Levonadifloxacin can be considered a strong empirical option for bone and joint infections due to its high bioavailability, bone penetration, and effective biofilm activity.
Dr. Yogesh Vaidya, Pinnacle Orthocentre Hospital, Mumbai, Maharashtra, India. E-mail: vaidyayogesh044@gmail.com
Introduction: Bone and joint infections (BJIs) present significant therapeutic challenges due to factors such as limited drug penetration at the infection site, biofilm formation, and concerns regarding the safety of prolonged antibiotic use. If left untreated, these infections may cause lasting disability or death. To address this, an investigator-initiated study evaluated the clinical efficacy and safety of levonadifloxacin in patients with BJIs.
Materials and Methods: This was a prospective, multicentric, investigator-initiated study designed to assess the real-world effectiveness and safety of levonadifloxacin, administered either orally or intravenously, in adult patients (≥21 years) with BJIs. A total of 97 participants were enrolled. The treatment duration ranged up to 28 days. That exact duration of treatment was individualized based on the patient’s response and the physician’s discretion. Clinical response was evaluated on days 4, 8, 14, and 25, whereas the microbiological response was assessed on day 28. Safety monitoring included clinical and laboratory assessments, and the final evaluation was conducted at the end-of-treatment (EoT) (day 28) and test-of-cure (ToC) visit (day 35).
Results: Patients received levonadifloxacin for a minimum duration of 14 days (mean duration of treatment was 26.8 days). On day 4, clinical improvement (CI) was observed in 82.6% of patients, and improvement was further seen in 90.2% of patients on day 14, and CI was seen in all the patients on day 28. Significant reduction in wound size (length, width, and total wound area – from 52.2 cm2 to 11.2 cm2) was observed over a period of 28 days with levonadifloxacin (P < 0.0001) with improved mobility. Clinical cure was achieved in 97.8% at EoT and 96.4% at ToC, with no patients with clinical failure. Microbiological success reached 93.5%, with no clinical failures or notable changes in hematology, electrocardiogram, vital signs, or laboratory parameters. Its promising biofilm activity, availability of intravenous and oral formulations, high bioavailability, and adequate bone penetration make levonadifloxacin a compelling empirical choice over traditional agents such as vancomycin, linezolid, and teicoplanin.
Conclusion: Levonadifloxacin (IV/oral) provided good clinical efficacy in patients with BJIs. Moreover, it was found to be safe and well-tolerated even after administration for 35 days.
Keywords: Levonadifloxacin, clinical cure, microbiological success, bone and joint infection.
Bone and joint infections (BJIs), encompassing conditions such as osteomyelitis and prosthetic joint infections, may originate from hematogenous dissemination, direct inoculation following trauma or surgical intervention, or contiguous spread from adjacent soft-tissue infections. Management of BJIs typically necessitates a combination of surgical intervention (debridement) and prolonged antimicrobial therapy [1]. Inadequate or delayed treatment can lead to substantial functional impairment, including stiffness, chronic pain, deformities, and in severe cases, osteonecrosis or spread to other areas giving rise to sepsis and even death. The microbial etiology of BJIs is predominantly characterized by Gram-positive organisms, especially Staphylococcus aureus and coagulase-negative Staphylococci [1,2]. Consequently, antibiotics with potent Gram-positive coverage remain the cornerstone of therapeutic options for these infections. Several critical factors must be considered when selecting an appropriate treatment regimen for BJIs. In many cases, patients prefer oral therapy due to considerations of convenience and cost [3]. The decision between all-intravenous (IV) therapy, all-oral therapy, or a sequential IV-to-oral switch is influenced by the acute or chronic nature of the infection and the associated risk of complications (along with the presence of comorbidities). For an antibiotic to be clinically effective, it must achieve adequate concentrations within bone tissue, which is inherently limited in vascular supply [4]. In addition, the presence of biofilm at the infection site further complicates treatment outcomes [5]. To ascertain clinical efficacy, if an oral antibiotic is chosen, the serum concentration should be comparable to that achieved by IV administration. Furthermore, as mentioned earlier, prolonged therapy is often needed for chronic BJI cases, and in such scenarios, the chosen oral antibiotic should possess adequate safety so that the therapy can be completed until the desired clinical outcome is achieved. In the Indian context, most cases of BJIs are managed with antibiotics selected on an empirical basis. With about 40–50% of S. aureus being methicillin-resistant S. aureus (MRSA) and considering the risk for co-infection with Gram-negative organisms, an MRSA-active, broad-spectrum antibiotic is a preferable empirical choice [6,7]. With patient, pathogen- and antibiotic-specific factors having an influence on the clinical outcome, there is a strong unmet need over the existing antibiotics for better management of BJIs. Levonadifloxacin is a novel, broad-spectrum antibiotic belonging to the benzoquinolizine subclass of fluoroquinolones, developed by Wockhardt Ltd. It is highly effective against multi-drug-resistant Gram-positive bacteria, most notably MRSA. It targets both DNA gyrase and topoisomerase IV, two enzymes critical for bacterial DNA replication. It preferentially targets DNA gyrase, which helps it overcome common quinolone resistance mechanisms in S. aureus. Levonadifloxacin is available in both IV and oral formulations. Along with potent antibacterial activity against MRSA, it is also active against other Gram-positive pathogens [8,9]. Both formulations have been approved for the treatment of adult patients with skin concerns (acute bacterial and structural infection), including diabetic foot infection and concurrent bacteraemia [10]. The in vitro, non-clinical pharmacokinetic–pharmacodynamic (PK/PD) as well as clinical data suggest a potential role of levonadifloxacin in the management of BJIs. Conventional therapies for BJIs are often limited by suboptimal bone penetration, prolonged IV administration, adverse effects, and rising antimicrobial resistance, whereas surgical interventions carry high morbidity and relapse risk [11,12,13]. Given these limitations, there is a compelling rationale to evaluate the safety and efficacy of levonadifloxacin in real-world clinical settings for patients with BJIs. Accordingly, this investigator-initiated study was conducted to assess the therapeutic outcomes of levonadifloxacin in patients with clinically diagnosed BJIs. Participants received either IV, oral, or sequential IV-to-oral levonadifloxacin, or both, evaluating safety and efficacy parameters.
Study design
This was a prospective, multicenter investigator-initiated study to evaluate the real-world efficacy and safety of levonadifloxacin (administered orally and/or intravenously) in treating BJIs.
Ethics
Ethics approval for this study was obtained from the institutional ethics committees (IECs) of all participating sites (Approval number and date: HCH-EC_01/20210701 date July 26, 2021; DYP/IEC/12/2021 date June 10, 2021; IEC/Pharm/CT/347/February/2021 date February 20, 2021; IHEC/202/01/04 date September 23, 2021; and DYP/IEC/09-024/2020 date November 12, 2020) the study was registered under CTRI/2020/12/029718. All study procedures adhered to the Indian Council of Medical Research and the Central Drugs Standard Control Organisation good clinical practice guidelines, institutional and national ethical standards, and the Declaration of Helsinki (1975, revised in 2013). Written informed consent was obtained from all participants, and confidentiality of patient identity was strictly maintained.
Study participants
Patients aged 18 years (any gender) and above with clinically documented BJIs were recruited at six orthopedic centers in India. These infections encompassed a variety of clinical conditions, such as osteomyelitis, septic arthritis, prosthetic/implant-related infections, diabetic-foot infections complicated by osteomyelitis, and discitis/spinal osteomyelitis. The clinical signs and symptoms included redness, warmth, pain, tenderness, swelling, wound discharge, and purulent discharge. Laboratory parameters included elevated serum C-reactive protein (CRP), increased total leukocyte count, increased erythrocyte sedimentation rate, and evidence of BJI on radiograph.
Inclusion criteria
- Male or female subjects aged 18 years and above
- Subjects must be willing to participate in the study and provide a written informed consent
- Patients with Gram-positive bacteremia – BJI (osteomyelitis, septic arthritis, prosthetic infection, discitis/spinal osteomyelitis, and diabetic foot infection with osteomyelitis, etc.) except mycobacterial infection, to be treated with levonadifloxacin (oral or IV) as per the investigator’s judgment.
Exclusion criteria
- Subjects with a history of hypersensitivity to any of the study drugs or the same class of drugs
- Subjects who received any experimental drug within 30 days before enrolment
- Subjects who, in the judgment of the investigator, are likely to be non-compliant or uncooperative during the study
- Subjects with any abnormality that the investigator deems to be clinically relevant, either on medical history, physical examination, or electrocardiogram (ECG).
Study outcomes
Primary outcome
The primary outcome for this study was clinical response on days 4, 8, 14, 28 (end-of-treatment [EoT]), and day 35 (test-of-cure [ToC]). Clinical response was evaluated as “Clinical Cure (CC),” “Clinical Improvement (CI),” “Clinical failure (CF)” or “Clinical indeterminate (CI)” as per a predefined criterion. Clinical cure (CC) was defined as complete resolution of clinical signs and symptoms of BJI from baseline, along with no hospitalization due to worsening of baseline BJI, no need for additional antimicrobial, reduction in size of wound, no purulent discharge, or no new or recurrent sinus tract. CI was defined as improvement in at least two clinical signs and symptoms (improvement in pain, redness, tenderness, swelling, purulent discharge, and size of wound) of BJI from baseline without additional antibiotics, no worsening of pain and/or point tenderness, and improvement in inflammation (measured by CRP). Clinical failure was failure to meet all criteria for clinical success, worsening/progression of clinical condition leading to hospitalization, or need for additional antimicrobial drugs. Clinical indeterminate was designated when the evaluation was not possible (lost to follow-up, major protocol violation, lack of data critical for assessment of clinical response) for any reason. In addition, the CI was assessed by the study investigators on days 4, 8, and 14. The selected treatment duration of 28–35 days aligns with standard guidelines for BJIs, which recommend prolonged antimicrobial therapy due to the chronic nature and poor vascularity of infected bone tissue [14].
Secondary outcomes
The secondary outcome for this study was microbiological response at the EoT. Microbiological response was evaluated as “Microbiological success/cure,” “Microbiological failure,” and “Microbiological indeterminate.” Microbiological success was defined as the absence of organism which was present at baseline and not isolated in the follow-up microbial testing over a period of 28 days from the start of levonadifloxacin therapy. Microbiological failure was defined as persistence of the pathogen(s) identified at baseline. Microbiological indeterminate was designated when the evaluation was not possible (lost to follow-up, major protocol violation, missing data necessary for clinical response) for any reason.
Safety outcome
Safety of levonadifloxacin was assessed based on adverse events (AEs) reported after the start of therapy, vital signs, and physical examination findings, clinical laboratory evaluation, and ECG collected during the study.
Statistical analysis
The study was conducted over a 13-month period across six clinical sites, with the sample size formally calculated by a statistician. Descriptives were presented for demography and study outcomes. Measurement data were presented as means and standard deviation, whereas categorical data were presented as numbers with percentages. All patients who received at least one dose of levonadifloxacin were included in the intent-to-treat (ITT) population for safety analysis. The modified ITT (mITT) population comprised 92 patients who completed the prescribed treatment regimen and had evaluable follow-up data. Efficacy analyses were conducted using the mITT dataset, whereas safety evaluations were based on the full ITT cohort of 97 patients, including the five individuals without follow-up data.
Demography
Out of 97 patients, 92 patients completed the study, hence included in the mITT dataset. Five patients did not complete the study or had incomplete data. Table 1 presents the demographics of the patients in the mITT dataset.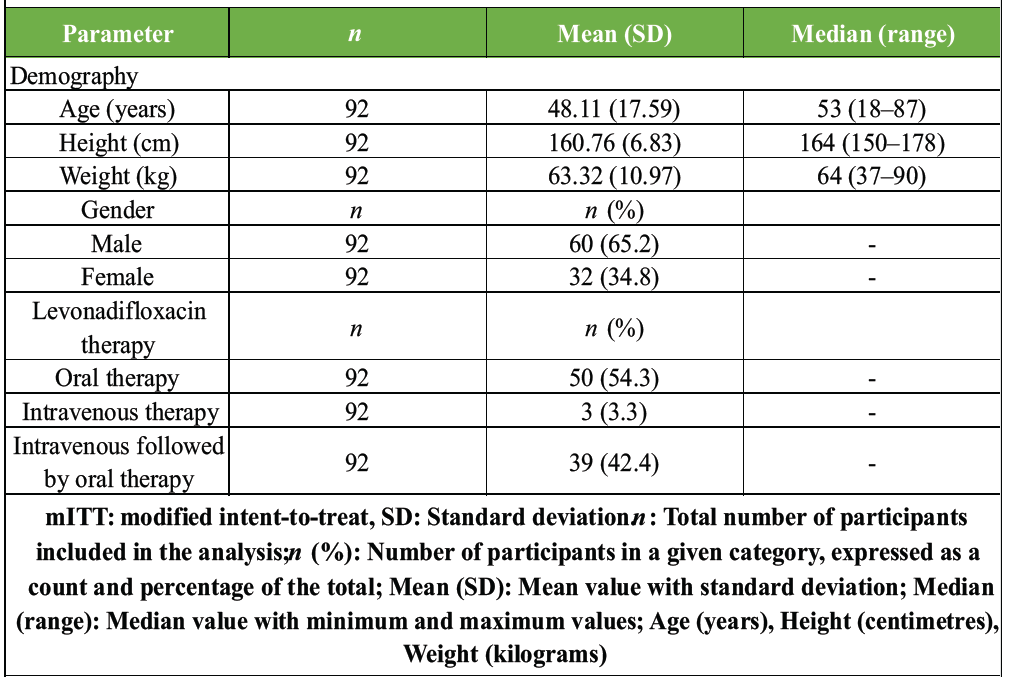
Table 1: Profile of patients and levonadifloxacin therapy received by the patients (mITT analysis set)The median age of the patients was 53 years (range: 18–87). Of the 92 patients, 65.2% were male, and 34.8% were female. Fifty (54.3%) patients received oral therapy, 3.3% received IV therapy, and 42.4% received IV followed by oral levonadifloxacin therapy. Patients received levonadifloxacin (IV or oral or sequential IV and oral) for a minimum duration of 14 days (mean duration of treatment was 26.8 days) to a maximum of 56 days.
At baseline, pain and tenderness of affected areas were observed in 98.9% and 96.7% patients, respectively at baseline. The mean wound area at baseline (n = 86) was 52.2 cm2 (range: 1–300 cm2). A total of 51 (97 samples) organisms were isolated at baseline, and 5 (92 samples) on day 28. However, microbial sensitivity data were available only for 46 isolates at baseline and 3 at EoT. The fewer number of isolates at EoT is attributed to either wound closure or the appearance of healthy granulation tissue, indicating resolution of infection. No resistance was observed among any of the 46 isolates tested. At baseline, the highest sensitivity was seen for S. aureus (7/8 sensitive), Achromobacter denitrificans (1/1), Acinetobacter junii (1/2), Chryseobacterium indologenes (1), Morganella morganii (1/3), Proteus mirabilis (1/4), Escherichia coli (1/6), Klebsiella pneumoniae (1/5), and Staphylococcus epidermidis (1/1). Organisms such as E. coli (5/6), Enterobacter cloacae (6/6), Enterobacter faecalis (2/2), Acinetobacter Baumannii (3/3), A. junii (1/2), K. pneumoniae (4/5), M. morganii (2/3), P. mirabilis (3/4), Proteus vulgaris (1/1), S. aureus (1/8), and Pseudomonas aeruginosa (3/3) showed intermediate susceptibility, suggesting partial responsiveness to levonadifloxacin.
At EoT, only 33.7% patients had pain and 30.4% had tenderness after levonadifloxacin therapy compared to baseline. Improvement in redness was seen in 71.0% patients, whereas swelling was resolved in 72.9% patients at EoT. Similar improvements were observed for warmth, wound discharge/drainage, pus, sinus tract, and localized movement restriction/decreased mobility. Significant reduction in wound size (length, width, and total wound area) was observed over a period of 28 days with levonadifloxacin therapy. (Fig. 1) At EoT, the mean wound area significantly reduced from 52.2 cm2 to 11.2 cm2 on day 28. (Table 2, 3)
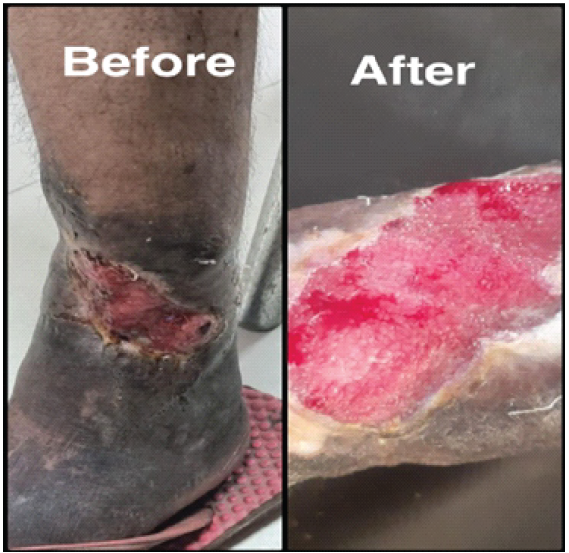
Figure 1: Healing progression of soft-tissue wound associated with bone and joint infection. Clinical photographs of the patient’s lower leg ulcer showing healing progression. The image on the left depicts the ulcer located on the medial aspect of the lower leg above the ankle at initial presentation, prior to treatment. The image on the right shows the same ulcer site after treatment, demonstrating healthy granulation tissue formation. Both images are standard clinical photographs taken in the outpatient setting, with the “after” image captured approximately 5 weeks following initiation of therapy.
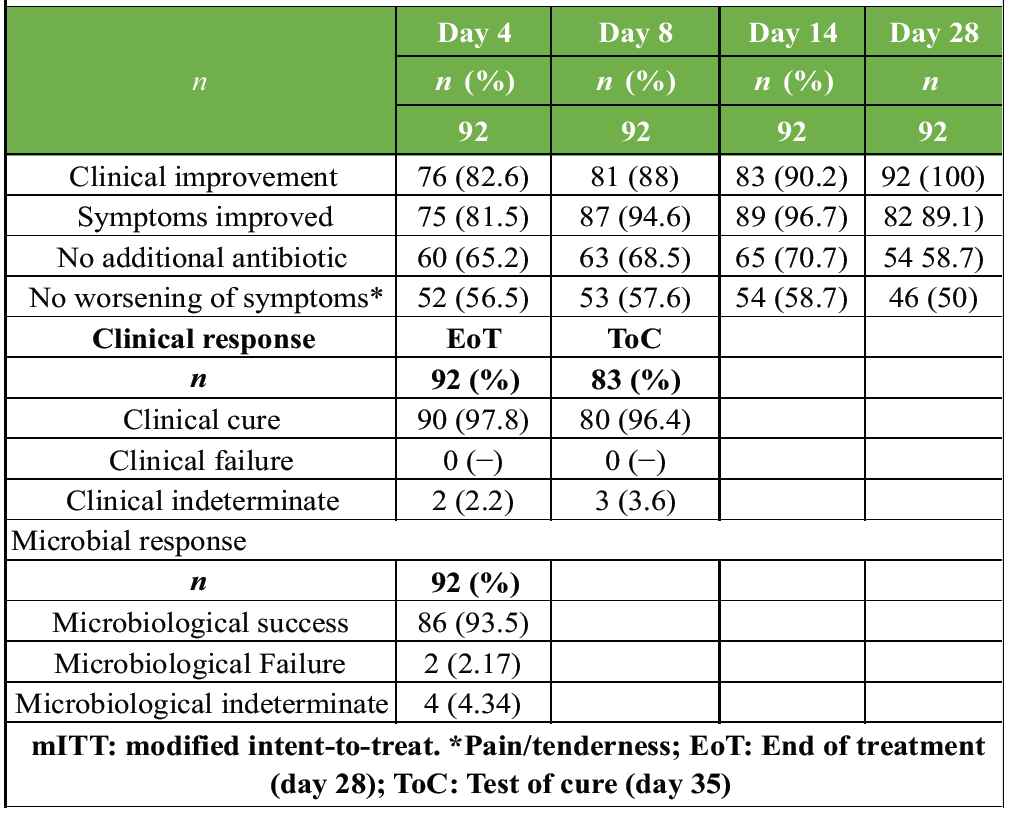
Table 2: Clinical outcomes with levonadifloxacin in the mITT dataset
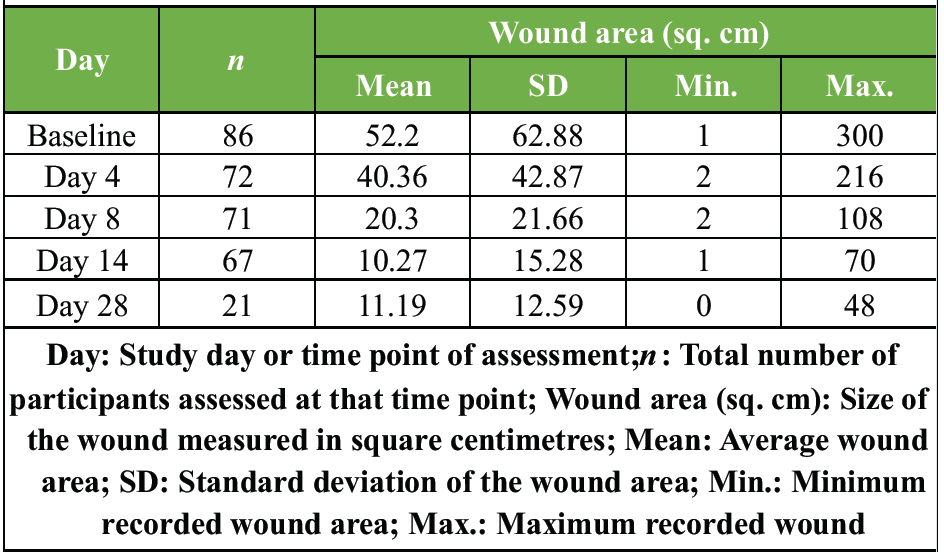
Table 3: Wound area at baseline and on days 4, 8, 14, and 28 of levonadifloxacin treatment
At EoT (day 28), the three organisms isolated were K. pneumoniae (1 sensitive), P. mirabilis (1 intermediate), and S. aureus (1 intermediate). The minimum inhibitory concentration (MIC) values for levonadifloxacin were A. baumannii (4–12 mg/L), A. junii (1 mg/L), Alcaligenes faecalis (8 mg/L), C. indologenes (5 mg/L), Citrobacter werkmanii (12 mg/L), Enterococcus faecalis (8 mg/L), E. coli (8 mg/L), K. pneumoniae (8 mg/L), P. mirabilis (6 mg/L), P. aeruginosa (1–8 mg/L), S. aureus (5–8 mg/L), S. aureus (MRSA) (12 mg/L), and S. epidermidis (Methicillin-Resistant Coagulase-Negative Staphylococci- MRCONS) (8 mg/L).
Primary outcome
Table 2 presents the clinical outcomes assessed in the mITT population at day 4, 8, 14, and 28. On day 4, CI was observed in 82.6% patients, and improvement was further seen in 90.2% of patients on day 14, and CI was seen in all the patients on day 28. At the EoT and ToC visits, clinical response data were available for a total of 92 and 83 patients, respectively. CC was achieved in 97.8% at EoT and 96.4% at ToC, with no patients with clinical failure. Indeterminate outcomes were observed in 2.2% (EoT) and 3.6% (ToC).
Secondary outcomes
At baseline, while overall culture growth was observed in 47.4% of tissue samples, with Gram-positive organisms in 16.3%, Gram-negative in 30.9%, and polymicrobial infections in 3.3% of patients. The culture negativity at baseline was probably due to prior antibiotic exposure, biofilm-associated infection, or a fastidious organism not detected by routine methods. By EoT, Gram-positive and Gram-negative isolates were found in only 1.1% and 2.2% of patients, respectively, with no evidence of polymicrobial growth, and culture positivity was reduced to 3.3%. Common organisms isolated at baseline were S. aureus (including MRSA), P. aeruginosa, and E. coli. These data support the broad-spectrum activity of levonadifloxacin, particularly against Gram-positive pathogens and select Gram-negative organisms. Overall, the study demonstrates a 93.5% microbiological success rate with no evidence of clinical failure. These outcomes are particularly significant given the widespread presence of difficult-to-treat pathogens such as S. aureus (including MRSA), P. aeruginosa, and E. coli, often associated with polymicrobial and resistant infections. From an antimicrobial standpoint, levonadifloxacin exhibits potent in vitro activity against biofilm-forming S. aureus, including MRSA strains. Its favorable PK profile, marked by 90% oral bioavailability and adequate bone penetration, supports its rational use in BJI. Moreover, comparative analyses suggest that levonadifloxacin performs better than traditional agents such as vancomycin, linezolid, and teicoplanin, which are known to have limitations in bone tissue efficacy [18,24,25,26].
This study demonstrates a strong clinical and microbiological efficacy of levonadifloxacin in treating BJIs, with a CC rate of 97.8% at EoT and 96.4% at ToC (day 35).
Safety
No changes from baseline in the vital parameters (systolic blood pressure and diastolic blood pressure), laboratory parameters (alanine aminotransferase and aspartate aminotransferase), or 12-lead ECG [QRS (ventricular depolarization), PR (start of atrial depolarization to the start of ventricular depolarization.), QT (start of ventricular depolarization to the end of ventricular repolarization.), and QTcF (QT corrected by Fridericia’s formula) intervals] were observed on day 28 with levonadifloxacin therapy. Of the 97 patients who received levonadifloxacin, a total of three AEs (cough, n = 1; fever, n = 1; and cold, n = 1) were reported in patients. All events were mild in severity, and none of the events were determined to be related to levonadifloxacin by the investigators. All events resolved without sequelae, and there was no discontinuation of levonadifloxacin therapy due to AEs.
BJIs present a considerable therapeutic challenge due to factors such as compromised vascularity, biofilm formation, and rising antimicrobial resistance – particularly from MRSA [15]. Levonadifloxacin has emerged as a promising therapeutic option owing to its broad-spectrum activity, potent anti-MRSA efficacy, and high oral bioavailability [16]. The effective management of BJIs relies on the ability of the antibiotic to reach the site of infection and exert antibacterial action. The presence of biofilms further adds a layer of complexity, impeding antibiotic efficacy [17]. Involvement of antibiotic-resistant pathogens such as MRSA further complicates the treatment course. Conventional anti-MRSA antibiotics have several drawbacks with respect to the treatment of BJIs (Tables 5 and 6). Glycopeptides such as vancomycin and teicoplanin are restricted to IV administration, exhibit poor bone tissue penetration, and also lack biofilm activity [17]. Although linezolid offers both IV and oral formulations, its use beyond 10 days is constrained by the risk of cytopenias. In contrast, levonadifloxacin offers both IV and oral routes – with 90% oral bioavailability – demonstrates adequate bone tissue penetration, and crucially, exhibits potent activity against MRSA biofilms [18]. The reported activity of levonadifloxacin against key pathogens is implicated in BJIs across multiple studies. In previous in vitro studies involving biofilm-embedded S. aureus, levonadifloxacin displayed a consistent ≥90% bacterial kill rate at clinically relevant concentrations, whereas vancomycin and daptomycin displayed inferior activity, and even linezolid failed to eradicate biofilm [19]. Furthermore, levonadifloxacin has shown a favorable safety profile during prolonged administration. The present study was conducted to evaluate whether these PDs and safety advantages translate into real-world clinical efficacy and tolerability in the treatment of BJIs. This prospective, multicenter, investigator-initiated study assessed the real-world efficacy and safety of oral and/or IV levonadifloxacin in 97 adults with BJIs across six orthopedic centers in India. CC rates were 97.8% at EoT and 96.4% at ToC (day 35), with microbiological success being 93.5% and no clinical failures reported. Common pathogens included MRSA, P. aeruginosa, and E. coli, underscoring the drug’s broad-spectrum utility. From an antimicrobial perspective, levonadifloxacin exhibits excellent in vitro activity against biofilm-producing S. aureus, including MRSA. Its high oral bioavailability (~90%) and adequate bone penetration make it a suitable choice for treating BJIs. When compared to standard agents such as vancomycin, linezolid, and teicoplanin, which have known limitations in terms of efficacy and safety in managing BJIs, levonadifloxacin presents a favorable profile. A total of three AEs (3.1%) were reported during the study. These included cough in 1 patient (1.0%), cold in 1 patient (1.0%), and fever in 1 patient (1.0%). All reported events were mild in nature. These findings suggest that levonadifloxacin is a well-tolerated and effective empirical option for BJIs, particularly in cases involving resistant or mixed infections, and support its potential role in future comparative clinical trials. None of the patients reported a serious AE or any AE leading to treatment discontinuation. Both the IV and oral formulations were well tolerated. This study demonstrated that, at the recommended dosing regimen, oral levonadifloxacin achieved adequate systemic exposure, penetrated bone tissue effectively, and provided broad coverage against common BJI pathogens with high CC rates as monotherapy. Its dual targeting of DNA gyrase and topoisomerase IV enables it to overcome common quinolone resistance mechanisms, whereas its favorable PK profile ensures high oral bioavailability, effective bone penetration, and robust anti-biofilm activity. Recent surveillance data from Indian tertiary care centers reported 100% susceptibility of MRSA isolates to levonadifloxacin, compared with markedly lower rates for conventional agents such as doxycycline and cotrimoxazole, and high resistance to older fluoroquinolones. These findings underscore levonadifloxacin’s potential as a reliable therapeutic option in BJIs where MRSA is a frequent pathogen, supporting its empirical use in real-world clinical practice [20]. Levonadifloxacin exhibits potent activity against key pathogens implicated in BJIs, particularly Gram-positive organisms such as S. aureus (including MRSA and resistant strains), with high susceptibility rates. It also shows promising MIC values against anaerobic bacteria, although specific susceptibility data for these organisms remain limited. Overall, levonadifloxacin represents a highly suitable therapeutic option for the management of complex BJIs [8,9,16,19,21,22,23]. BJIs, particularly those that are deep-seated or chronic, often exhibit acidic microenvironments due to poor vascularization, necrotic tissue, and biofilm formation. Under such conditions, the efficacy of many antibiotics is significantly reduced, limiting their therapeutic utility. Levonadifloxacin, however, retains potent antibacterial activity in acidic environments, making it especially suitable for the treatment of BJIs. This acid-stable profile, coupled with rapid bactericidal action, effective bone penetration, and robust anti-biofilm activity, supports its use as a viable monotherapy against key pathogens commonly implicated in BJIs [16]. Levonadifloxacin demonstrates broad-spectrum activity, rapid bactericidal action, and potent biofilm activity, whereas these features are limited or absent with the comparator antibiotics. Unlike teicoplanin and linezolid, levonadifloxacin is not associated with thrombocytopenia. It also shows enhanced antibacterial activity in acidic conditions commonly present in deep-seated BJIs. In terms of pharmacological properties, levonadifloxacin exhibits excellent bone tissue penetration, whereas linezolid also shows good penetration but lacks several other advantages. In addition, levonadifloxacin has a safety profile suitable for long-term use, whereas the comparator agents have limitations related to prolonged therapy [8,9,10,16,19,24,25,26,27,28,29]. The in vitro activity of levonadifloxacin against various bacterial isolates demonstrates favorable MIC values and high susceptibility rates for several clinically relevant pathogens. For S. aureus, the MIC50 and MIC90 values were 0.005 mg/L and 1.000 mg/L, respectively. MRSA showed MIC50 and MIC90 values of 0.500 mg/L and 1.000 mg/L with a susceptibility rate of 91.5%. The Bengal Bay clone of S. aureus and hetero-vancomycin-intermediate S. aureus demonstrated MIC50/MIC90 values of 0.250/0.500 mg/L with 100% susceptibility. For coagulase-negative Staphylococci, MIC50 and MIC90 values were 0.060 mg/L and 2.000 mg/L, with a susceptibility rate of 93.4%. Among Streptococci, β-hemolytic streptococci and viridans group streptococci both exhibited MIC50 and MIC90 values of 0.250 mg/L, with susceptibility rates of 98.3% and 97.2%, respectively. For anaerobic organisms, levonadifloxacin demonstrated MIC50/MIC90 values of 1/16 mg/L for Bacteroides spp., 0.25/1 mg/L for Prevotella spp. and Porphyromonas spp., 0.5/8 mg/L for Fusobacterium spp., 0.5/1 mg/L for Peptostreptococci, and 0.06/0.25 mg/L for Clostridium perfringens. These findings highlight the broad-spectrum antimicrobial activity of levonadifloxacin against key Gram-positive and anaerobic pathogens [8,9,16,19,21,22,23,30]. Levonadifloxacin has a broader antimicrobial coverage compared with the comparator antibiotics used in BJIs. It is active against methicillin-susceptible and S. aureus and MRSA, coagulase-negative Staphylococci, Streptococcus species, quinolone-susceptible Gram-negative organisms, and anaerobes. In contrast, teicoplanin, vancomycin, and linezolid mainly cover Gram-positive organisms and lack activity against Gram-negative pathogens, with linezolid showing only limited activity against anaerobes [18,24,25,26]. A key strength of this study lies in its real-world design, which enhances the generalizability of findings to routine clinical practice. The structured follow-up schedule spanning Days 4, 8, 14, 28, and 35 enabled a comprehensive longitudinal assessment of therapeutic response. Safety was rigorously monitored through serial ECG evaluations, laboratory investigations, and AE tracking. Furthermore, microbiological correlation was established by documenting microbial clearance, thereby reinforcing the clinical relevance and robustness of the study outcomes. While this study demonstrates the safety and efficacy of IV and oral levonadifloxacin in the treatment of BJIs under real-world conditions, several limitations merit consideration. Foremost, the study was investigator-initiated and non-comparative, lacking a control arm with standard-of-care agents such as vancomycin or linezolid, thereby limiting conclusions regarding relative efficacy and safety. The study was also conducted across a limited number of centers, potentially affecting the diversity and representativeness of the patient population. In addition, baseline microbiological cultures were available in fewer than 50% of patients, limiting the ability to correlate clinical outcomes with pathogen-specific responses and raising questions about the rationale for empirical therapy. Furthermore, the safety assessment was limited to a 35-day follow-up period, which may be insufficient to detect delayed adverse effects, particularly relevant for quinolones, which are known to cause late-onset side effects. Our inclusion criteria encompassed a broad spectrum of Gram-positive bacteremia associated with BJIs. While this enhances external validity and reflects real-world clinical practice, it may introduce heterogeneity in terms of prior antimicrobial exposure, presence of sequestrum, and varying resistance patterns. To mitigate these concerns, susceptibility testing was performed, prior drug use was documented, and patients requiring surgical source control were included only after appropriate intervention. Baseline microbiological cultures were available in fewer than half of patients, reflecting real-world challenges such as prior antibiotic exposure, biofilm-associated infections, and fastidious organisms. Nevertheless, the availability of cultures and susceptibility testing reports was limited, and no resistance was observed among the tested isolates. The empiric use of levonadifloxacin was therefore justified by its potent Gram-positive activity, including MRSA, high oral bioavailability, bone penetration, and anti-biofilm efficacy. While this approach enhances external validity, the limited culture data should be considered a study limitation. Longer follow-up is essential to assess relapse rates and late-onset AEs, particularly for quinolones. To strengthen future research, it is recommended to conduct randomized controlled comparative studies, expand the geographic scope of study centers, ensuring comprehensive microbiological documentation. These improvements would strengthen the evidence base and support broader clinical applicability.
This study addresses an important disease BJIs by evaluating levonadifloxacin, a novel antibiotic available in both IV and oral formulations, in a real-world, multicenter Indian setting involving 97 patients. The study demonstrates high microbiological and CC rates. It delineates a favorable safety profile even with prolonged use, which is particularly relevant given the limitations of existing MRSA-active therapies. Structured in accordance with standard guidelines, the study’s novelty lies in its real-world data, especially in cases involving severe, life-threatening infections and difficult-to-treat pathogens with chronic infections. Despite its limitations in being a non-comparative study, including restricted geographic representation and limited baseline microbiological data, the findings offer a compelling rationale for future randomized controlled trials. Such trials would help establish levonadifloxacin’s comparative efficacy and safety, thereby reinforcing its potential role in the broader clinical management of BJIs.
Levonadifloxacin demonstrated strong clinical and microbiological effectiveness in treating BJIs, with rapid improvement and sustained cure rates. Its dual IV and oral formulations, high bioavailability, and good bone penetration make it a practical choice for real-world management. The drug was well tolerated even with extended use, supporting its utility as a reliable therapeutic option in challenging osteoarticular infections.
References
- 1. Colston J, Atkins B. Bone and joint infection. Clin Med 2018;18:150-4. [Google Scholar] [PubMed]
- 2. Prabhoo R, Chaddha R, Iyer R, Mehra A, Ahdal J, Jain R. Overview of methicillin resistant Staphylococcus aureus mediated bone and joint infections in India. Orthop Rev (Pavia) 2019;11:8070. [Google Scholar] [PubMed]
- 3. Ferry T, Lustig S, Laurent F, Soriano A. Editorial: Innovative approaches in the management of bone and joint infection. Front Med (Lausanne) 2021;8:789092. [Google Scholar] [PubMed]
- 4. Thabit AK, Fatani DF, Bamakhrama MS, Barnawi OA, Basudan LO, Alhejaili SF. Antibiotic penetration into bone and joints: An updated review. Int J Infect Dis 2019;81:128-36. [Google Scholar] [PubMed]
- 5. Dastgheyb SS, Hammoud S, Ketonis C, Liu AY, Fitzgerald K, Parvizi J, et al. Staphylococcal persistence due to biofilm formation in synovial fluid containing prophylactic cefazolin. Antimicrob Agents Chemother 2015;59:2122-8. [Google Scholar] [PubMed]
- 6. Anupurba S, Sen MR, Nath G, Sharma BM, Gulati AK, Mohapatra TM. Prevalence of methicillin resistant Staphylococcus aureus in a tertiary referral hospital in eastern Uttar Pradesh. Indian J Med Microbiol 2003;21:49-51. [Google Scholar] [PubMed]
- 7. Tiwari H, Sapkota D, Sen MR. High prevalence of multidrug-resistant MRSA in a tertiary care hospital of Northern India. Infect Drug Resist 2008;1:57-61. [Google Scholar] [PubMed]
- 8. Bakthavatchalam YD, Shankar A, Muniyasamy R, Peter JV, Marcus Z, Triplicane Dwarakanathan H, et al. Levonadifloxacin, a recently approved benzoquinolizine fluoroquinolone, exhibits potent in vitro activity against contemporary Staphylococcus aureus isolates and Bengal Bay clone isolates collected from a large Indian tertiary care hospital. J Antimicrob Chemother 2020;75:2156-9. [Google Scholar] [PubMed]
- 9. Appalaraju B, Baveja S, Baliga S, Shenoy S, Bhardwaj R, Kongre V, et al. In vitro activity of a novel antibacterial agent, levonadifloxacin, against clinical isolates collected in a prospective, multicentre surveillance study in India during 2016-18. J Antimicrob Chemother 2020;75:600-8. [Google Scholar] [PubMed]
- 10. Bhatia A, Mastim M, Shah M, Gutte R, Joshi P, Kumbhar D, et al. Efficacy and safety of a novel broad-spectrum anti-MRSA agent levonadifloxacin compared with linezolid for acute bacterial skin and skin structure infections: A phase 3, openlabel, randomized study. J Assoc Physicians India 2020;68:30-6. [Google Scholar] [PubMed]
- 11. Hooper DC, Jacoby GA. Mechanisms of drug resistance: Quinolone resistance. Ann N Y Acad Sci 2015;1354:12-31. [Google Scholar] [PubMed]
- 12. Tande AJ, Patel R. Prosthetic joint infection. Clin Microbiol Rev 2014;27:302-45. [Google Scholar] [PubMed]
- 13. Gould K, Ager S. Clinical update on linezolid in the treatment of gram-positive bacterial infections. Infect Drug Resist 2012;5:87-102. [Google Scholar] [PubMed]
- 14. Saavedra-Lozano J, Falup-Pecurariu O, Faust SN, Girschick H, Hartwig N, Kaplan S, et al. Bone and joint infections. Pediatr Infect Dis J 2017;36:788-99. [Google Scholar] [PubMed]
- 15. Peng J, Guo C, Yang C, Zhang L, Yang F, Huang X, et al. Phage therapy for bone and joint infections: A comprehensive exploration of challenges, dynamics, and therapeutic prospects. J Glob Antimicrob Resist 2024;39:12-21. [Google Scholar] [PubMed]
- 16. Bhagwat SS, Nandanwar M, Kansagara A, Patel A, Takalkar S, Chavan R, et al. Levonadifloxacin, a novel broad-spectrum anti-mrsa benzoquinolizine quinolone agent: Review of current evidence. Drug Des Devel Ther 2019;13:4351-65. [Google Scholar] [PubMed]
- 17. Saseedharan S, Dubey D, Singh RK, Zirpe K, Choudhuri AH, Mukherjee DN, et al. Treatment challenges in the management of difficult-to-treat gram-positive infections: A consensus view apropos therapeutic role of novel anti-MRSA antibiotics, levonadifloxacin (IV) and alalevonadifloxacin (oral). Indian J Med Microbiol 2024;47:100528. [Google Scholar] [PubMed]
- 18. Mehta Y, Mishra KC, Paliwal Y, Rangappa P, Sinha S, Bhapkar S. Meeting the unmet need in the management of MDR gram-positive infections with oral bactericidal agent levonadifloxacin. Crit Care Res Pract 2022;2022:2668199. [Google Scholar] [PubMed]
- 19. Tellis M, Joseph J, Khande H, Bhagwat S, Patel M. In vitro bactericidal activity of levonadifloxacin (WCK 771) against methicillin- and quinolone-resistant Staphylococcus aureus biofilms. J Med Microbiol 2019;68:1129-36. [Google Scholar] [PubMed]
- 20. Mehta Y, Sutar AR, Zirpe K, Kothari JN, Alapati C, Pathak M, et al. Prescription-event monitoring study on safety and efficacy of levonadifloxacin (oral and I.V.) in management of bacterial infections: Findings of real-world observational study. Int J Appl Basic Med Res 2022;12:30-6. [Google Scholar] [PubMed]
- 21. Fatima N, Tejan N, Dinkar A, Kumar A, Patel SS, Yaduvanshi N, et al. Assessment of In vitro Activity of Levonadifloxacin Against Clinical Bacterial Isolates in Patients Admitted in ICU in a Tertiary Care Hospital. In Review; 2025. Available from: https://www.researchsquare.com/article/rs-7029796/v1 [Last accessed on 2025 Sep 15]. [Google Scholar] [PubMed]
- 22. Bhagwat SS, McGhee P, Kosowska-Shick K, Patel MV, Appelbaum PC. In vitro activity of the quinolone WCK 771 against recent U.S. Hospital and community-acquired Staphylococcus aureus pathogens with various resistance types. Antimicrob Agents Chemother 2009;53:811-3. [Google Scholar] [PubMed]
- 23. Flamm RK, Rhomberg PR, Sader HS. In Vitro Activity of the Novel Lactone Ketolide Nafithromycin (WCK 4873) against Contemporary Clinical Bacteria from a Global Surveillance Program. Antimicrob Agents Chemother. 2017 Dec;61(12):e01230-17. doi:10.1128/AAC.01230-17 [Google Scholar] [PubMed] [CrossRef]
- 24. Bakthavatchalam YD, Rao SV, Isaac B, Manesh A, Nambi S, Swaminathan S, et al. A comparative assessment of clinical, pharmacological and antimicrobial profile of novel anti-methicillin-resistant Staphylococcus aureus agent levonadifloxacin: Therapeutic role in nosocomial and community infections. Indian J Med Microbiol 2019;37:478-87. [Google Scholar] [PubMed]
- 25. Behra-Miellet J, Calvet L, Dubreuil L. Activity of linezolid against anaerobic bacteria. Int J Antimicrob Agents 2003;22:28-34. [Google Scholar] [PubMed]
- 26. Dubois J, Dubois M. Levonadifloxacin (WCK 771) exerts potent intracellular activity against Staphylococcus aureus in THP-1 monocytes at clinically relevant concentrations. J Med Microbiol 2019;68:1716-22. [Google Scholar] [PubMed]
- 27. Liu Y, Liu T, Xiao K, Wang J, Yan P, Fang X, et al. Effects of oral versus intravenous linezolid administration on treatment effect and incidence of thrombocytopenia in patients with severe infection. Intensive Care Res 2024;4:171-9. [Google Scholar] [PubMed]
- 28. Saseedharan S, Zirpe K, Mehta Y, Dubey D, Sutar A, Debnath K, et al. Efficacy and safety of oral and IV levonadifloxacin therapy in management of bacterial infections: Findings of a prospective, observational, multi-center, post-marketing surveillance study. Cureus 2024;16:e55178. [Google Scholar] [PubMed]
- 29. Seng P, Vernier M, Gay A, Pinelli PO, Legré R, Stein A. Clinical features and outcome of bone and joint infections with streptococcal involvement: 5-year experience of interregional reference centres in the south of France. New Microbes New Infect 2016;12:8-17. [Google Scholar] [PubMed]
- 30. Greenwood D. Microbiological properties of teicoplanin. J Antimicrob Chemother 1988;21 Suppl A:1-13. [Google Scholar] [PubMed]










