. Orthop Surg 2021;13:1707-20. [Google Scholar] [PubMed]
- 35. Yan L, Ge L, Dong S, Saluja K, Li D, Reddy KS, et al. Evaluation of comparative efficacy and safety of surgical approaches for total hip arthroplasty. JAMA Netw Open 2023;6:e2253942. [Google Scholar] [PubMed]
- 36. Ramadanov N, Voss M, Hable R, Hakam HT, Prill R, Salzmann M, et al. Postoperative harris hip score versus harris hip score difference in hip replacement: What to report? Orthop Surg 2025;17:3-21. [Google Scholar] [PubMed]
- 37. Putman S, Preda C, Girard J, Duhamel A, Migaud H. Mapping and crosswalk of the oxford hip score and different versions of the hip disability and osteoarthritis outcome score. Clin Orthop Relat Res 2021;479:1534-44. [Google Scholar] [PubMed]
- 38. Lazaru P, Bueschges S, Ramadanov N. Direct anterior approach (DAA) vs. conventional approaches in total hip arthroplasty: A RCT meta-analysis with an overview of related meta-analyses. PLoS One 2021;16:e0255888. [Google Scholar] [PubMed]
- 39. Fleisher IT, Thompson MC, Mensah CJ, Joseph AD, McLawhorn AS, Padgett DE, et al. Development and validation of crosswalks between the western Ontario & mcmaster universities osteoarthritis index and hip disability and osteoarthritis outcome score joint replacement/knee injury and osteoarthritis outcome score joint replacement. J Arthroplasty 2022;37:1034-9.e3. [Google Scholar] [PubMed]
- 40. Higgins BT, Barlow DR, Heagerty NE, Lin TJ. Anterior vs. posterior approach for total hip arthroplasty, a systematic review and meta-analysis. J Arthroplasty 2015;30:419-34. [Google Scholar] [PubMed]
- 41. Yue C, Kang P, Pei F. Comparison of direct anterior and lateral approaches in total hip arthroplasty: A systematic review and meta-analysis (PRISMA). Medicine (Baltimore) 2015;94:e2126. [Google Scholar] [PubMed]
- 42. Ladurner A, Schöfl T, Calek AK, Zdravkovic V, Giesinger K. Direct anterior approach improves in-hospital mobility following hemiarthroplasty for femoral neck fracture treatment. Arch Orthop Trauma Surg 2022;142:3183-92. [Google Scholar] [PubMed]
- 43. Jin MW, Zhang L, Chu XB, Lv SJ, Zhang JJ, Tong PJ, et al. Comparison of clinical efficacy between direct anterior approach and posterolateral approach in primary total hip arthroplasty. Eur Rev Med Pharmacol Sci 2023;27:5604-13. [Google Scholar] [PubMed]
- 44. Awad ME, Farley BJ, Mostafa G, Saleh KJ. Direct anterior approach has short-term functional benefit and higher resource requirements compared with the posterior approach in primary total hip arthroplasty: A meta-analysis of functional outcomes and cost. Bone Joint J 2021;103-B:1078-87. [Google Scholar] [PubMed]
- 45. Khan IA, Magnuson JA, Arshi A, Krueger CA, Freedman KB, Fillingham YA. Direct anterior approach in hip hemiarthroplasty for femoral neck fractures: Do short-term outcomes differ with approach? A systematic review and meta-analysis. JBJS Rev 2022;10(9):e21.00202. [Google Scholar] [PubMed]
- 46. Gazendam A, Bozzo A, Ekhtiari S, Kruse C, Hiasat N, Tushinski D, et al. Short-term outcomes vary by surgical approach in total hip arthroplasty: A network meta-analysis. Arch Orthop Trauma Surg 2022;142:2893-902. [Google Scholar] [PubMed]
- 47. Latijnhouwers DA, Niels LA, Verdegaal SH, Nelissen RG, Vlieland TP, Kaptijn HH, Gademan MG. Activities and participation after primary total hip arthroplasty; posterolateral versus direct anterior approach in 860 patients. Acta orthopaedica 2022;93:613-22. [Google Scholar] [PubMed]
- 48. Zhou Z, Li Y, Peng Y, Jiang J, Zuo J. Clinical efficacy of direct anterior approach vs. other surgical approaches for total hip arthroplasty: A systematic review and meta-analysis based on RCTs. Front Surg 2022;9:1022937. [Google Scholar] [PubMed]
- 49. Kunze KN, Huddleston HP, Romero J, Chiu YF, Jerabek SA, McLawhorn AS. Accuracy and precision of acetabular component position does not differ between the anterior and posterior approaches to total hip arthroplasty with robotic assistance: A matched-pair analysis. Arthroplasty Today 2022;18:68-75. [Google Scholar] [PubMed]
- 50. Heinz T, Vasilev H, Anderson PM, Stratos I, Jakuscheit A, Horas K, et al. The direct anterior approach (daa) as a standard approach for total hip arthroplasty (tha) in coxa profunda and protrusio acetabuli? A radiographic analysis of 188 cases. J Clin Med 2023;12:3941. [Google Scholar] [PubMed]