Snake envenomation of the upper extremity may result in significant soft-tissue sequelae and high operative burden.
Dr. Mark Solarz, Department of Orthopaedics and Sports Medicine, Temple University Hospital, Philadelphia, Pennsylvania. E-mail: mark.solarz@tuhs.temple.edu; Phone: 215-707-2111
Introduction: Snake envenomation is exceedingly rare in the United States. Nonetheless, herein we describe the case of a 26-year-old male who presented to our urban emergency department (ED) with monocled cobra (Naja kaouthia) bites to his bilateral upper extremities, subsequent monitoring for the development of compartment syndrome, and eventual treatment of a subacute wound infection.
Case Report: The patient presented almost immediately after sustaining bites to his bilateral hands while descaling his pet cobra. Approximately 4 h later, he developed signs of neurotoxicity, which improved following the administration of antivenom. Orthopedic surgery was consulted to evaluate for possible compartment syndrome of the right upper extremity, but fortunately, he did not develop concerning symptoms on serial examinations. Six days later, the patient returned to the ED with purulent drainage from the right volar palm wound. The patient was admitted and started on intravenous antibiotics. However, due to persistent purulent drainage, he underwent two operative procedures for drainage of a palmar abscess. The patient’s post-operative course was uncomplicated, and he recovered baseline function of his right hand.
Conclusion: Snake envenomation of the upper extremity can result in significant soft-tissue sequelae. Orthopedic practitioners must be aware of these complications and should also recognize signs and symptoms of acute intoxication when managing these patients.
Keywords: Snake envenomation, compartment syndrome, soft-tissue abscess, hand infection.
Although relatively infrequent, approximately 8,000–10,000 patients are treated for snake bites at emergency departments (EDs) across the United States every year [1]. A majority of these injuries are inflicted by non-venomous species, but a small fraction of injuries from venomous species result in envenomation, significantly altering the treatment algorithm [2,3]. Members of the Elapidae and Viperidae families are predominantly responsible for snake envenomation in the United States, with most occurring in private residences, presumably by individuals who keep these species as pets [2]. Although native to Southeast Asia, envenomation by Naja spp., commonly known as cobras, and specifically Naja kaouthia (monocled cobra), has been described in the United States and is regarded as of the highest medical importance according to the World Health Organization’s classification of venomous snakes due to their extreme lethality [4,5,6]. N. kaouthia presents a unique hazard compared with other cobra species as it is also capable of spitting its venom, which can affect the eyes [7]. Due to the rapid-onset neurotoxic paralysis that often ensues following N. kaouthia envenomation, antivenom is the first-line definitive treatment with other supportive interventions primarily aimed at preventing respiratory failure [3,5,8].
In addition to this severe neuromuscular blockade, snake envenomation can also result in morbid sequelae such as compartment syndrome and infection at the site of the bite. While the direct effects of envenomation can often mimic signs of compartment syndrome despite normal soft-tissue pressures, several instances of true compartment syndrome following snake envenomation have been described, often requiring treatment with fasciotomy [3,9,10,11]. Nonetheless, wound infection following a snake bite is much more common, occurring in roughly one out of every five cases in the Americas [12,13]. Infection rates for Elapidae species specifically exceed 60% and may require surgical intervention or long-term antibiotic therapy [12,14]. The rarity of Elapidae envenomation in the United States, coupled with the severity of the injury, may present a challenge to clinicians who encounter such a situation in the ED. Herein, we describe the case report of a 26-year-old male who presented with monocled cobra (N. kaouthia) bites to his bilateral upper extremities and subsequently underwent evaluation for compartment syndrome and developed a subacute wound infection following initial envenomation treatment.
A 26-year-old male presented to the ED almost immediately after sustaining bites to his bilateral hands while descaling his pet snake. He described venom coming out of the wounds located on the volar aspect of the right palm proximal to the index finger palmodigital crease. His right hand was diffusely swollen to the wrist and moderately tender to palpation (Fig. 1).
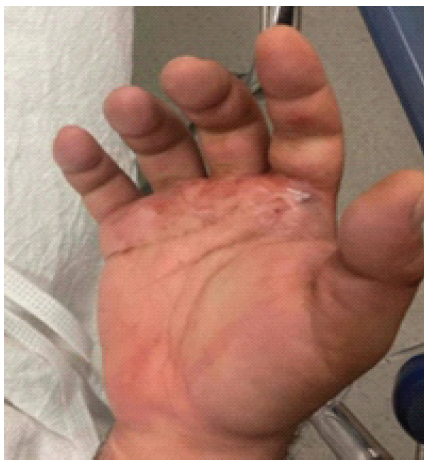
Figure 1: Right volar palm wound at initial presentation to the emergency department.
Multiple punctate wounds were present on the left hand, and motion at the digits and wrist was significantly limited secondary to pain. Sensation in the median and ulnar nerve distributions was intact bilaterally, and all digits were well perfused. Vital signs were stable, and no laboratory abnormalities were present at the time of initial workup. Approximately 90 min following arrival at the ED, the patient became diaphoretic, tachypneic, and bradycardic, which was attributed to a vasovagal response. He recovered quickly without intervention.
Approximately 4 h following initial presentation, the patient developed bilateral ptosis and became lethargic with slurred speech. His vital signs remained stable, so the decision to proceed with mechanical ventilation was delayed until antivenom arrived. As N. kaouthia is not housed in any local zoos, antivenom was urgently flown from a regional metropolitan zoo to our facility. Approximately 5 h following the initial envenomation, 10 vials of antivenom were administered, and the patient’s ptosis and speech improved. At this point, the orthopedic hand team was engaged to evaluate the patient for possible compartment syndrome of the right upper extremity. The patient was comfortable without any clinical signs of compartment syndrome at the time of evaluation and on subsequent serial examinations. Tissue compartments were soft and compressible despite diffuse swelling of the distal extremity. Pain with passive stretch of the joints and paraesthesias were absent. The digits remained well perfused with pulse oximetry in the high 90th percentiles throughout his intensive care unit stay. The patient was admitted for inpatient observation and remained stable throughout the rest of his course. D-dimer (1098 ng/mL) and fibrinogen (330 mg/dL) became mildly elevated 6 h following the administration of antivenom, but no signs of coagulopathy were present. The patient was discharged 3 days after initial admission and was counseled regarding return precautions (Fig. 2). He was not provided with antibiotic prophylaxis at that time.
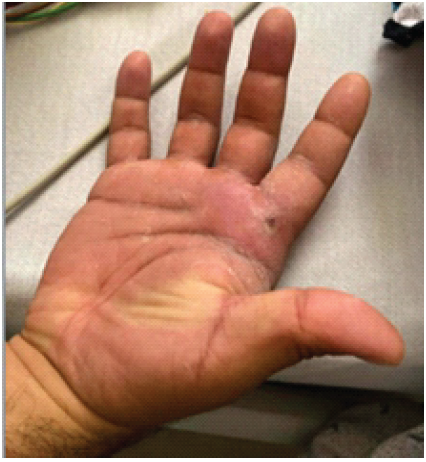
Figure 2: Right volar palm wound on the day of discharge.
Three days following discharge, the patient again presented to the ED with about 24 h of pain and purulent drainage of the right volar palm (Fig. 3).
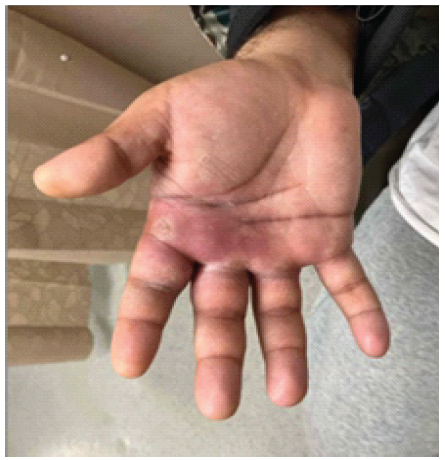
Figure 3: Right volar palm abscess at the second emergency department presentation
The patient was afebrile with mildly elevated neutrophils, elevated erythrocyte sedimentation rate (40 mm/h), and reduced hemoglobin (13.4 g/dL). A bedside irrigation and debridement (I&D) of an abscess at the volar palm overlying the second metacarpal was performed, and cultures grew Morganella morganii. The patient was admitted to the inpatient ward and started on ceftriaxone. Computed tomography with contrast of the right hand was obtained, which at that time showed no obvious fluid collection or abscess formation. However, the patient continued to experience pain and swelling of the right hand with persistent purulent drainage. The patient was taken to the operating room for a formal incision and drainage of the right volar hand on the 2nd day of admission (Fig. 4).
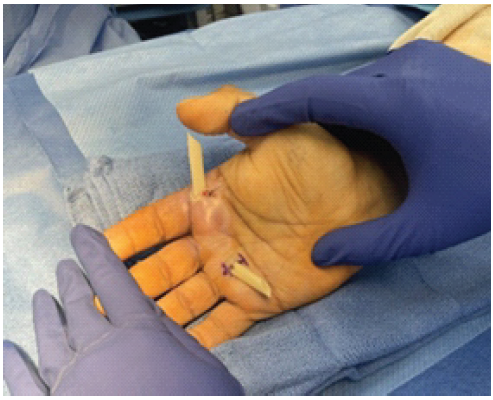
Figure 4: Right volar palm immediately following operative incision and drainage.
Due to continued drainage with dressing changes on post-operative day 2, he required a return to the operating room for a repeat I&D. Cultures from both surgical drainages grew ampicillin-sensitive Enterococcus faecalis and beta-lactamase-positive Bacteroides fragilis. Ceftriaxone therapy was stopped following receipt of these cultures and was replaced with amoxicillin-clavulanate and levofloxacin.
The patient left the hospital through patient-directed discharge the same day of his second operative irrigation and drainage. Incisions remained clean and dry at the outpatient visit on post-operative day 1. Minimal erythema and moderate swelling of the right volar palm were noted with negligible serosanguinous fluid expressed from the wound. Subsequent outpatient wound assessments revealed a well-healing incision with progressively decreased pain, swelling, and restoration of full active digital motion. Sutures were removed on post-operative day 12, and the patient was compliant with oral antibiotics for 6 weeks (Fig. 5).

Figure 5: Right volar palm at last follow-up, approximately 5 weeks post-injury.
Snake envenomation of the upper extremity can result in significant soft-tissue sequelae. Substantial swelling, erythema, and pain can mimic the signs of compartment syndrome, and although true compartment syndrome following N. kaouthia envenomation has been described, our patient’s clinical presentation was not consistent with this complication [3,15]. Nonetheless, this patient developed an abscess of the bite wound that required two operative interventions and a 6-week course of oral antibiotic therapy. Gram-negative species, particularly M. morganii, are the most commonly isolated bacterial species following snake bite infections, with a prevalence of 63.0% [12]. In contrast, Gram-positive and anaerobic species can be identified in 40.0% and 4.0% of cases, respectively [12]. Interestingly, microorganisms from all three categories were isolated from this patient’s operative cultures. This can complicate the selection of an appropriate antibiotic regimen, as a single agent alone is unlikely to provide sufficient coverage of a polymicrobial infection. In this instance, amoxicillin–clavulanate was selected for its activity against B. fragilis and E. faecalis, whereas M. morganii is often susceptible to fluoroquinolones such as levofloxacin [16,17].
Surgical intervention following snake bite infection is a common complication of these injuries, but it is not without its own hazards [12,13]. Heightened vigilance is required when the abscess affects the palm, as superficial neurovascular structures are at risk of injury during the procedure. High-pressure lavage can cause soft-tissue injury and wound healing complications, which may be further exacerbated by local necrosis caused by venom exposure [3,18,19]. With these in mind, a clear discussion of the risks and benefits of abscess drainage is warranted for these patients.
This patient’s post-envenomation course was further complicated by post-synaptic blockade of nicotinic acetylcholine receptors, resulting in a brief episode of ptosis and lethargy [8]. The signs of N. kaouthia neurotoxins are first manifested by disruption to the cranial nerves, resulting in ptosis, ophthalmoplegia, and slurred speech [20]. It is essential that all treating providers be aware of these signs so that appropriate intervention can occur before neuromuscular blockade worsens. Severe envenomation or delay of antivenom administration can result in more widespread flaccid paralysis that affects the diaphragm, leading to respiratory collapse [8,20]. In addition, N. kaouthia are capable of spitting their venom into the eyes, which can present with intense pain, vision changes, corneal edema, and scleral hemorrhage [7].
We describe the case of a 26-year-old male who presented to our ED with N. kaouthia bites to his bilateral hands. A consultation to evaluate for possible compartment syndrome was requested due to diffuse swelling and pain of the right hand, although this patient fortunately did not develop compartment syndrome. Following a brief episode of cholinergic blockade that improved with antivenom administration, the patient developed a polymicrobial abscess at one of the bite wounds, which required two operative procedures for drainage.
While rare in developed countries, snake envenomations can occur, which often result in severe soft-tissue complications. The appropriate management of these injuries requires a multidisciplinary approach that addresses the initial systemic toxicology, acute evaluation for compartment syndrome, and close monitoring for delayed abscess formation or deep infections that may arise.
References
- 1. O’Neil ME, Mack KA, Gilchrist J, Wozniak EJ. Snakebite injuries treated in United States emergency departments, 2001-2004. Wilderness Environ Med 2007;18:281-7. [Google Scholar] [PubMed]
- 2. Warrick BJ, Boyer LV, Seifert SA. Non-native (exotic) snake envenomations in the U.S., 2005-2011. Toxins (Basel) 2014;6:2899-911. [Google Scholar] [PubMed]
- 3. Seifert SA, Armitage JO, Sanchez EE. Snake envenomation. N Engl J Med 2022;386:68-78. [Google Scholar] [PubMed]
- 4. Chowdhury MA, Müller J, Al Haidar IK, Rahman MM, Noman M, Ghose A, et al. Interspecific and intraspecific variability in venom composition of Naja naja and Naja kaouthia (Reptilia: Elapidae) populations from different habitats in Bangladesh. J Proteomics 2025;322:105544. [Google Scholar] [PubMed]
- 5. Greene SC, Osborn L, Bower R, Harding SA, Takenaka K. Monocled cobra (Naja kaouthia) envenomations requiring mechanical ventilation. J Emerg Med 2021;60:197-201. [Google Scholar] [PubMed]
- 6. Warrell DA, World Health Organization, Regional Office for South-East Asia. Guidelines for the Management of Snakebites. 2nd ed. New Delhi: World Health Organization, Regional Office for South-East Asia; 2010. [Google Scholar] [PubMed]
- 7. Rahman MM, Uddin MA, Sayeed AA, Noman M, Auawal A, Islam MR, et al. Venom-spit ophthalmia: A novel envenomation of monocled cobra (Naja kaouthia). Toxicon 2025;254:108221. [Google Scholar] [PubMed]
- 8. Kakati H, Patra A, Mukherjee AK. Composition, pharmacology, and pathophysiology of the venom of monocled cobra (Naja kaouthia)- a medically crucial venomous snake of southeast Asia: An updated review. Toxicon 2024;249:108056. [Google Scholar] [PubMed]
- 9. Bucaretchi F, De Capitani EM, Hyslop S, Mello SM, Fernandes CB, Bergo F, et al. Compartment syndrome after South American rattlesnake (Crotalus durissus terrificus) envenomation. Clin Toxicol (Phila) 2014;52:639-41. [Google Scholar] [PubMed]
- 10. Bucaretchi F, De Capitani EM, Hyslop S, Mello SM, Madureira PR, Zanardi V, et al. Compartment syndrome after Bothrops jararaca snakebite: Monitoring, treatment, and outcome. Clin Toxicol (Phila) 2010;48:57-60. [Google Scholar] [PubMed]
- 11. Varney SM, Alindogan AA, Stuteville H, Roth BA, Watkins S, Ng PC, et al. Fasciotomy following North American pit viper envenomation in Texas 2004-2021. Clin Toxicol (Phila) 2024;62:314-21. [Google Scholar] [PubMed]
- 12. Bonilla-Aldana DK, Bonilla-Aldana JL, Ulloque-Badaracco JR, Al-Kassab-Córdova A, Hernandez-Bustamante EA, Alarcón-Braga EA, et al. Snakebite-associated infections: A systematic review and meta-analysis. Am J Trop Med Hyg 2024;110:874-86. [Google Scholar] [PubMed]
- 13. Yeh H, Gao SY, Lin CC. Wound infections from Taiwan cobra (Naja atra) bites: Determining bacteriology, antibiotic susceptibility, and the use of antibiotics-a Cobra BITE study. Toxins (Basel) 2021;13:183. [Google Scholar] [PubMed]
- 14. Brenes-Chacon H, Gutiérrez JM, Avila-Aguero ML. Use of antibiotics following snakebite in the era of antimicrobial stewardship. Toxins (Basel) 2024;16:37. [Google Scholar] [PubMed]
- 15. Tongpoo A, Trakulsrichai S, Putichote K, Sriapha C, Wananukul W. Recurrent neurotoxic envenoming of cobra bite. Toxicon 2019;167:180-3. [Google Scholar] [PubMed]
- 16. Wu PH, Chen CH, Lin HH, Tseng KH, Ko WC, Ho MW, et al. Geographic patterns of antimicrobial susceptibilities for Bacteroides spp. Worldwide: Results from the Antimicrobial Testing Leadership and Surveillance (ATLAS) programme, 2007-2020. Int J Antimicrob Agents 2023;62:106822. [Google Scholar] [PubMed]
- 17. Zhu W, Liu Q, Liu J, Wang Y, Shen H, Wei M, et al. Genomic epidemiology and antimicrobial resistance of Morganella clinical isolates between 2016 and 2023. Front Cell Infect Microbiol 2025;14:1464736. [Google Scholar] [PubMed]
- 18. Chiaramonti A, Robertson A, Nguyen T, Jaffe DE, Hanna EL, Holmes R, et al. Pulsatile lavage of musculoskeletal wounds causes muscle necrosis and dystrophic calcification in a rat model. J Bone Joint Surg Am 2017;99:1851-8. [Google Scholar] [PubMed]
- 19. Zeitlin J, Arango SD, Flynn JC, Hammarstedt JE, Weir TB, Miller AJ. Esmarch exsanguination in acute hand infections: Evaluating risk of infection spread. Surg Infect (Larchmt) 2025;26:688-93. [Google Scholar] [PubMed]
- 20. Tansuwannarat P, Tongpoo A, Phongsawad S, Sriapha C, Wananukul W, Trakulsrichai S. A retrospective cohort study of cobra envenomation: Clinical characteristics, treatments, and outcomes. Toxins (Basel) 2023;15:468. [Google Scholar] [PubMed]