Accurate distinction of non-neoplastic bone lesions from neoplastic mimics requires integrated clinicoradiological–pathological correlation to avoid overtreatment.
Dr. Karthik Shunmugavelu, Department of Dentistry, PSP Medical College Hospital and Research Institute, Kanchipuram, Tamil Nadu, India. E-mail: drkarthiks1981@gmail.com
Introduction: From inflammatory to neoplastic disorders, a variety of pathological bone lesions can manifest. Only 0.5% of all cancer kinds are bone tumors, making them comparatively rare. Cartilaginous, osteoid, fibrous, and bone marrow components make up bone. Benign or malignant tumors can develop in any tissue. Most of the current research focuses on uncommon tumors and their even less frequent molecular events, even though the ordinary surgical pathologist evaluates significantly more non-neoplastic orthopedic pathology on a regular basis. Our observations from concentrated seminars and consultations strongly suggest that a knowledge and diagnosis gap still exists in certain non-neoplastic orthopedic disorders. Revision arthroplasty and the assessment of infection in periprosthetic joints are among the most common intraoperative consultations conducted in the United States, both in academic and private facilities. In this technique, pathologists are essential because they assist in evaluating intraoperatively whether patients need antibiotic spacers before reimplantation. Since they (and their surgeons) wrongly think that a thorough pathological evaluation yields little therapeutically important information, several pathology departments have stopped examining arthroplasty specimens. Bone tumor-like lesions are those that resemble tumors in appearance and cytogenetics but exhibit clinical behavior that suggests they are not malignant. Tumor-like bony lesions are significant because they are widespread and resemble real bone tumors, including malignant lesions, on radiographs.
Materials and Methods: Major databases such as Medline were explored detailed literature search in resulting in a systematic review pertaining to non-neoplastic orthopedic pathology updates: Common problems and pitfalls and how to avoid them.
Results: Six original research scientific articles dated between 2020 and 2024 pertaining to mentioned topic were highlighted.
Conclusion: Although primary musculoskeletal neoplasms account for <1% of all musculoskeletal surgical pathology cases, they are disproportionately emphasized in the surgical pathology literature compared to non-neoplastic orthopedic pathology. Detailed information regarding the non-neoplastic orthopedic pathology updates: common problems and pitfalls and how to avoid them is discussed in this systematic review.
Keywords: Musculoskeletal, tumors, bone tumors, diagnostic imaging, histopathology, non-neoplastic lesions.
According to a review of Global Burden of Disease 2019 data, musculoskeletal disorders affect 1.71 billion people globally and are the leading cause of disability and the requirement for rehabilitation. Of the 570 million common cases of musculoskeletal disorders globally, low back pain is the primary cause, resulting from a variety of traumas and illnesses, including bone diseases. Although they can affect people of any age, bone-related diseases are more common as people age. Osteoporosis is linked to cardiovascular disease and arterial calcifications, osteoarthritis (OA) to sarcopenia, and both to disability, reduced quality of life, morbidity, and mortality. These conditions frequently coexist with other non-communicable diseases and raise the chance of developing additional comorbidities. Bone fractures, OA, rheumatoid arthritis, spondyloarthritis, benign or malignant primary bone tumors, and bone cancer metastases can all cause bone-related discomfort. Bone pain is also linked to rare and inherited bone and joint conditions that afflict children and young adults, such as fibrous dysplasia and osteogenesis imperfecta. A diverse group of neoplasms known as bone tumors can develop directly from the bone (primary tumors) or spread to the bone from other parts of the body (secondary or metastatic cancers). Benign and malignant kinds of primary bone tumors are further classified, and each exhibits distinct biological characteristics, clinical symptoms, and treatment responses. Understanding the intricacy of these circumstances and creating effective management plans depends on this classification. Osteosarcoma is the most prevalent primary malignant bone tumor, especially in young adults and adolescents during periods of fast growth. The formation of osteoid tissue or immature bone by malignant cells is a characteristic of this tumor type. Bone tumors are rare, and histological analysis is essential for the precise diagnosis and treatment of bone lesions. In many respects, the pathologist’s role is like a lens that focuses several light beams on a single place. The most relevant final diagnosis can be obtained by tempering microscopic findings by superimposing imaging results, clinical information, and the surgeon’s personal impression, both before and after surgery, on the glass slide.
“Non-neoplastic” AND “bone” AND “pathology’’ were the words used in MEDLINE database using advance search strategy targeting different article categories between 2020 and 2024. The result was 58 articles, out of which we selected 6 articles based in the inclusion criteria. Inclusion criteria were case studies and scientific literature between 2020 and 2024. Exclusion criteria were those of scientific literature irrelevant to the specific search. This systematic review was conducted to determine the importance of non-neoplastic orthopedic pathology updates: Common problems and pitfalls, and how to avoid them following the guidelines of the Preferred Reporting Items for systematic reviews and meta-analyses PubMed, Lilacs, Embase, Scopus, and Web of Science were the source of electronic databases. The search strategy used Boolean operators (AND and OR): (ALL [“non-neoplastic”] AND [bone OR musculoskeletal OR pathology OR histopathology OR diagnostics] AND [incidence]). The following data were collected: First author, year, country of study, type of study and outcome. The quality of studies was assessed using the Strengthening the Reporting of Observational Studies checklist.
Six articles were included in this systematic review based on the selection criteria. We analyzed and mentioned in the six articles reviewed. This included only relevant research articles and excluded articles pertaining to non-specific search terms (Table 1).
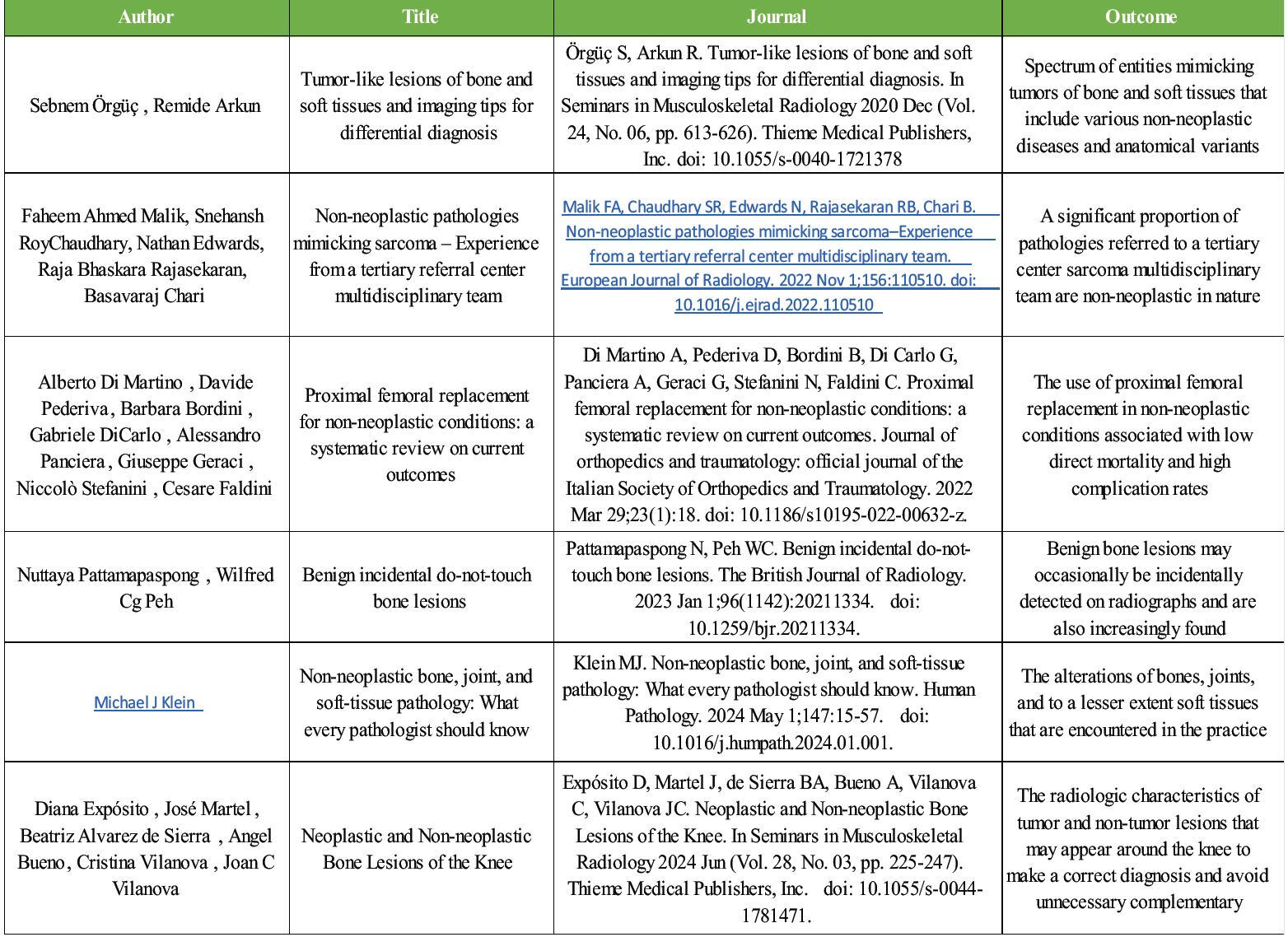
Table 1: An overview
Bone tumors represent a heterogeneous collection of neoplasms that originate either directly from the bone or metastasize to the bone from other sites in the body [1]. Understanding the prevalence and distribution of bone tumors is crucial for healthcare providers to develop effective screening, diagnostic, and treatment strategies [2]. The surveillance, epidemiology, and end results (SEER) database, maintained by the National Cancer Institute, collects and publishes cancer incidence and survival data from population-based cancer registries covering approximately 34.6% of the U.S population [3]. Techniques such as magnetic resonance imaging (MRI), computed tomography (CT), and positron emission tomography scans have significantly enhanced the ability to detect and diagnose bone tumors at earlier stages [4]. The incidence rates of bone tumors vary widely across different populations and regions. According to the SEER database, the overall incidence rate of primary malignant bone tumors is approximately 0.9/100,000 individuals [5]. Osteosarcoma accounts for about 35% of all primary malignant bone tumors, followed by chondrosarcoma (30%) and Ewing’s sarcoma (16%) [6]. Globally, the incidence rates show significant variability. For instance, the incidence of osteosarcoma is higher in some Asian countries compared to Western countries [7]. Several risk factors have been identified for the development of bone tumors. Age is a significant risk factor, with certain types of tumors being more prevalent in specific age groups. Gender also plays a role, with some tumors common in males than females [8]. Genetic predispositions, such as hereditary retinoblastoma and Li-Fraumeni syndrome, increase the risk of developing bone tumors. In addition, exposure to radiation and certain chemicals has been linked to an increased risk of bone tumors [9]. Biopsy is the gold standard for diagnosing bone tumors, as it allows for histopathological examination of the tumor tissue. Molecular and genetic testing can also provide valuable information for diagnosis and treatment planning [10]. Bone incidentalomas are not uncommonly found on radiographs and increasingly, CT or MRI performed for other clinical reasons, typically pain or injury at a bony site [11]. Although mostly asymptomatic, the incidental bone lesions detected on imaging may be developmental or acquired, be regarded as normal variants, or even be normal, that is, pseudolesions [12]. Anatomically, long bones include femur; flat bones include pelvis; short bones include hand and feet, and irregular bones include wrist, ankle and spine [13]. The human bone is affected by various pathological conditions, which comprise; degenerative diseases, inflammatory diseases, metabolic diseases and neoplastic lesions. Neoplastic lesions constitute; benign, malignant and metastatic lesions [14]. Diagnosis of bone lesions is a great challenge to a practicing pathologist and requires a comprehensive support of clinical, laboratory, radiological and histopathological correlation [15]. The need for accurate demographic details, such as age, gender and the affected site, becomes important, for the simple reason that many benign lesions mimic malignant lesions [16]. Age-related fractures are projected to exceed over 3 million in 2025. The prevalence of degenerative joint disease (DJD)/OA worldwide is 7%, affecting almost 500 million people [17]. There are approximately 719,000 total knee arthroplasties and 332,000 total hip arthroplasties annually, and these numbers do not reflect total shoulder replacements and other similar surgeries in the hands and feet [18]. Additional non-neoplastic specimens likely to be encountered, often due to non-healing ulcers, gangrene, or osteomyelitis, non-osteochondroma exostoses, entheses, trigger finger and carpal tunnel syndrome (CTS) biopsies, and loose bodies [19]. DJD/OA is the most common form of joint disease worldwide. It is a disease of articular cartilage, characterised by progressive erosion and loss of the articular cartilage surface with secondary changes [20]. DJD/OA may be a primary disorder or occur secondary to other pre-existing conditions, including trauma, inflammation, crystal deposition disease, osteonecrosis, and congenital abnormalities [21]. Most arise as a primary condition, possibly related to ageing and overuse, with most patients older than 60 years of age, and some have suggested that underutilization may be an even greater risk factor [22]. Clinical symptoms include joint pain, often worse in the morning, stiffness and reduced the range of motion. Patients with secondary DJD/OA report similar symptoms but often present at an earlier age [23]. Significant chronic lymphoplasmacytic inflammation, especially if accompanied by synovial hyperplasia and surface fibrin/fibrinoid necrosis, should raise the possibility of inflammatory arthritis [24]. The most common “loose body” is the osteocartilaginous loose bodies (OLB) associated with severe DJD/OA. OLB displays concentric layering, composed of variable amounts of viable proliferating cartilage and necrotic bone [25]. OLB should be distinguished from the far less common synovial chondromatosis (SC), a benign but locally aggressive hyaline cartilage neoplasm occurring within the synovium, bursa and/ or tendon sheaths surrounding joints [26]. Compared with OLB, the “loose bodies” of SC are typically more uniform in size and shape, occur in the absence of DJD/OA and typically involve the large joints of younger patients, between 30 and 50 years of age, with males affected twice as often as females [27]. Avascular necrosis (AVN), also known as aseptic necrosis, may occur in any bone and can be traumatic or atraumatic but, especially in the hip, is pathologically defined as subchondral osteonecrosis in the absence of hip dislocation/fracture and infection [28]. Risk factors include trauma, chronic steroid use, autoimmune disease, especially systemic lupus erythematous, decompression sickness, radiation, alcohol use, organ transplant particularly kidney, sickle cell anemia, leukemia, and Gaucher’s disease [29]. Reliable recognition of the condition is important as patients with AVN have an increased risk (at least 50%) of developing the disease in the contralateral hip as well as other bone sites [30]. An important differential diagnosis to consider for both DJD/OA with secondary osteonecrosis and AVN is subchondral insufficiency fracture (SIF). It is a small fracture/microfractures occurring due to deficient elastic resistance underneath the articular cartilage [31]. Classic clinical setting is elderly women with osteoporosis; however, it may also affect younger adults, particularly active and obese males. Other risk factors include renal and kidney transplant and systemic lupus erythematosus [32]. Unlike DJD/OA and AVN, SIF patients report acute onset pain usually because of minor injury such as twisting or long walks with heavy bags [33].
With progression, a “healing” linear fracture callus may replace the initial acute fracture. A subset appears to resolve with conservative therapy, but other cases undergo rapid progression and collapse, leading to destructive arthropathy [34]. Rapidly progressive/rapidly destructive arthritis represents a rapidly progressive and non- infectious degenerative change, occurring over a short period of time, culminating in femoral head collapse [35]. There is significant overlap with so- called neuropathic joint; however, underlying mechanisms remain unclear. The disease occurs mostly in elderly women, involving unilateral or bilateral hip joints with progressive destruction of the joint [36]. Radiologically, rapid loss of articular surface with joint space narrowing is characteristic, and collapse of the femoral head may be appreciated. Osteophytes are not formed due to the short time interval [37]. Sequential radiographs illustrating rapid loss of articular surface with joint space narrowing are characteristic, and complete collapse of the femoral head may be appreciated [38]. Gout has plagued humanity for more than 4500 years; Population studies reveal a pooled global range of prevalence of <1–6.8% and an incidence of 0.58–2.89/1000 person-years [39]. It is caused by persistent chronic hyperuricemia resulting in deposition of monosodium urate crystals in soft tissues and joints, excess dietary purine or underexcretion [40]. Regardless of etiology, the clinical manifestations of gouty arthritis encompass four distinct syndromes – asymptomatic hyperuricemia, nephrolithiasis, acute gouty arthritis and chronic tophaceous gout; the latter two are of primary concern to the pathologist [41]. The most common clinical manifestation is a painful arthritis, frequently monoarticular and most often afflicting, at least initially, the first metatarsophalangeal joint, generally resolving within days to weeks, only to recur later [42]. Overall, treatment involves relieving the pain of acute gout, using non-steroidal anti-inflammatory drugs, as well as the prevention of future attacks with colchicine and/or allopurinol [43]. Like gouty arthritis, calcium pyrophosphate deposition (CPPD) deposition disease is a form of arthritis characterized by crystal deposition within the joints. The clinical features of CPPD deposition disease vary quite considerably, ranging from asymptomatic, incidental joint calcifications (chondrocalcinosis) to acute arthritis, closely resembling gout (pseudogout) [44]. Amyloidosis (AL) in carpal tunnel release AL represents a group of disorders involving protein misfolding, which results in aggregates of extracellular deposits of amyloid fibrils, leading eventually to organ dysfunction [45]. CTS represents the most common peripheral neuropathy of the upper extremity. Indeed, it is even more frequently observed in systemic AL, and the vast majority have bilateral symptoms [46]. Pathological fractures result from local or generalized bone disorder leading to qualitative reduction in bone strength, and disorders of production, resorption, remodeling and local bone destruction are chief causes of pathological fractures [47]. Various causes lead to abnormal bone quality, leading to pathological fracture, and primary factors of causation are the load pattern and altered strength of the bone. The pattern, clinical proile and management data are essential to know the burden of the problem [48]. Regarding an already fractured bone, surgical fixation has been linked to better function and improved quality of life. A multidisciplinary approach is advocated for the management [49]. As tumors like lesions of the bone are not neoplasms, they need not be treated as aggressively as other bone tumors. Most of the cases can be left alone. They need to be treated only if they are symptomatic or likely to produce a pathological fracture [50].
A crucial area of oncology, bone tumors are distinguished by their distinct epidemiological characteristics and substantial effects on afflicted patients. This systematic study provides important information about the incidence rates, survival rates, and risk factors related to various types of bone tumors. Given that patients with localized disease had substantially higher survival rates than those with metastatic disease, the review emphasizes the significance of early detection and therapy intervention. The need for specialized treatment approaches is highlighted by the observed differences in survival rates among various tumor forms. Growth patterns, environmental exposures, and genetic predispositions are key risk factors that have been identified and can inform targeted screening procedures for high-risk groups. The growing identification of bone cancers, made possible by improvements in imaging methods and increased consciousness among medical practitioners, points to a promising trend in early diagnosis. Bone tumor-like lesions are those that resemble tumors in appearance and cytogenetics but exhibit clinical behavior that suggests they are not malignant. The tumor-like bony lesions are significant because they are highly prevalent and resemble real bone tumors, including malignant lesions, on radiographs. Bone lesions that resemble tumors might be aggressive, like aneurysmal bone cysts, active, like simple bone cysts, or latent, like non-ossifying fibromas. For their management, there are no clear rules available. Unlike other bone tumors, they do not require the same level of rigorous treatment.
Non-neoplastic bone lesions often mimic malignancy on imaging and histology. Careful linicoradiological correlation is essential to avoid misdiagnosis and unnecessary aggressive treatment. Most lesions require conservative management rather than oncologic intervention
References
- 1. Franchi A. Epidemiology and classification of bone tumors. Clin Cases Miner Bone Metab 2012;9:92-5. [Google Scholar] [PubMed]
- 2. Mirzaei S, Saghari S, Bassiri F, Raesi R, Zarrabi A, Hushmandi K, et al. NF‐κB as a regulator of cancer metastasis and therapy response: A focus on epithelial-mesenchymal transition. J Cell Physiol 2022;237:2770-95. [Google Scholar] [PubMed]
- 3. Tarver T. Cancer facts and figures 2012. American cancer society (ACS). J Consum Health Internet 2012;16:366-7. [Google Scholar] [PubMed]
- 4. Chakarun CJ, Forrester DM, Gottsegen CJ, Patel DB, White EA, Matcuk GR Jr. Giant cell tumor of bone: Review, mimics, and new developments in treatment. Radiographics 2013;33:197-211. [Google Scholar] [PubMed]
- 5. Xu Y, Shi F, Zhang Y, Yin M, Han X, Feng J, et al. Twenty‐year outcome of prevalence, incidence, mortality and survival rate in patients with malignant bone tumors. Int J Cancer 2024;154:226-40. [Google Scholar] [PubMed]
- 6. Freeman AK, Sumathi VP, Jeys L. Primary malignant tumours of the bone. Surgery (Oxford) 2018;36:27-34. [Google Scholar] [PubMed]
- 7. Mirabello L, Troisi RJ, Savage SA. International osteosarcoma incidence patterns in children and adolescents, middle ages and elderly persons. Int J Cancer 2009;125:229-34. [Google Scholar] [PubMed]
- 8. Frentzel-Beyme R, Becher H, Salzer-Kuntschik M, Kotz R, Salzer M. Factors affecting the incident juvenile bone tumors in an Austrian case-control study. Cancer Detect Prev 2004;28:159-69. [Google Scholar] [PubMed]
- 9. Miwa S, Yamamoto N, Hayashi K, Takeuchi A, Igarashi K, Tsuchiya H. Surgical site infection after bone tumor surgery: Risk factors and new preventive techniques. Cancers (Basel) 2022;14:4527. [Google Scholar] [PubMed]
- 10. Hashemi M, Abbaszadeh S, Rashid M, Amini N, Anaraki KT, Motahhary M, et al. STAT3 as a newly emerging target in colorectal cancer therapy: Tumorigenesis, therapy response, and pharmacological/nanoplatform strategies. Environ Res 2023;233:116458. [Google Scholar] [PubMed]
- 11. Velasco BT, Ye MY, Chien B, Kwon JY, Miller CP. Prevalence of incidental benign and malignant lesions on radiographs ordered by orthopaedic surgeons. J Am Acad Orthop Surg 2020;28:e356-62. [Google Scholar] [PubMed]
- 12. Levine J, Petkovska I, Landa J, Bates DD, Capanu M, Fuqua JL 3rd, et al. Bone lesions on baseline staging rectal MRI: Prevalence and significance in patients with rectal adenocarcinoma. Abdom Radiol (NY) 2021;46:2423-31. [Google Scholar] [PubMed]
- 13. Schaefer M, Black SM, Schaefer MC, Scheuer L. Juvenile Osteology. London: Academic Press; 2009. [Google Scholar] [PubMed]
- 14. Bamanikar SA, Pagaro PM, Kaur P, Chandanwale SS, Bamanikar A, Buch AC. Histopathological study of primary bone tumours and tumour-like lesions in a medical teaching hospital. J Krishna Inst Med Sci Univ 2015;4:46-55. [Google Scholar] [PubMed]
- 15. Deoghare SB, Prabhu MH, Ali SS, Inamdar SS. Histomorphological spectrum of bone lesions at tertiary care centre. Int J Life Sci Sci Res 2017;3:980-5. [Google Scholar] [PubMed]
- 16. Modi D, Rathod GB, Delwadia KN, Goswami HM. Histopathological study of bone lesions-A review of 102 cases. Int Arch Integr Med 2016;3:27-36. [Google Scholar] [PubMed]
- 17. Amin S, Achenbach SJ, Atkinson EJ, Khosla S, Melton LJ 3rd. Trends in fracture incidence: A population‐based study over 20 years. J Bone Miner Res 2014;29:581-9. [Google Scholar] [PubMed]
- 18. Kumar KV, Raghul VR, Pradeep E, Pandian H, Vijay A, Sheik M. Single stance radiography of the knee joint – a novel approach to assess the degree of knee osteoarthritis. J Orthop Case Rep 2024;14:184-9. [Google Scholar] [PubMed]
- 19. Yakkanti RR, Haziza S, Wasserman NA, Annapareddy A, Ratnakar V, Karri SR, et al. Relative frequency of avascular necrosis of the hip as indication for primary total hip arthroplasty in the USA vs. India J Orthop 2023;36:1-6. [Google Scholar] [PubMed]
- 20. Johnson VL, Hunter DJ. The epidemiology of osteoarthritis. Best Pract Res Clin Rheumatol 2014;28:5-15. [Google Scholar] [PubMed]
- 21. Zhang Y, Jordan JM. Epidemiology of osteoarthritis. Clin Geriatr Med 2010;26:355-69. [Google Scholar] [PubMed]
- 22. Alexander CJ. Relationship between the utilisation profile of individual joints and their susceptibility to primary osteoarthritis. Skeletal Radiol 1989;18:199-205. [Google Scholar] [PubMed]
- 23. Zhang L, Mirza SZ, DiCarlo EF. Subarticular inflammatory pseudoabscesses: A pathologic study with clinical correlation. Am J Surg Pathol 2020;44:633-40. [Google Scholar] [PubMed]
- 24. O’Connell JX, Nielsen GP, Rosenberg AE. Subchondral acute inflammation in severe arthritis: A sterile osteomyelitis? Am J Surg Pathol 1999;23:192-7. [Google Scholar] [PubMed]
- 25. Bianchi S, Martinoli C. Detection of loose bodies in joints. Radiol Clin North Am 1999;37:679-90. [Google Scholar] [PubMed]
- 26. Davis RI, Hamilton A, Biggart JD. Primary synovial chondromatosis: A clinicopathologic review and assessment of malignant potential. Hum Pathol 1998;29:683-8. [Google Scholar] [PubMed]
- 27. Neumann JA, Garrigues GE, Brigman BE, Eward WC. Synovial chondromatosis. JBJS Rev 2016;4:e2. [Google Scholar] [PubMed]
- 28. Chau MM, Klimstra MA, Wise KL, Ellermann JM, Tóth F, Carlson CS, et al. Osteochondritis dissecans: Current understanding of epidemiology, etiology, management, and outcomes. J Bone Joint Surg Am 2021;103:1132-51. [Google Scholar] [PubMed]
- 29. Lavernia CJ, Sierra RJ, Grieco FR. Osteonecrosis of the femoral head. J Am Acad Orthop Surg 1999;7:250-61. [Google Scholar] [PubMed]
- 30. Goker B, Block JA. Risk of contralateral avascular necrosis (AVN) after total hip arthroplasty (THA) for non-traumatic AVN. Rheumatol Int 2006;26:215-9. [Google Scholar] [PubMed]
- 31. Yamamoto T, Bullough PG. Subchondral insufficiency fracture of the femoral head: A differential diagnosis in acute onset of coxarthrosis in the elderly. Arthritis Rheum 1999;42:2719-23. [Google Scholar] [PubMed]
- 32. Yamamoto T, Karasuyama K, Iwasaki K, Doi T, Iwamoto Y. Subchondral insufficiency fracture of the femoral head in males. Arch Orthop Trauma Surg 2014;134:1199-203. [Google Scholar] [PubMed]
- 33. Yamamoto T, Schneider R, Bullough PG. Subchondral insufficiency fracture of the femoral head: Histopathologic correlation with MRI. Skeletal Radiol 2001;30:247-54. [Google Scholar] [PubMed]
- 34. Yamamoto T. Subchondral insufficiency fractures of the femoral head. Clin Orthop Surg 2012;4:173-80. [Google Scholar] [PubMed]
- 35. Yamamoto T, Bullough PG. The role of subchondral insufficiency fracture in rapid destruction of the hip joint: A preliminary report. Arthritis Rheum 2000;43:2423-7. [Google Scholar] [PubMed]
- 36. Hu L, Zhang X, Kourkoumelis N, Shang X. The mysteries of rapidly destructive arthrosis of the hip joint: A systemic literature review. Ann Palliat Med 2020;9:1220-9. [Google Scholar] [PubMed]
- 37. Layfield LJ, Crim JR. Destructive arthropathy of the femoral head: Pathologic findings in a case series. Am J Clin Pathol 2022;157:273-8. [Google Scholar] [PubMed]
- 38. Chau HT, Wong PY, Pan NY, Ma KF. Rapidly destructive hip osteoarthritis: A diagnosis not to miss. Br J Radiol 2024;97:1526-33. [Google Scholar] [PubMed]
- 39. Dehlin M, Jacobsson L, Roddy E. Global epidemiology of gout: Prevalence, incidence, treatment patterns and risk factors. Nat Rev Rheumatol 2020;16:380-90. [Google Scholar] [PubMed]
- 40. Dermawan JK, Goldblum A, Reith JD, Kilpatrick SE. The incidence and significance of calcium pyrophosphate dihydrate deposits in histologic examinations of total hip, knee, and shoulder joint arthroplasties. Arch Pathol Lab Med 2021;145:1558-63. [Google Scholar] [PubMed]
- 41. Shidham VB, Galindo LM, Gupta D, Jhala N, Shidham GB. Histology: Urate crystals in tissue: A novel staining method for formalin-fixed, paraffin-embedded sections. Lab Med 1998;29:109-13. [Google Scholar] [PubMed]
- 42. Shidham V, Shidham G. Staining method to demonstrate urate crystals in formalin-fixed, paraffin-embedded tissue sections. Arch Pathol Lab Med 2000;124:774-6. [Google Scholar] [PubMed]
- 43. McCarty DJ. Calcium pyrophosphate dihydrate crystal deposition disease-1975. Arthritis Rheum 1976;19:275-85. [Google Scholar] [PubMed]
- 44. Mccarty DJ Jr., Kohn NN, Faires JS. The significance of calcium phosphate crystals in the synovial fluid of arthritic patients: The “pseudogout syndrome” I. Clinical aspects. Ann Intern Med 1962;56:711-37. [Google Scholar] [PubMed]
- 45. Shetty NS, Pampana A, Patel N, Maurer MS, Goyal P, Li P, et al. Carpal tunnel syndrome and transthyretin amyloidosis in the all of us research program. Mayo Clin Proc 2024;99:1101-11. [Google Scholar] [PubMed]
- 46. Donnelly JP, Hanna M, Sperry BW, Seitz WH Jr. Carpal tunnel syndrome: A potential early, red-flag sign of amyloidosis. J Hand Surg Am 2019;44:868-76. [Google Scholar] [PubMed]
- 47. Zore Z, Filipović Zore I, Matejčić A, Kamal M, Arslani N, Knezović Zlatarić D. Surgical treatment of pathologic fractures in patients with metastatic tumors. Coll Antropol 2009;33:1383-6. [Google Scholar] [PubMed]
- 48. De Mattos CB, Binitie O, Dormans JP. Pathological fractures in children. Bone Joint Res 2012;1:272-80. [Google Scholar] [PubMed]
- 49. Snyder BD, Hauser-Kara DA, Hipp JA, Zurakowski D, Hecht AC, Gebhardt MC. Predicting fracture through benign skeletal lesions with quantitative computed tomography. J Bone Joint Surg Am 2006;88:55-70. [Google Scholar] [PubMed]
- 50. Dominic KP, Dijoe D, Manathara LT. Tumour like lesions and their management: A retrospective study. Int J Res Orthop 2017;4:159-65. [Google Scholar] [PubMed]