[box type=”bio”] Learning Point of the Article: [/box]
Following total hip arthroplasty, consideration should be given to metal ion toxicity due to trunnion wear in patients with cobalt/chromium femoral components, even with relatively low ion levels.
Case Report | Volume 9 | Issue 2 | JOCR March – April 2019 | Page 48-51 | Michael S Reich, Pooya Javidan, Vikram K Garg, Steven N Copp. DOI: 10.13107/jocr.2250-0685.1366
Authors: Michael S Reich[1],[4], Pooya Javidan[2],[4], Vikram K Garg[3], Steven N Copp[4]
[1]Department of Orthopaedics, Texas Tech University Health Sciences Center El Paso, 4801 Alberta Ave, El Paso, TX 79905, USA.
[2]Department of Orthopaedics, Southern California Permanente Medical Group, 250 Travelodge Dr, El Cajon, CA 92020, USA.
[3]Department of Rheumatology and Internal Medicine, Scripps Clinic, 310 Santa Fe Drive, Encinitas, CA 92024, USA.
[4]Department of Orthopaedic Surgery, Scripps Clinic, 10666 North Torrey Pines Road, MS116, La Jolla, CA 92037, USA.
Address of Correspondence:
Dr. Steven N. Copp,
Department of Orthopaedic Surgery, Scripps Clinic,10666 North Torrey Pines Road, MS116, La Jolla, CA 92037, USA.
E-mail: copp.steven@scrippshealth.org
Abstract
Introduction: Metal toxicity secondary to corrosion and wear has been reviewed in the arthroplasty literature with evidence supporting dermatologic, neurologic, and cardiac involvement. This is the first report of a novel case with the occurrence of systemic pathology with only mildly elevated serum cobalt (Co) and chromium (Cr) levels due to trunnion wear in a metal-on-polyethylene articulation.
Case Report: A 45-year-old female who had undergone a revision total hip arthroplasty developed concerning dermatologic, neurologic, and cardiac symptoms. Symptoms were initially attributed to an unknown autoimmune etiology. She had mildly elevated cobalt and chromium levels and subsequently underwent a second revision, this time with complete exchange of the cobalt/chromium components. By her 2-month follow-up, she reported partial resolution of symptoms. She continues to improve more than a year postoperatively.
Conclusion: Consideration should be given to metal ion toxicity due to trunnion wear in patients with cobalt/chromium femoral components, even with relatively low ion levels.
Keywords: Total hip arthroplasty, Metal ion toxicity, Trunnion wear.
Introduction
Modularity in total hip arthroplasty (THA) design has introduced surgical flexibility and implant options for both the surgeon and patient. With more widespread use, concerns of implant interaction at the modular junctions have been raised [1, 2]. More recently, the phenomenon of head-neck junction corrosion has been recognized as a cause of hip pain following arthroplasty [3, 4]. Reports of clinical symptoms, implant failure, and elevated metal ion levels causing adverse local tissue reactions (ALTRs), similar to previously described presentations in metal-on-metal (MoM) hip designs have been reported [5, 6, 7]. While these parallels have been explored, systemic manifestations including allergic dermatitis, neurological complications, and cardiac pathology due to metal toxicity have been less common and isolated almost exclusively to MoM articulations [8, 9]. We discuss a patient who presented with signs of systemic metal toxicity including dermatologic, neurologic, and cardiac manifestations secondary to metallosis at the head-neck junction of a modular implant. She went on to have improvement in her clinical symptoms following revision arthroplasty.
Case Report
The patient was verbally informed of the intent to publish the findings and consent was obtained through independent discussion for deidentified clinical images and clinical data to be published. The patient is now a 47-year-old female with a family history of rheumatoid arthritis, Hashimoto’s thyroiditis, and Grave’s disease. She had a non-contributory personal medical history before the onset of the symptoms described below, except for having developmental dysplasia of the right hip, multiple reconstructive surgeries as a child, and ultimately undergoing a right THA at age 22. She denied a metal allergy. She initially did well after her primary THA but went on to develop acetabular osteolysis which was treated with revision of her acetabular component with femoral head exchange 11 years later. The retained femoral component was an anatomic medullary locking (AML) hip stem (DePuy, Warsaw, Indiana), a cobalt-chromium-molybdenum alloy porous-coated femoral implant. This stem was retained along with implantation of a new 32 +5mm cobalt (Co)/chromium (Cr) femoral head. There was no gross evidence of corrosion or wear at the trunnion at the time of the revision surgery. Two years after her revision surgery, she sought care from rheumatology, cardiology, and her primary care physician.
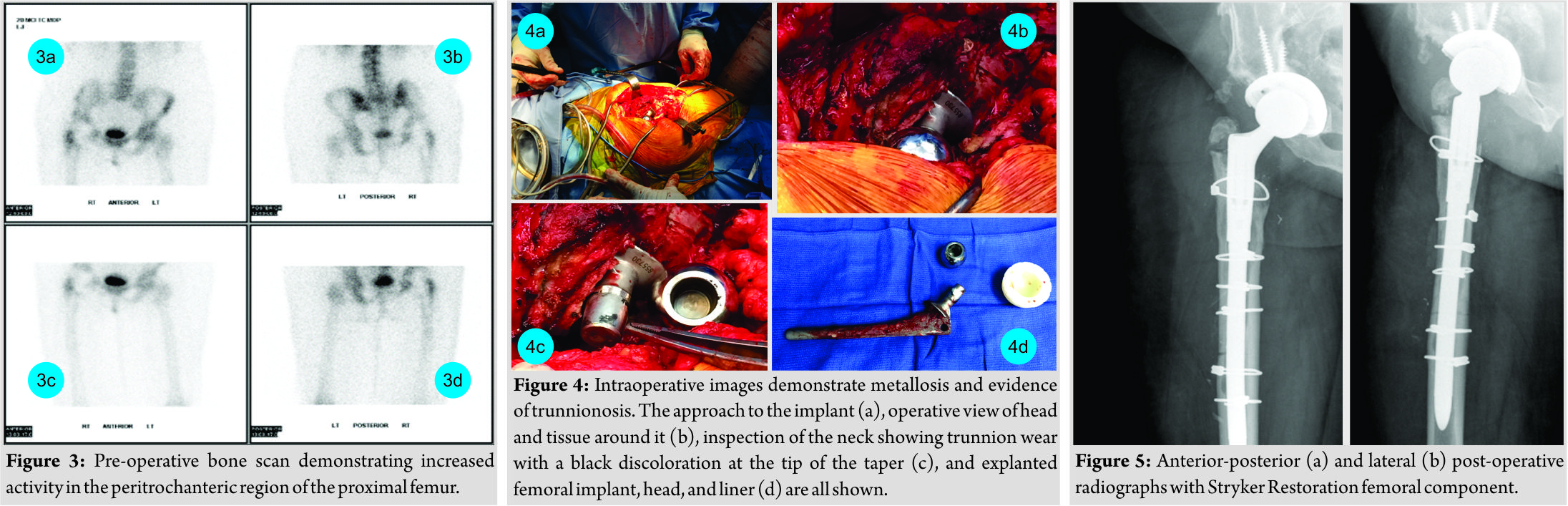
Discussion
While metal toxicity has been described in the arthroplasty literature, novel to this case is the occurrence of systemic pathology with only mildly elevated serum Co and Cr levels due to trunnion wear in a metal-on-polyethylene articulation. Furthermore, this represents the first report of clinically significant systemic metal toxicity linked to trunnion wear with the AML stem, though under revision circumstances, a widely implanted femoral stem with excellent long-term survivorship, and clinical outcomes reported at 20 years [11, 12]. Thresholds for concerning Co and Cr levels are still debated. For the evaluation of ALTR, some propose 4, 5, or 7 mcg/L as the cutoff for what should prompt further workup[10, 11, 13]. Systemic cobalt toxicity has been reported before with marginally elevated ion levels[14, 15], but levels <10 mcg/L should not lead to systemic symptoms[13]. Extensive laboratory studies were performed on our patient to evaluate for rheumatologic disorders, but in the end, the only notable findings were the patient’s dermatopathology and elevated Co and Cr levels. The patient’s dermatologist, rheumatologist, cardiologist, toxicologist, and two unaffiliated orthopedic surgeons all concurred that the patient’s presentation could be reflective of systemic metal ion toxicity. Reports of systemic symptoms in the setting of a metal-on-polyethylene articulation, with the causative source most likely wear at the head-neck junction, remain uncommon. A certain subset of patients may exist with clinical syndromes similar to the ones described in this case report whose symptoms could have been overlooked in the past. As in this case, these manifestations may be directly linked to chronic and low-level Co and Cr exposure. Consideration should be given to the possibility of trunnion wear in this stem as a cause of low-level metal toxicity. This becomes especially important in patients with similar clinical manifestations for whom the etiology of their symptoms have eluded definitive diagnosis.
Conclusion
This novel case of a 45-year-old female with the occurrence of systemic pathology with only mildly elevated serum cobalt (Co) and chromium (Cr) levels in a revised hip with metal-on-polyethylene articulation indicates that consideration should be given to metal ion toxicity due to trunnion wear, even with relatively low ion levels.
Clinical Message
In patients experiencing dermatologic, neurologic, and cardiac symptoms following total hip arthroplasty with cobalt/chromium femoral components, consideration should be given to metal ion toxicity due to trunnion wear, even in patients with relatively low ion levels.
References
1. Goldberg JR, Gilbert JL. In vitro corrosion testing of modular hip tapers. J Biomed Mater Res B Appl Biomater 2003;64:78-93.
2. Royhman D, Patel M, Runa MJ, Jacobs JJ, Hallab NJ, Wimmer MA, et al. Fretting-corrosion in hip implant modular junctions: New experimental set-up and initial outcome. Tribol Int 2015;91:235-45.
3. Mistry JB, Chughtai M, Elmallah RK, Diedrich A, Le S, Thomas M, et al. Trunnionosis in total hip arthroplasty: A review. J Orthop Traumatol 2016;17:1-6.
4. Jennings JM, Dennis DA, Yang CC. Corrosion of the head-neck junction after total hip arthroplasty. J Am Acad Orthop Surg 2016;24:349-56.
5. Gill IP, Webb J, Sloan K, Beaver RJ. Corrosion at the neck-stem junction as a cause of metal ion release and pseudotumour formation. J Bone Joint Surg Br 2012;94:895-900.
6. Cooper HJ, Urban RM, Wixson RL, Meneghini RM, Jacobs JJ. Adverse local tissue reaction arising from corrosion at the femoral neck-body junction in a dual-taper stem with a cobalt-chromium modular neck. J Bone Joint Surg Am 2013;95:865-72.
7. Mao X, Tay GH, Godbolt DB, Crawford RW. Pseudotumor in a well-fixed metal-on-polyethylene uncemented hip arthroplasty. J Arthroplasty 2012;27:493.e13-7.
8. Bradberry SM, Wilkinson JM, Ferner RE. Systemic toxicity related to metal hip prostheses. Clin Toxicol (Phila) 2014;52:837-47.
9. Wong CC, Nixon RL. Systemic allergic dermatitis caused by cobalt and cobalt toxicity from a metal on a metal hip replacement. Contact Dermatitis 2014;71:113-4.
10. Zywiel MG, Cherian JJ, Banerjee S, Cheung AC, Wong F, Butany J, et al. Systemic cobalt toxicity from total hip arthroplasties: Review of a rare condition part 2. Measurement, risk factors, and step-wise approach to treatment. Bone Joint J 2016;98-B:14-20.
11. Engh CA, Jr., Claus AM, Hopper RH, Jr., Engh CA. Long-term results using the anatomic medullary locking hip prosthesis. Clinical orthopaedics and related research 2001; (393): 137.
12. Belmont PJ Jr., Powers CC, Beykirch SE, Hopper RH Jr., Engh CA Jr., Engh CA, et al. Results of the anatomic medullary locking total hip arthroplasty at a minimum of twenty years. A concise follow-up of previous reports. J Bone Joint Surg Am 2008;90:1524-30.
13. Paustenbach DJ, Galbraith DA, Finley BL. Interpreting cobalt blood concentrations in hip implant patients. Clin Toxicol (Phila) 2014;52:98-112.
14. Mao X, Wong AA, Crawford RW. Cobalt toxicityan emerging clinical problem in patients with metal-on-metal hip prostheses? Med J Aust 2011;194:649-51.
15. Tower SS. Arthroprosthetic cobaltism: Neurological and cardiac manifestations in two patients with metal-on-metal arthroplasty: A case report. J Bone Joint Surg Am 2010;92:2847-51.
 |
 |
 |
 |
| Dr. Michael S Reich | Dr. Pooya Javidan | Dr. Vikram K Garg | Dr. Steven N Copp |
| How to Cite This Article: Reich M S, Javidan P, Garg V K, Copp S N. Chronic Systemic Metal Ion Toxicity from Wear on a Revised Cobalt-chromium Trunnion. Journal of Orthopaedic Case Reports 2019 Mar-Apr; 9(2): 48-51. |
[Full Text HTML] [Full Text PDF] [XML]
[rate_this_page]
Dear Reader, We are very excited about New Features in JOCR. Please do let us know what you think by Clicking on the Sliding “Feedback Form” button on the <<< left of the page or sending a mail to us at editor.jocr@gmail.com





