Megaprosthetic reconstruction in limb salvage surgery achieves meaningful functional recovery; however, complication rates remain substantial and are influenced by anatomical site, implant characteristics, and duration of follow-up.
Rishabh Guliani, Department of Orthopaedics, Dayanand Medical College and Hospital, Ludhiana, Punjab, India. E-mail: gulianirishab@gmail.com
Introduction: Limb salvage surgery (LSS) has replaced amputation in most cases of primary malignant bone tumors, achieving comparable oncologic control while preserving limb function and quality of life. Among reconstruction techniques, megaprosthetic replacement offers immediate stability, modular flexibility, and early rehabilitation potential. However, variability in tumor site, prosthesis design, and post-operative care continues to influence functional outcomes and complication rates across regions.
Materials and Methods: A comprehensive scoping review of studies published between 2014 and 2025 was conducted using PubMed, Scopus, Google Scholar, and EBSCO databases. Nineteen studies involving more than 600 patients who underwent LSS with megaprosthetic reconstruction for primary or recurrent bone tumors were analysed. Extracted data included patient demographics, tumor type, prosthesis characteristics, functional outcomes (musculoskeletal tumor society [MSTS], Toronto extremity salvage score [TESS], range of motion [ROM], gait parameters, Karnofsky Performance Status [KPS], return to work), and complications such as infection, mechanical failure, and aseptic loosening.
Results: Functional recovery was favourable, with mean MSTS and TESS scores ranging from 66% to 89%. Distal femur reconstructions demonstrated the highest functional outcomes (>80%), while proximal femur and humerus reconstructions showed slightly reduced scores (66–73%) due to soft-tissue limitations and reduced mobility. Improvements in ROM, gait symmetry, and KPS indicated successful restoration of biomechanics and independence. Complications included deep infections (5–24%), aseptic loosening (6–15%), and mechanical failures (up to 12.5%), most commonly in expandable implants. Despite these, limb retention exceeded 88% at 5 years, and long-term implant survival remained encouraging across different centers. Socioeconomic disparities and limited access to rehabilitation were identified as key factors affecting recovery and patient satisfaction.
Conclusion: Megaprosthetic reconstruction remains a cornerstone of modern LSS, providing durable oncologic safety and functional restoration. Early physiotherapy, infection prevention, and careful prosthesis selection are vital for optimising outcomes. Future studies should include standardized reporting of functional and psychosocial parameters and integrate socioeconomic factors to ensure equitable, patient-centred recovery worldwide.
Keywords: Bone tumour, endoprosthesis, functional outcome, limb salvage surgery, megaprosthesis, reconstruction.
Limb salvage surgery (LSS) has become the preferred treatment for primary malignant bone tumors of the extremities, replacing amputation in nearly 90–95% of eligible cases while achieving comparable oncologic control [1,2]. This approach emphasizes wide tumor excision, preservation of limb function, and improved quality of life for patients. The majority of these tumors occur in load-bearing bones, with the distal femur accounting for approximately 37% of cases, followed by the proximal tibia (19%), proximal humerus, and proximal femur [3]. The principal tumor types treated with limb salvage procedures include osteosarcoma, Ewing’s sarcoma, chondrosarcoma, and giant cell tumors. Advances in neoadjuvant chemotherapy, cross-sectional imaging, and reconstructive techniques have significantly improved the safety and functional success of limb-sparing surgery, leading to 5-year survival rates exceeding 60–70% in non-metastatic osteosarcoma patients [4].
Restoring limb biomechanics and function following tumor resection remains a key challenge. Various reconstructive strategies have been developed, including biological options such as allografts, autografts, arthrodesis, and rotationplasty. However, modular endoprosthetic reconstruction (megaprosthesis) has gained the widest acceptance due to its intraoperative adaptability, immediate structural stability, and facilitation of early mobilization [5,6]. These implants are particularly advantageous for metaphyseal and epiphyseal lesions where joint preservation is not feasible.
Functional outcomes following megaprosthetic reconstruction are generally favourable, with 70–85% of patients achieving good to excellent results. Functional recovery has been most commonly assessed using the musculoskeletal tumor society (MSTS) score and, in several recent studies, the Toronto extremity salvage score (TESS). Other complementary indicators – such as range of motion (ROM), gait analysis (e.g., 6 min walk test or gait deviation index [GDI]), pain reduction, Karnofsky Performance Status (KPS), and return to work capability have also been reported, providing a broader understanding of post-operative functional restoration. Despite these encouraging outcomes, residual limitations, including restricted joint motion, gait asymmetry, persistent pain, and reduced activity levels, may still impact overall quality of life.
Complications remain a major concern. Deep infections occur in approximately 10–20% of cases, while aseptic loosening, mechanical failures, and periprosthetic fractures continue to threaten long-term prosthesis survival and may necessitate revision surgery or, in severe instances, secondary amputation [7]. These complications are influenced by tumor site, prosthesis design, and soft-tissue reconstruction Quality.
Given the complexity of reconstruction and variability in outcomes across anatomical sites, understanding functional recovery patterns, complication profiles, and prosthesis longevity is crucial for optimizing patient outcomes. Therefore, this review aims to synthesize current evidence on the functional and non-functional outcomes of megaprosthetic reconstruction following LSS, with particular attention to site-specific challenges and complication rates.
This scoping review was conducted according to the Arksey and O’Malley scoping framework [8] and reported with reference to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 statement to enhance transparency in study identification and selection. The study selection process is illustrated in the PRISMA flow diagram (Fig. 1) [9].
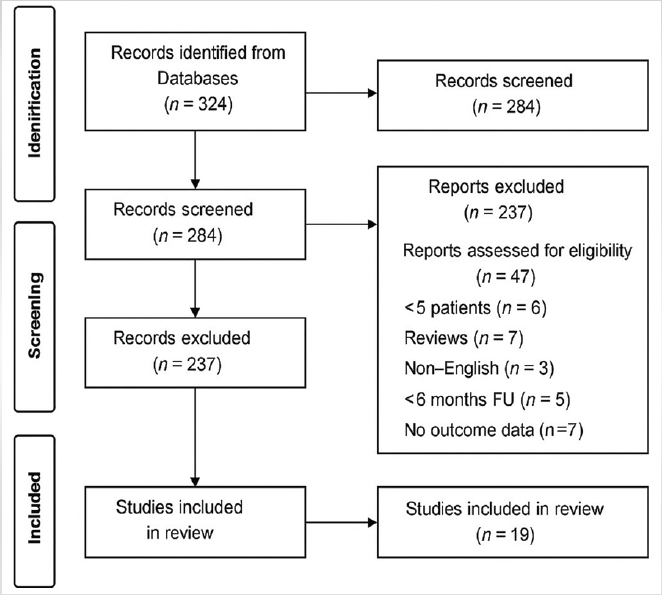
Figure 1: Preferred Reporting Items for Systematic Reviews and Meta-Analyses 2020 flow
diagram of study selection.
A comprehensive search was performed across PubMed, Scopus, Google Scholar, and EBSCO databases for studies published between January 2014 and May 2025, guided by the population, intervention, comparison, outcome framework (Table 1).
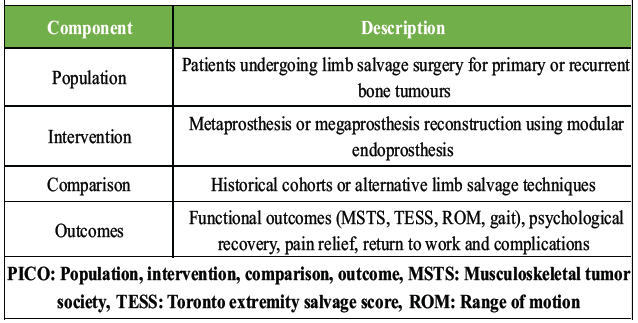
Table 1: The PICO framework guided search strategy and selection criteria
A structured combination of MeSH terms and keywords related to bone tumors, LSS, endoprosthetic or megaprosthetic reconstruction, and functional or complication outcomes was applied, with detailed search logic outlined in (Table 2).
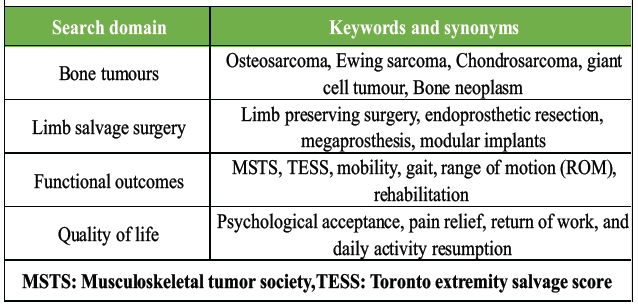
Table 2: Keywords
The initial search yielded 324 records, and after duplicate removal and screening, 47 full-text articles were assessed, of which 19 met the inclusion criteria. Eligible studies were English language publications (2014–2025), available in full text, focusing on LSS using modular or custom megaprosthetic reconstruction for primary or recurrent bone tumours, and reporting at least one functional outcome (e.g., MSTS, TESS, ROM, pain, or quality of life) and at least one complication outcome (infection, mechanical failure, aseptic loosening, or revision surgery). Excluded were studies with fewer than five patients, conference abstracts, reviews, non-English papers, non-extractable data, or follow-up shorter than 6 months.
Methodological quality and risk of bias of the included non-randomized observational studies were assessed using the Newcastle Ottawa scale (NOS) for cohort studies. The critical appraisal skills programme checklist [10] was used as a supportive framework to aid critical appraisal and interpretation of study findings. The assessment evaluated three key domains: Selection of study cohorts, comparability of cohorts based on study design or analysis, and adequacy of outcome assessment and follow-up. Risk of bias judgments for individual NOS domains were summarized and visualised using the risk of bias tool, employing a generic risk of bias framework. After the bias assessment, data were extracted using a structured template (Table 3) documenting study design, sample size, demographics, tumor type and site, prosthesis details, surgical margins, follow-up duration, functional outcomes, and complication profiles.
Due to heterogeneity in study design, prosthesis models, and outcome reporting, results were synthesised through a qualitative narrative approach rather than meta-analysis. The review further analysed patterns in functional recovery, complication rates, prosthesis survivorship, and regional practice trends, while identifying gaps in evidence concerning long-term implant performance, rehabilitation protocols, and design innovation to inform future research directions.Study scope and patient demographics
This review included 19 studies published between 2014 and 2025, encompassing a total of 610 patients who underwent LSS with megaprosthetic reconstruction for primary or recurrent bone tumors. Patient ages ranged from 13 to 47 years, with several studies focusing on pediatric and adolescent groups receiving expandable prostheses. Geographically, India contributed eight studies, followed by China (three), Egypt (two), France (two), and one each from Spain, Bulgaria, Denmark, and Pakistan. Study designs comprised prospective cohorts, retrospective series, cross-sectional, and case–control studies, with follow-up durations ranging from 6 months to more than 8 years.
Risk of bias assessment
Overall, the included studies demonstrated predominantly moderate risk of bias across NOS domains. Most studies showed low risk in selection representativeness and outcome assessment, reflecting well-defined cohorts and the use validated functional outcome measures. However, comparability between cohorts was frequently limited due to a retrospective study design and a lack of adjustment for confounding variables. In several studies, the absence of a clearly defined non-exposed comparator group contributed to higher risk ratings in selection-related domains. Follow-up duration and adequacy were generally moderate, with variability in reporting of loss to follow-up. A summary of the domain-specific risk of bias assessment is presented in Fig. 2.
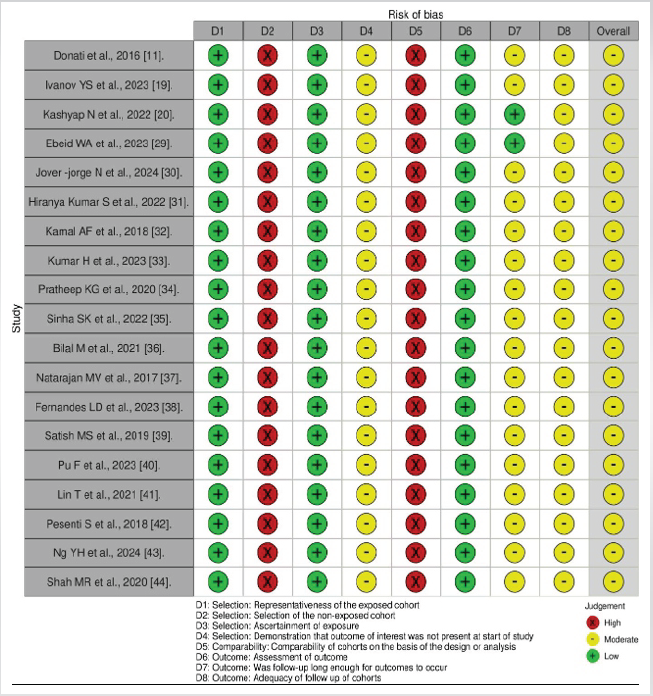
Figure 2: Risk of bias assessment of included non-randomized studies using the Newcastle Ottawa scale.
Surgical techniques and prosthesis types
Across the included literature, both modular and custom megaprostheses were used, with common systems including MUTARS, RESTOR, GMRS, and silver-coated or cemented variants. Modular implants predominated in India, China, and Egypt because of their cost-effectiveness, intraoperative adaptability, and ease of revision, whereas custom implants were favoured in European centers for precise anatomical reconstruction in complex resections. Silver-coated prostheses demonstrated lower infection rates (7.9% vs. 16.7%) and improved revision-free survival [11,12], while cemented fixation allowed earlier weight bearing and faster mobilization. Modular systems also facilitated simpler revision procedures in cases of loosening or mechanical failure [13,14,15].
Functional outcomes
Functional recovery was favourable, influenced by rehabilitation intensity, early mobilisation, prosthesis design, and anatomical site [16]. The MSTS score was the predominant outcome tool, while three studies also used the TESS. Additional parameters such as ROM, gait and biomechanical analysis (6-min Walk Test, Timed Up and Go, GDI), pain scores (Visual Analog Scale), KPS, and return to work or activities of daily living measures were variably reported.
Across all cohorts, MSTS/TESS scores ranged from 66.6% to 89.7%, indicating good to excellent limb function. The distal femur remained the most frequently reconstructed site and consistently yielded mean MSTS scores exceeding 80%, attributed to better biomechanics, soft-tissue coverage, and structured physiotherapy [17]. Proximal femur and proximal humerus reconstructions showed lower functional scores (66–73%), reflecting abductor weakness and limited joint mobility [18,19] (Fig. 3).
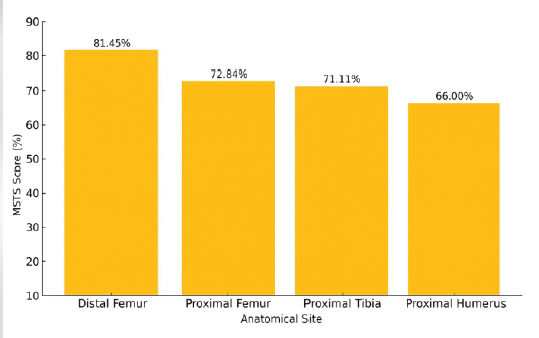
Figure 3: Mean musculoskeletal tumour society scores by anatomical site.
ROM data were reported in eight studies, with mean knee flexion ranging between 80° and 110° post-rehabilitation, extension lag of 5–20° in proximal tibial cases, and shoulder abduction between 70–90° after proximal humeral reconstruction. Biomechanical assessments were presented in three European studies, which demonstrated near-normal gait kinematics with GDI values of 86–89 compared to 94 in healthy controls and mildly prolonged 6MWT and TUG times. In Pu et al. (2023), KPS improved significantly from 50 preoperatively to 70–90 postoperatively, indicating restored independence. Early physiotherapy and weight bearing within 6–8 weeks were consistently associated with superior function, faster gait recovery, and higher satisfaction [20].
Non-functional outcomes
Non-functional results, including oncologic control, prosthesis longevity, and psychosocial recovery, were encouraging. Local recurrence ranged from 6.45% to 9.3% [13,14], while metastatic spread occurred in 7–18.2% of patients [17,18]. Five-year limb retention rates exceeded 88% in nearly all cohorts, with several reporting stable 10-year survivorship [11,12]. For example, Ebeid et al. (2023) documented 82.1% wide surgical margins and only 7% secondary amputations due to recurrence or severe complications. Distal femur reconstructions achieved the best local control [14], whereas proximal humerus and pelvic reconstructions carried slightly higher recurrence risks [19]. Psychosocial and return to work outcomes were addressed in five studies. Fernandes et al. (2023) reported 70% return to employment with a mild reduction in work capacity compared to controls, while Satish et al. (2019) and Jover-Jorge et al. (2024) observed better emotional well-being, self-image, and social reintegration among limb salvage patients compared to amputees. Ebeid et al. (2023) and Ivanov et al. (2023) similarly reported high patient satisfaction and improved quality of life following structured rehabilitation [21].
Complications and survivorship
Complications influenced long-term outcomes, with deep infection being the most common (5.1–23.6%), particularly in Egyptian and Chinese cohorts, despite the use of silver coatings [11,13]. Aseptic loosening occurred in 6.25–14.5% of patients, primarily in Indian and Pakistani studies [14,15]. Mechanical failures – including prosthetic breakage, hinge malfunction, periprosthetic fracture, and expansion mechanism defects – were observed in 6.5–12.5% of cases, with higher incidence among skeletally immature patients undergoing expandable reconstructions due to repeated lengthening and growth stresses [22]. A bar chart demonstrating pooled mean incidence of major complications following megaprosthesis reconstruction, including infection, aseptic loosening, mechanical failure, local recurrence, and metastasis (Fig. 4). Despite these complications, implant survival remained robust, with 5-year limb retention consistently above 88% and multiple studies reporting durable functionality beyond 10 years [11,12].
Recovery after LSS extends far beyond restoring bone and muscle – it represents the restoration of independence, identity, and quality of life. Across the 19 core studies (n > 600), patients frequently described regaining the ability to walk independently, return to work, and reclaim personal autonomy as defining milestones of recovery. Collectively, these outcomes reaffirm that megaprosthetic reconstruction is not merely a technical triumph but a means of restoring function and dignity. Long-term survivorship data have been equally encouraging, with several reports demonstrating that proximal femoral endoprostheses achieve more than 85% survival at 10 years, underscoring their durability in experienced oncologic centers [23].
Interpretation of these results, however, must be cautious given the predominance of retrospective designs, limited randomization, and heterogeneity in implant types and follow-up durations across studies. Functional outcomes, most commonly assessed through the MSTS and TESS, consistently demonstrated good to excellent recovery, with mean values ranging between 66% and 89%. These scores reflect an overall improvement in limb function, pain reduction, emotional acceptance, and independence in daily activities – highlighting that megaprosthetic reconstruction achieves both mechanical and functional recovery. Parallel gains in joint mobility and gait performance confirmed smoother biomechanical adaptation and efficient ambulation following structured rehabilitation. Improvements observed in performance indices such as the GDI and Timed Up and Go tests suggest that patients achieved near-normal walking symmetry and endurance. Similarly, higher KPS and increased return to work rates underscored the restoration of physical capacity, self-care ability, and social reintegration. Together, these multidimensional improvements confirm that LSS restores not only the integrity of the limb but also the patient’s overall quality of life and independence.
Hardes et al. demonstrated that silver-coated prostheses reduce infection risk by nearly half, offering a valuable preventive strategy against one of the most devastating complications [24]. Nevertheless, mechanical failures – including hinge fractures, component fatigue, aseptic loosening, and expansion mechanism defects remain significant challenges, particularly among children and adolescents treated with expandable implants, where repeated lengthening increases mechanical stress and revision rates [25]. Early initiation of rehabilitation and progressive weight bearing after reconstruction have been repeatedly shown to enhance functional recovery, accelerate ambulation independence, and improve MSTS and TESS scores among limb salvage patients [26].
Beyond surgical technique, social and economic contexts profoundly influence long-term outcomes. Gronchi et al. (2021) emphasised that optimal sarcoma management should be centralised in multidisciplinary reference centers where surgeons, oncologists, physiotherapists, and psychologists collaborate to optimise oncologic control, functional recovery, and emotional adaptation [27]. Duijts et al. (2014) further noted that even after successful physical recovery, many cancer survivors face ongoing psychosocial barriers, including anxiety and challenges returning to work or social activities [28]. In resource-limited settings, modular systems are often preferred due to their affordability and versatility; however, restricted access to structured rehabilitation and socioeconomic constraints can impede functional recovery and quality of life outcomes. Future studies should incorporate socioeconomic indicators such as income, education, and rehabilitation access to present a more comprehensive picture of recovery trajectories.
From an oncologic standpoint, achieving wide surgical margins remains the cornerstone of durable limb preservation. Ebeid et al. reported that 82% of patients with histologically clear margins retained their limbs at 5 years, reaffirming the delicate balance between oncologic safety and functional preservation [29]. By systematically searching four major databases, we were able to map both outcomes and complications rates over 600 patients worldwide. This provides a broad and realistic picture of how megaprosthetic reconstruction perform across different anatomical sites and healthcare settings. Because the included studies varied significantly in their study design, prosthetic models and follow-up durations, a statistical meta analysis was unable to be conducted. Furthermore, our review is limited by available literature as most of the underlying studies are retrospective and single-center series. Progress in this field now depends on methodological refinement. Moving forward, the establishment of multi-center registries that incorporate standardised definitions, validated patient-reported outcome measures (PROM’s), and socioeconomic variable is essential. This will enable better comparison and help guide global best practices.
This scoping review demonstrates that megaprosthetic LSS provides durable oncologic control and reliable functional restoration for patients with primary or recurrent bone tumours. Functional outcomes were consistently favourable, particularly in distal femur reconstructions, with strong long-term implant survival across studies. Although complications such as infection, aseptic loosening, and mechanical failure persist, modern modular and silver-coated prosthetic designs combined with structured rehabilitation have markedly improved long-term results. Future progress will depend on multi-center collaboration, standardized outcome measures, and the integration of socioeconomic and rehabilitation factors into future research, ensuring that advances in implant technology translate into equitable, patient-centered improvements in mobility and quality of life.
Megaprosthetic reconstruction remains a technical cornerstone of modern limb salvage surgery (LSS). While it offers immediate stability and early mobilization, the balance between functional gain and long term complications requires careful patient selection and site specific expectations.
References
- 1. Simon MA, Aschliman MA, Thomas N, Mankin HJ. Limb-salvage treatment versus amputation for osteosarcoma of the distal end of the femur. J Bone Joint Surg Am 1986;68:1331-7. [Google Scholar] [PubMed]
- 2. Rougraff BT, Simon MA, Kneisl JS, Greenberg DB, Mankin HJ. Limb salvage compared with amputation for osteosarcoma of the distal end of the femur. A long-term oncological, functional, and quality-of-life study. J Bone Joint Surg Am 1994;76:649-56. [Google Scholar] [PubMed]
- 3. Dharanikota A, Arjunan R, Dasappa A. Factors affecting prognosis and survival in extremity osteosarcoma. Indian J Surg Oncol 2021;12:199-206. [Google Scholar] [PubMed]
- 4. Bacci G, Mercuri M, Longhi A. Neoadjuvant chemotherapy for osteosarcoma of the extremity. J Clin Oncol 2005;23:8845-52. [Google Scholar] [PubMed]
- 5. Grimer RJ, Aydiner B, Wafa H, Carter SR, Jeys L, Abudu A, et al. Very long-term outcomes after limb salvage surgery with end oprosthesis for bone tumors. Clin Orthop Relat Res 2016;474:706-15. [Google Scholar] [PubMed]
- 6. Jeys LM, Kulkarni A, Grimer RJ, Carter SR, Tillman RM, Abudu A. Endoprosthetic reconstruction for the treatment of musculoskeletal tumors of the appendicular skeleton and pelvis. J Bone Joint Surg Am 2008;90:1265-71. [Google Scholar] [PubMed]
- 7. Henderson ER, Groundland JS, Pala E, et al. Failure mode classification for tumor endoprostheses. Clin Orthop Relat Res 2011;469:1704-3. [Google Scholar] [PubMed]
- 8. Arksey H, O’Malley L. Scoping studies: Towards a methodological framework. Int J Soc Res Methodol 2005;8:19-32. [Google Scholar] [PubMed]
- 9. Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021;372:n71. [Google Scholar] [PubMed]
- 10. Critical Appraisal Skills Programme (CASP). CASP Checklists. Available from: https://casp-uk.net/casp-tools-checklists [Last accessed on 2025 May ???]. [Google Scholar] [PubMed]
- 11. Donati F, Di Giacomo G, D’Adamio S, Ziranu A, Careri S, Rosa MA, et al. Silver-coated hip megaprosthesis in oncological limb savage surgery. Biomed Res Int 2016;2016:9079041. [Google Scholar] [PubMed]
- 12. Zajonz D, Birke U, Ghanem M, Prietzel T, Josten C, Roth A, et al. Silver-coated modular megaendoprostheses in salvage revision arthroplasty after periimplant infection with extensive bone loss – a pilot study of 34 patients. BMC Musculoskelet Disord 2017;18:383. [Google Scholar] [PubMed]
- 13. Ahlmann ER, Menendez LR, Kermani C, Gotha H. Survivorship and clinical outcome of modular endoprosthetic reconstruction for neoplastic disease of the lower limb. J Bone Joint Surg Br 2006;88:790-5. [Google Scholar] [PubMed]
- 14. Shehadeh A, Noveau J, Malawer M, Henshaw R. Early complications and outcomes of cemented endoprosthetic reconstruction after resection of primary bone tumors. Ann Surg Oncol 2010;17:524-35. [Google Scholar] [PubMed]
- 15. Pala E, Trovarelli G, Calabrò T, Angelini A, Abati CN, Ruggieri P. Survival of modern knee tumor megaprostheses: Failures, functional results, and a comparative statistical analysis. Clin Orthop Relat Res 2015;473:891-9. [Google Scholar] [PubMed]
- 16. Batta V, Coathup MJ, Parratt MT, et al. Effect of rehabilitation and patient factors on functional outcome after endoprosthetic replacement for bone tumors of the lower limb. J Bone Joint Surg Br 2010;92:1152-8. [Google Scholar] [PubMed]
- 17. Puri A, Gulia A, Agarwal MG, Reddy K, Patil V. Functional outcome of limb salvage surgery with megaprosthesis for primary bone tumours of the lower limb. Indian J Orthop 2012;46:470-6. [Google Scholar] [PubMed]
- 18. De Gori M, D’Arienzo A, Andreani L, Beltrami G, Campanacci DA, De Biase P, et al. Complications and survival of megaprostheses after resection of bone metastases. J Biol Regul Homeost Agents 2017;31:43-50. [Google Scholar] [PubMed]
- 19. Ivanov YS, Tsenkov T. Functional results after reconstruction with modular tumor endoprostheses in patients with malignant bone tumors. Folia Med (Plovdiv) 2023;65:80-6. [Google Scholar] [PubMed]
- 20. Kashyap N, Runu R, Ahmed W, Kumar I, Subash A. A retrospective cohort analysis of limb salvage surgery using mega prosthesis in bone tumours at a tertiary care centre in Eastern India. Cureus 2022;14:e28959. [Google Scholar] [PubMed]
- 21. Aksnes LH, Bauer HC, Jebsen NL, Folleras G, Allert C, Haugen GS, et al. Limb-sparing surgery preserves more function and quality of life than amputation: A long-term follow-up of extremity sarcoma patients. Acta Orthop 2008;79:106-12. [Google Scholar] [PubMed]
- 22. Öztürk R. Expandable endoprostheses in skeletally immature patients: Where we are. World J Orthop 2024;15:312-7. [Google Scholar] [PubMed]
- 23. Wedin R, Bauer HC. Surgical treatment of skeletal metastatic lesions of the proximal femur: Endoprosthesis or reconstruction nail? J Bone Joint Surg Br 2005;87:1653-7. [Google Scholar] [PubMed]
- 24. Hardes J, Von Eiff C, Streitbuerger A, Balke M, Budny T, Henrichs MP, et al. Reduction of periprosthetic infection with silver-coated megaprostheses in patients with bone sarcoma. J Surg Oncol 2010;101:389-95. [Google Scholar] [PubMed]
- 25. Dotan A, Dadia S, Bickels J, Nirkin A, Flusser G, Issakov J, et al. Expandable endoprosthesis for limb-sparing surgery in children: Long-term results. J Child Orthop 2010;4:391-400. [Google Scholar] [PubMed]
- 26. Shehadeh A, El Dahleh M, Salem A, Sarhan Y, Sultan I, Henshaw RM, et al. Standardization of rehabilitation after limb salvage surgery for sarcomas improves patients’ outcome. Hematol Oncol Stem Cell Ther 2013;6:105-11. [Google Scholar] [PubMed]
- 27. Gronchi A, Miah AB, Dei Tos AP, Abecassis N, Bajpai J, Bauer S, et al. Soft tissue and visceral sarcomas: ESMO-EURACAN-GENTURIS clinical practice guidelines for diagnosis, treatment and follow-up(☆). Ann Oncol 2021;32:1348-65. [Google Scholar] [PubMed]
- 28. Duijts SF, Van Egmond MP, Spelten E, Van Muijen P, Anema JR, Van Der Beek AJ. Physical and psychosocial problems in cancer survivors beyond return to work: A systematic review. Psychooncology 2014;23:481-92. [Google Scholar] [PubMed]
- 29. Ebeid WA, Hassan MH. Functional outcome following proximal tibial osteosarcoma resection and reconstruction by modular endoprosthesis. Ann Surg Oncol 2023;30:1914-25. [Google Scholar] [PubMed]
- 30. Jover-Jorge N, González-Rojo P, Amaya-Valero JV, Baixuali-García F, Calva-Ceinós C, Angulo-Sánchez MÁ, et al. Evaluating functional outcomes and quality of life in musculoskeletal tumor patients with distal femoral megaprostheses: A case-control study. World J Surg Oncol 2024;22:341. [Google Scholar] [PubMed]
- 31. Kumar H, Mittemari KN, Nimmagadda VC, Abraham AT, Nadagouda S, Choudry D. The functional outcome of limb salvage of proximal femur tumors with modular endoprosthesis: A prospective study. Cureus 2023;15:e50375. [Google Scholar] [PubMed]
- 32. Kamal AF, Pitarini A, Prabowo Y. Megaprosthesis limb salvage surgery: Outcome and challenges in treating advanced bone tumour cases in vast archipelago in Indonesia. A case series. Int J Surg Open 2018;11:30-6. [Google Scholar] [PubMed]
- 33. Pratheep KG, Karthikeyan S, Pandiaselvan A. Analysis of the functional outcome of limb salvage procedure using custom-made endoprosthesis for bone tumours: A case series. Int J Sci Res 2020;9:144-7. [Google Scholar] [PubMed]
- 34. Sinha SK, Das A, Das A, Ghosh S. Limb salvage surgery with endoprosthesis reconstruction in management of locally advanced primary bone tumours: A functional outcome evaluation. Int J Res Orthop 2022;8:214-21. [Google Scholar] [PubMed]
- 35. Bilal M, Jilani S, Rafi I, Shakeel O, Jabeen W. Early outcomes of limb salvage surgery with mega-prosthesis: A single center experience. J Acute Dis 2021;10:208-15. [Google Scholar] [PubMed]
- 36. Natarajan MV, Sameer MM, Upadhyay U, Kumar MD. Custom mega prosthesis in metachronous osteosarcoma. Ann Natl Acad Med Sci 2020;53:73-8. [Google Scholar] [PubMed]
- 37. Fernandes LD, Villadsen A, Holm CE, Sørensen MS, Zebis MK, Andersen LL, et al. Work ability and physical activity after major bone sarcoma resection and reconstruction with tumour prosthesis of the lower extremities: A cross-sectional study. Disabil Rehabil 2023;45:2597-603. [Google Scholar] [PubMed]
- 38. Satish MS, Vijay S, Raja A, Veeraiah S. Quality of life and functional outcome of patients treated with revision limb salvage surgery and amputation: A cross-sectional study. J Bone Soft Tissue Tumors 2019;5:11-4. [Google Scholar] [PubMed]
- 39. Pu F, Yu Y, Shao Z, Wu W, Feng J, Chen F, et al. Clinical efficacy of customized modular prosthesis in the treatment of femoral shaft metastases. Front Oncol 2023;13:1115898. [Google Scholar] [PubMed]
- 40. Lin T, Jin Q, Mo X, Zhao Z, Xie X, Zou C, et al. Experience with periprosthetic infection after limb salvage surgery for patients with osteosarcoma. J Orthop Surg Res 2021;16:93. [Google Scholar] [PubMed]
- 41. Pesenti S, Peltier E, Pomero V, Authier G, Roscigni L, Viehweger E, et al. Knee function after limb salvage surgery for malignant bone tumor: Comparison of megaprosthesis and distal femur allograft with epiphysis sparing. Int Orthop 2018;42:427-36. [Google Scholar] [PubMed]
- 42. Ng YH, Chai YC, Mazli N, Jaafar NF, Ibrahim S. Outcome of endoprosthesis used in limb salvage surgery in a Malaysian orthopaedic oncology centre. Malays Orthop J 2024;18:60-5. [Google Scholar] [PubMed]
- 43. Shah MR, Shah MM, Agrawal AK, Shah MD, Desai SM. Functional outcome of limb salvage surgery by mega-prosthesis for malignant bone tumors-mid and long-term follow-up. Int J Curr Res Rev 2020;12:189-94. [Google Scholar] [PubMed]