Combined post-operative denosumab and bisphosphonate therapy may represent a biologically rational adjuvant strategy to reduce recurrence risk in high-risk spinal giant cell tumors.
Dr. Sumant Samuel, Department of Orthopaedics, Christian Medical College, Vellore, Tamil Nadu, India. E-mail: sumantsamuel@gmail.com
Introduction: Giant cell tumor (GCT) of the spine is a rare, locally aggressive bone tumor with a high risk of recurrence, particularly when wide surgical margins cannot be achieved. Although en bloc spondylectomy provides the best oncological control, it is frequently not feasible due to anatomical constraints. Systemic adjuvants have therefore been explored to reduce recurrence risk. Denosumab suppresses osteoclast-mediated bone resorption, while bisphosphonates exert direct anti-tumor effects on neoplastic stromal cells. However, the intentional combined post-operative use of denosumab and oral bisphosphonate therapy in spinal GCT (SGCT) has not been previously reported. This case highlights the feasibility and biological rationale of this combined adjuvant strategy.
Case Report: A 27-year-old male presented with severe low back pain and bilateral lower limb radicular pain of 6 months’ duration. Imaging demonstrated a destructive lesion involving the third lumbar vertebral body with paravertebral soft-tissue extension. A diagnosis of Enneking stage III GCT was made after biopsy. The patient received pre-operative denosumab followed by gross total resection of the affected vertebra with posterior stabilization and anterior column reconstruction. Postoperatively, denosumab therapy was continued for 6 months, and the patient was simultaneously commenced on oral bisphosphonate therapy (weekly alendronate followed by monthly ibandronate). The patient had complete resolution of symptoms. Serial imaging demonstrated progressive osseous consolidation and stable instrumentation with no evidence of residual or recurrent disease. At 4½ years of follow-up, the patient remained radiologically disease free without treatment-related adverse events.
Conclusion: This report demonstrates the feasibility of combining post-operative denosumab and oral bisphosphonate therapy as an adjuvant strategy for high-risk SGCT when wide surgical margins are not achievable. By targeting different cellular pathways involved in tumor biology, this approach may provide complementary tumor control, thereby reducing the risk of recurrence.
Keywords: Spinal, giant cell tumor, bisphosphonates, alendronate, ibandronate, denosumab.
Giant cell tumor (GCT) of bone is a benign but locally aggressive neoplasm composed of neoplastic mononuclear stromal cells intermixed with osteoclast-like giant cells [1,2]. Though commonly seen in the epiphyses of long bones, spinal GCT (SGCT) is rarer and presents greater therapeutic challenges because regional anatomy limits the ability to obtain wide margins and increases perioperative morbidity [3,4]. Enneking stage III tumors with soft-tissue extension carry a particularly high risk of local recurrence, which can be difficult to surgically treat [3].
The use of adjuvants has therefore been explored to decrease the incidence of local recurrence. Bisphosphonates (notably zoledronic acid) have been used to suppress osteoclast activity and – based on in vitro and clinical series – may exert direct antitumor effects on the neoplastic stromal cells [2]. Denosumab, a fully human monoclonal antibody targeting receptor activator of nuclear factor kappa B ligand (RANKL), was introduced more recently and has markedly changed the management of aggressive and unresectable GCT [1,4]. However, denosumab does not induce apoptosis of the neoplastic stromal cells, raising concerns about recurrence after withdrawal [1,2]. Because bisphosphonates and denosumab act on different cellular components and molecular pathways, there is a biologically plausible rationale for complementary (or even synergistic) use: Denosumab suppresses osteoclast-mediated osteolysis while bisphosphonates can directly inhibit stromal cell proliferation and induce apoptosis. In SGCT – where recurrence risk and surgical morbidity are high – exploring whether combined biologic/antiresorptive strategies reduce recurrence is therefore timely. Our case illustrates the feasibility of combining post-operative denosumab with oral bisphosphonate therapy, supporting the rationale for systematic investigation of such combined strategies in high-risk SGCT.
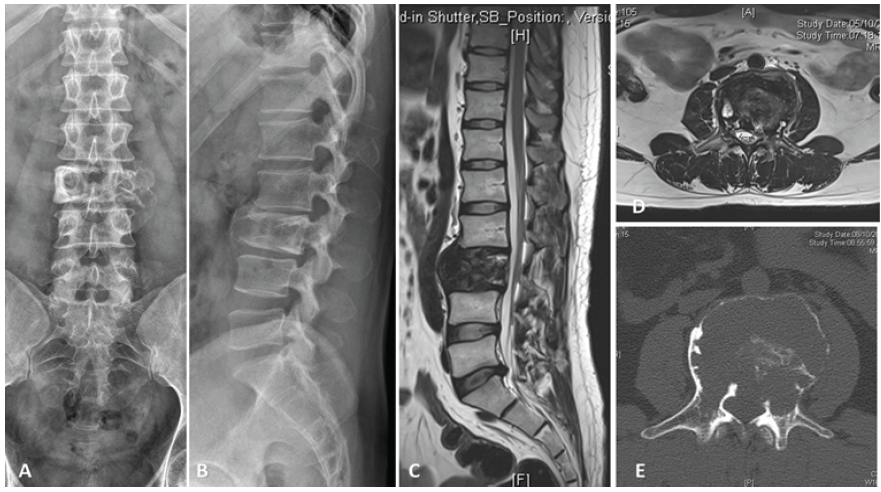
Figure 1: 27 year old male with (A and B) L3 vertebral body lesion. Magnetic resonance imaging (MRI) (C and D) and computed tomography (CT) scan (E) revealed left paravertebral soft-tissue component. A diagnosis of Enneking stage III Giant cell tumor of the L3 vertebral body involving zones 3–10 and layers A–D (Weinstein-Boriani-Biagini Classification [5]) was made following CT-guided biopsy.
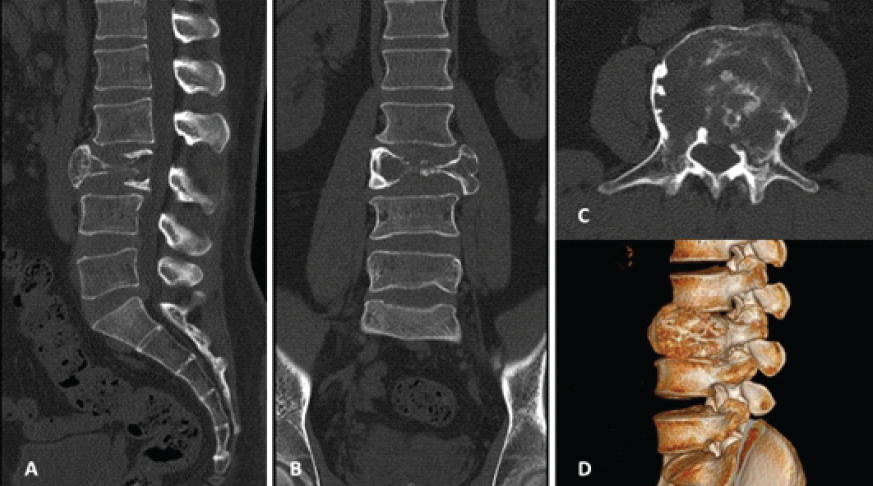
Figure 2: Sagittal [A], coronal [B], axial [C], and 3D reconstruction [D] computed tomography scan images after 6 months of Denosumab showed
improvement in cortical margin with mild increase in sclerosis.
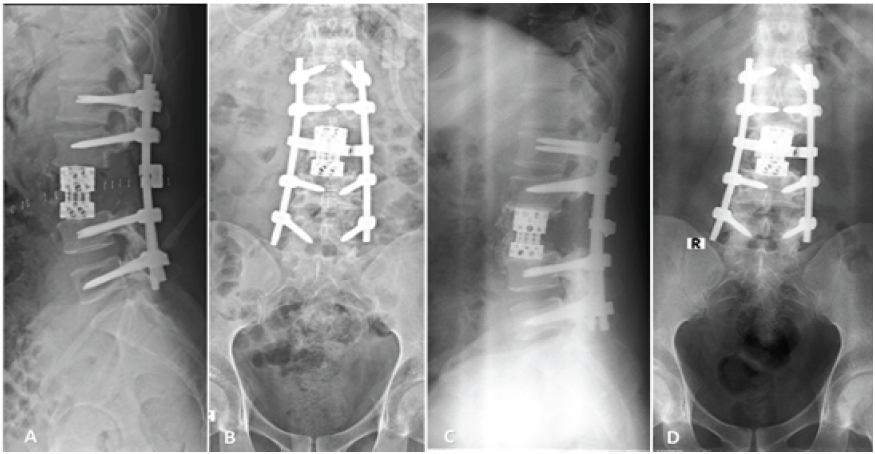
Figure 3: Lateral [A] and Anteroposterior (AP) [B] radiographs following L1 to L5 pedicle screw stabilization, proceed L3 gross total spondylectomy and
anterior column reconstruction. Lateral [C] and AP [D] radiographs 4.5 years after surgery showing osseous consolidation and stable instrumentation with
no evidence of recurrent disease.
SGCT is rare and surgically demanding [3,4]. En bloc spondylectomy offers the best oncologic control when achievable, but anatomy and proximity to neural and vascular structures may often preclude safe wide margins [3]. En bloc resection may also not be technically feasible as the tumor is friable and easily collapses during surgical manipulation with risk of consequent tumor cell seeding [6]. When en bloc resection is not possible, meticulous piecemeal gross total resection may still provide acceptable local control if no residual tumor is left [7]. These surgical realities and safety concerns related to the use of local adjuvants underscore interest in systemic adjuvants to reduce recurrence in SGCT [3].
Historically, bisphosphonates were used to reduce recurrence and improve pain in GCT [1]. Bisphosphonates bind to hydroxyapatite and are internalized by osteoclasts at sites of active resorption, where they inhibit farnesyl pyrophosphate synthase in the mevalonate pathway [8]. This disrupts prenylation of small GTP-binding proteins, leading to osteoclast dysfunction and apoptosis [2,8]. More importantly, in the context of GCT, in vitro and animal studies show that bisphosphonates not only induce apoptosis in osteoclast-like giant cells but also cause apoptosis in the neoplastic GCT stromal cells by exerting the same direct cytotoxic effect – suggesting a role in the reduction of local recurrence beyond just osteoclast – like giant cell suppression [2,9]. Several surgical series and retrospective cohorts suggest lower recurrence rates when bisphosphonates accompany surgery in the spine, and bisphosphonate-assisted strategies were widely adopted before denosumab availability [10,11]. Published data supports use of bisphosphonates as a biologically plausible adjuvant, but definitive level-1 evidence is lacking [1,12].
The introduction of denosumab shifted practice towards its pre-operative and adjuvant use for the management of aggressive GCT [1,4,12]. In contrast to bisphosphonates, Denosumab has a different mechanism of action and prevents RANKL from binding to its receptor, which inhibits the differentiation, activation, and survival of osteoclast-like giant cells [1]. This leads to rapid elimination of osteoclast-like giant cells, producing sclerosis and radiological mineralization that can facilitate less extensive resections and reduce intraoperative bleeding [1,4]. Clinically, it is effective for unresectable or metastatic GCT and for pre-operative downstaging. However, several important caveats exist: (a) denosumab preferentially targets only osteoclast-like giant cells and does not induce apoptosis of the neoplastic stromal cells [2]; (b) pre-operative denosumab can produce dense peripheral sclerosis that obscures residual tumor and may increase recurrence following curettage [1]; and (c) optimal post-operative duration remains undefined and recurrence after drug cessation has been reported [1]. Most contemporary reviews therefore recommend cautious, selective use of pre-operative denosumab (for downstaging where needed) and close long-term surveillance after discontinuation [12].
The differential, complementary mechanisms of denosumab and bisphosphonates provide a strong biological rationale for combined therapy. Denosumab rapidly suppresses RANKL-driven osteoclastogenesis and reduces bone resorption and tumor bulk, improving surgical planes, while bisphosphonates have direct anti-stromal effects (dose-dependent growth inhibition and apoptosis in multiple GCT stromal cell lines) targeting the proliferative neoplastic component that denosumab leaves relatively unaffected [2]. In vitro data directly comparing the two agents show significant stromal apoptosis with zoledronic acid but minimal apoptotic effect with denosumab, supporting the notion that bisphosphonates address a biologic gap left by denosumab [2].
Though Zoledronic acid is the most widely used bisphosphonate for the adjuvant treatment of GCT, other drugs in this class, including oral bisphosphonates, have also been shown to be efficacious [9,13,14]. Although all bisphosphonates have a similar mechanism of action, they differ in potency and adverse effect profile [8]. While Zoledronic acid has high anti-resorptive potency, alendronate and ibandronate carry lesser risk of adverse effects like renal injury, hypocalcemia, and osteonecrosis of the jaw (ONJ) [8].
Clinically, randomized evidence is not yet available to recommend routine concurrent administration. Safety concerns must be considered before combining agents: Both drug classes carry risks such as hypocalcemia and ONJ [8]. Any combined approach should therefore include dental assessment, serum calcium/Vitamin D optimization, and close monitoring for ONJ and hypocalcemia [3].
We used oral bisphosphonates (alendronate followed by ibandronate) along with Denosumab to decrease the possibility of adverse effects seen with the more potent Zoledronic acid. As a single-patient case report, this observation is inherently limited, and the relative contribution of combined adjuvant therapy cannot be clearly distinguished from the effects of surgical resection and stabilization. Although causal inference cannot be drawn from an individual case, the present case demonstrates the feasibility of combined post-operative denosumab and bisphosphonate therapy. To the best of our knowledge, there are no previously published reports describing the intentional combined post-operative use of Denosumab and oral bisphosphonates in SGCT to reduce the risk of recurrence. The purpose of this report is not to advocate a treatment strategy based on a single case, but rather to present the biological rationale for a novel combined approach for a challenging clinical scenario and to illustrate this concept through an individual case. The case highlights a novel strategy that warrants further investigation in aggressive SGCTs.
This report demonstrates the feasibility of combining post-operative Denosumab and oral bisphosphonate therapy as an adjuvant strategy for high-risk SGCT when wide surgical margins are not achievable. By targeting different cellular pathways involved in tumor biology, this approach may provide complementary tumor control.
Spinal giant cell tumors carry a high recurrence risk as wide margins are difficult to achieve. Combined post-operative denosumab and bisphosphonate therapy may represent a biologically rational adjuvant strategy in high-risk spinal giant cell tumors when en bloc resection is not feasible. Denosumab suppresses osteoclast-mediated osteolysis, while bisphosphonates exert direct anti-stromal effects. This complementary mechanism may help reduce recurrence in high-risk cases.
References
- 1. Van Der Heijden L, Lipplaa A, Van Langevelde K, Bovée JV, Van De Sande MA, Gelderblom H. Updated concepts in treatment of giant cell tumor of bone. Curr Opin Oncol 2022;34:371-8. [Google Scholar] [PubMed]
- 2. Lau CP, Huang L, Wong KC, Kumta SM. Comparison of the anti-tumor effects of denosumab and zoledronic acid on the neoplastic stromal cells of giant cell tumor of bone. Connect Tissue Res 2013;54:439-49. [Google Scholar] [PubMed]
- 3. Boriani S, Cecchinato R, Cuzzocrea F, Bandiera S, Gambarotti M, Gasbarrini A. Denosumab in the treatment of giant cell tumor of the spine. Preliminary report, review of the literature and protocol proposal. Eur Spine J 2020;29:257-71. [Google Scholar] [PubMed]
- 4. Goldschlager T, Dea N, Boyd M, Reynolds J, Patel S, Rhines LD, et al. Giant cell tumors of the spine: Has denosumab changed the treatment paradigm? J Neurosurg Spine 2015;22:526-33. [Google Scholar] [PubMed]
- 5. Boriani S, Weinstein JN, Biagini R. Primary bone tumors of the spine. Terminology and surgical staging. Spine (Phila Pa 1976) 1997;22:1036-44. [Google Scholar] [PubMed]
- 6. Boriani S, Bandiera S, Casadei R, Boriani L, Donthineni R, Gasbarrini A, et al. Giant cell tumor of the mobile spine: A review of 49 cases. Spine (Phila Pa 1976) 2012;37:E37-45. [Google Scholar] [PubMed]
- 7. Liu J, Hu P, Zhou H, Wang B, Liu X, Wu F, et al. Complications and prognosis of primary thoracic and lumbar giant cell tumors treated by total tumor resection. BMC Musculoskelet Disord 2023;24:281. [Google Scholar] [PubMed]
- 8. Papapetrou PD. Bisphosphonate-associated adverse events. Hormones (Athens) 2009;8:96-110. [Google Scholar] [PubMed]
- 9. Pannu CD, Kandhwal P, Raghavan V, Khan SA, Rastogi S, Jayaswal A. Role of bisphosphonates as adjuvants of surgery in giant cell tumor of spine. Int J Spine Surg 2018;12:695-702. [Google Scholar] [PubMed]
- 10. Xu W, Wang Y, Wang J, Yang X, Liu W, Zhou W, et al. Long-term administration of bisphosphonate to reduce local recurrence of sacral giant cell tumor after nerve-sparing surgery. J Neurosurg Spine 2017;26:716-21. [Google Scholar] [PubMed]
- 11. Yin H, Yang X, Xu W, Li B, Li B, Wang T, et al. Treatment and outcome of primary aggressive giant cell tumor in the spine. Eur Spine J 2015;24:1747-53. [Google Scholar] [PubMed]
- 12. Tsukamoto S, Mavrogenis AF, Masunaga T, Honoki K, Fujii H, Kido A, et al. Current concepts in the treatment of giant cell tumor of bone: An update. Curr Oncol 2024;31:2112-32. [Google Scholar] [PubMed]
- 13. Yu X, Xu M, Xu S, Su Q. Clinical outcomes of giant cell tumor of bone treated with bone cement filling and internal fixation, and oral bisphosphonates. Oncol Lett 2013;5:447-51. [Google Scholar] [PubMed]
- 14. Zhang W, Zhang Y, Li P, Rhodesm SD, Wang Y, Xue X, et al. Administration of sodium ibandronate in the treatment of complicated giant cell tumor of the spine. Spine (Phila Pa 1976) 2011;36:E1166-72. [Google Scholar] [PubMed]








