Early split-thickness skin grafting, when performed after confirmed source control, can provide durable soft-tissue coverage with excellent functional and esthetic outcomes in upper extremity necrotizing fasciitis, even in anatomically and cosmetically sensitive regions such as the dorsum of the hand.
Dr. Arpam Dutta, Mercy Health St. Vincent Medical Center, Toledo, Ohio. E-mail: arpamd24@gmail.com
Introduction: Necrotizing fasciitis (NF) is a rare, rapidly progressive soft-tissue infection requiring emergent surgical intervention. Upper extremity involvement poses unique reconstructive challenges due to a thin, soft-tissue coverage and risk of tendon exposure.
Case Report: We present the case of a 68-year-old female with NF of the left forearm and hand, treated with three debridements followed by split-thickness skin grafting on hospital day 6. A combination of meshed and unmeshed grafts was used, with the unmeshed graft placed over the dorsum of the hand for improved cosmesis. The patient demonstrated excellent graft uptake and preserved hand function at long-term follow-up. This case highlights the feasibility of early grafting in sensitive areas with NF when adequate source control is achieved.
Conclusion: Early skin grafting, when carefully timed, can result in favorable functional and esthetic outcomes in complex upper extremity infections.
Keywords: Necrotizing fasciitis, forearm infection, dorsum of the hand infection, split-thickness skin graft.
Necrotizing fasciitis (NF), also known as “flesh-eating disease,” is a severe, rapidly progressing skin and soft-tissue infection that leads to necrosis of the muscle fascia and subcutaneous tissue [1]. It is considered a rare condition, with an incidence ranging from 0.3 to 5 cases/100,000. Common risk factors include IV drug use, immunosuppression, and diabetes mellitus [2]. The most common pathogens leading to single-site infections include Gram-positive organisms (Staphylococcus aureus and Streptococci). Given its aggressive nature, NF requires emergent intervention such as antibiotic coverage, fluids, surgical debridement, and grafting [1]. NF of the upper extremity poses unique reconstructive challenges due to a thin, soft-tissue envelope, risk of tendon exposure, and poor vascularity, especially in the dorsum of the hand [3,4]. Split-thickness grafts (STSG) are frequently used in this situation. Graft timing remains a difficult clinical decision requiring careful consideration. Grafting over an area with residual bacteria can lead to further progression, making adequate source control a vital consideration [5]. We present a case of NF in a 68-year-old female who underwent four surgeries, including irrigation and debridement, as well as a combination of meshed and unmeshed STSG of the left forearm and dorsum of the hand.
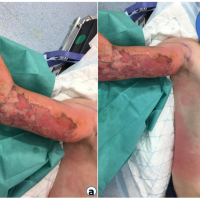
A 68-year-old female presented to our emergency department on May 02, 2023, complaining of left forearm pain secondary to a large dorsal wound. She had a minor surgical history with a past medical history of autoimmune diseases and morbid obesity. Three days prior, she fell and landed on her left forearm, sustaining a soft-tissue injury. She reported that she kept this wrapped for several days. She noted purulence when the wound opened the day prior. On examination, there was a large dorsal wound with substantial purulence and foul-smelling discharge. X-rays and computed tomography showed severe soft-tissue edema with ulcers and subcutaneous gas at the dorsum of the forearm and hand. Her laboratory tests revealed a white blood cell count of 11.2 × 103/µL (reference range, 4.00 × 103/µL–11.00 × 103/µL), an elevated absolute neutrophil count of 9.85 × 103/µL (reference range, 1.8 × 103 /µL–8.00 × 103/µL), an elevated C-reactive protein level of 315.2 mg/dL (reference range, <0.5 mg/dL), and an elevated erythrocyte sedimentation rate of 437 mm/h (reference range, 0–30 mm/h). The LRINEC (Laboratory Risk Indicator Score for Necrotizing Soft-Tissue Infection) score at time of admission was 9 (hemoglobin, 12.8; sodium, 128; glucose, 183; and creatinine, 1.7). Cultures taken at this time grew Streptococcus pyogenes (Fig 1).
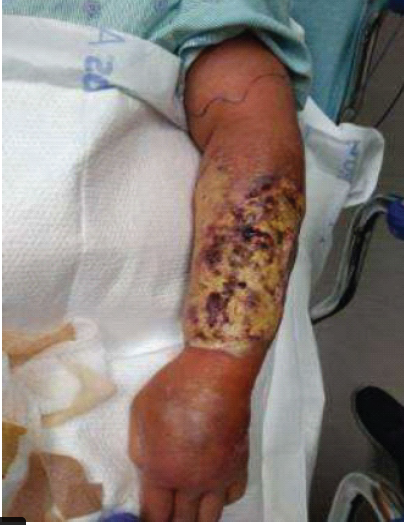
Figure 1: Patient’s initial injury presentation to the emergency department.
On physical examination, there was significant purulence, which prompted us to take this patient emergently to the operating room (OR). Upon excising the wound, we noted purulence and gray-colored fluid, similar to dishwater pus and consistent with NF. The forearm was debrided circumferentially to clean margins. (Fig. 2).
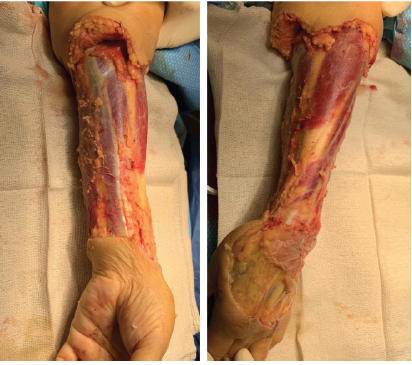
Figure 2: Intraoperative Findings During Initial Surgical Debridement for Necrotizing Fasciitis.
The infection tracked proximally to the elbow, which was not evident during initial evaluation, prompting more proximal incisional debridement. After thorough excision, viable tissue was observed, and wound care was halted at the elbow. The patient was placed on broad-spectrum antibiotics, including vancomycin, zosyn, and clindamycin. On hospital day-1, she returned to the OR. The upper arm appeared viable without evident signs of infected tissue progression. The remaining necrotic tissue on the forearm and hand was sharply debrided. On hospital day 3, the patient’s arm, forearm, and hand continued to demonstrate viable tissue. This prompted closure of the proximal incisions with plans to skin graft the hand and forearm in the coming days. On hospital day-5, she received an STSG, harvested from the anterior-lateral bilateral thighs at a 10/1000 of an inch thickness and meshed in a 1:1.5 ratio. There was a significant step-off at the proximal aspect of the wound, given the patient’s body habitus and complete excision of the subcutaneous tissues of the forearm. Utilizing 2-0 Monocryl, we elected to pull the intact skin proximally down to the level of the excised tissue, allowing for a more cosmetic transition to her graft. A 150 cm2 portion of the graft was left unmeshed for placement on the dorsal aspect of the hand for improved cosmesis. The patient was discharged on hospital day 14 with plans to follow-up in the clinic. Upon her 6-week follow-up, the graft bed showed excellent integration. Other than over the grafted skin, the patient was neurovascularly intact, able to form a fist fully, and had a full range of motion at the elbow and wrist without pain (Fig 2). During her last outpatient follow-up at the 22-month mark on November 12, 2024, the patient noted minor residual numbness and occasional burning sensation to the grafted areas of the left forearm. The patient was able to make a full composite fist with full finger abduction and adduction, lacking only slight terminal flexion of the ring finger (Fig. 3 and 4). The elbow range of motion was 0–125°. No limitations to pronation or supination were observed (Fig 5).(Table 1)
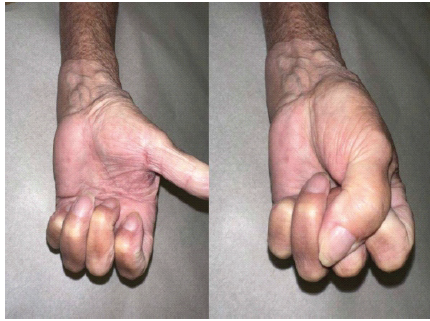
Figure 3: Hand motion at 22-month follow-up.
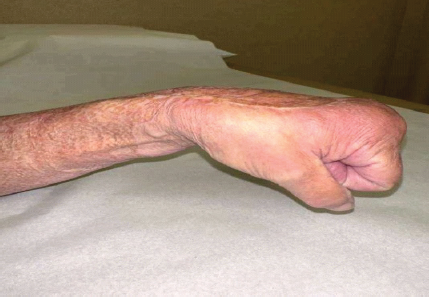
Figure 4: Wrist motion at 22-month follow-up.
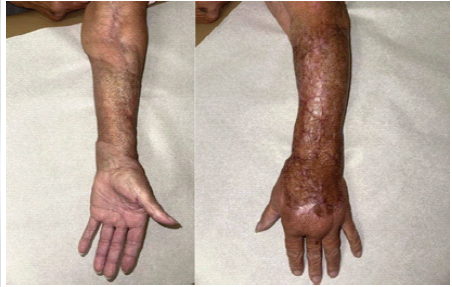
Figure 5:Supination and pronation at 22-month follow-up.

Table 1: Range of motion 22 months after initial debridement
NF is a true surgical emergency, with isolated extremity cases reporting mortality rates >20% [6]. The rapid and destructive nature of this disease makes early recognition, aggressive debridement, and antibiotic therapy necessary to minimize morbidity. Early diagnosis and prompt action, therefore, remain key to successful treatment and ultimate survival of these patients. Early source control is imperative to successful treatment, with patients often requiring an average of 3 surgical debridement operations [6]. In our case, the patient required 3 debridement procedures before the final skin grafting was applied. If multiple debridement procedures fail to control the infection, amputation of the affected extremity is required. Ultimate soft tissue coverage is unique to each case and presenting circumstances. In a systematic review of NF of the extremities, Angoules et al. found that definitive soft tissue coverage required split thickness skin grafting, rotational flap, amputation, or limb disarticulation in 48.4%, 4.5%, 20.9%, and 1.4% of cases, respectively [6]. The amount of debridement and exposed tissues will dictate which treatments are most appropriate for each scenario. In our patient, given the amount of soft tissue exposed due to the need for extensive debridement, we elected for coverage with STSG. Our patient’s infection with S. pyogenes required debridement of the dorsum of the hand. The dorsal surface of the hands has a thin, soft tissue layer under which the extensor tendons directly lie. Extensor tendons of the hand are, therefore, uniquely at risk for exposure when skin defects occur over the dorsum of the hand. The failure rate of STSG is reported to be very high when performed on tendon-exposed wound beds because tendons are fibrous connective tissues with a relatively poor blood supply [3,4]. Fortunately, our case involved retainment of most of the fascia overlying the tendons of the hand, which allowed for the application of the skin graft. Most of the forearm contained only exposed muscle and was deemed appropriate for skin grafting. If tendons are exposed, techniques have been proposed to graft over the tendons [7]. Others have also advocated for the use of products such as collagen-based or polyglactin synthetic substitutes to help facilitate the growth of a granulation layer and wound bed appropriate for skin grafting [8]. We elected to use an unmeshed graft for the dorsum of the hand. As the dorsum of the hand is more difficult to cover with clothing, this lends the dorsum of the hand to be more readily visible. We therefore sought the most esthetically pleasing option for this smaller, more sensitive area. In a systematic review by Samuel et al. comparing meshed versus unmeshed STSGs, it was found that unmeshed grafts offer a more esthetically pleasing appearance, due to a more uniform skin surface and greater resilience to movement [9]. It was felt that by applying an unmeshed graft to the dorsum of the hand, this would create a more continuous, smoother skin surface with a more uniform texture for this more sensitive location than a meshed graft would. Of note, however, unmeshed grafts do carry an increased risk of fluid collection due to being unperforated as well as requiring a larger donor site [9]. A knowledge of the advantages and disadvantages regarding these grafts is essential, and clinical discretion should be used when applying unmeshed grafts. Timing of skin grafting proves a difficult clinical decision with careful consideration. We elected to take our patient back in 6 days for definitive skin grafting. It was felt that after repeat debridement and wound checks, the necrotic/infected tissue was eliminated, and a healthy wound bed remained. Due to the risk of remnant bacteria causing poor wound healing, it is not recommended to close these wounds early. Historically, the primary method for management included serial debridement and dressing changes, allowing for slow granulation with eventual muscle and skin grafting once the infection has cleared [5,10].
Achieving source control while preserving limb function remains challenging. In our case, we felt that adequate control was achieved by day-6 and chose to skin graft at that time. Therefore, the use of early STSG, specifically unmeshed grafting for cosmetically sensitive areas, can provide acceptable esthetic outcomes, with acceptable preservation of limb function.
Upper extremity NF poses a reconstructive challenge due to limited soft tissue coverage and high risk of tendon exposure. This case illustrates that definitive skin grafting as early as hospital day-6, following serial debridement and careful assessment of wound viability, can result in durable healing and sustained hand function, offering a practical framework for timing reconstruction in complex forearm and hand infections.
References
- 1. Wallace HA, Perera TB. Necrotizing fasciitis. In: StatPearls. Treasure Island, FL: StatPearls Publishing; 2023. [Google Scholar] [PubMed]
- 2. Fernando SM, Tran A, Cheng W, Rochwerg B, Kyeremanteng K, Seely AJ, et al. Necrotizing soft tissue infection: Diagnostic accuracy of physical examination, imaging, and LRINEC score: A systematic review and meta-analysis. Ann Surg 2019;269:58-65. [Google Scholar] [PubMed]
- 3. Takasugi H, Akahori O, Nishihara K, Tada K. Three-dimensional architecture of blood vessels of tendons demonstrated by corrosion casts. Hand 1978;10:9-15. [Google Scholar] [PubMed]
- 4. Schmidt-Rohlfing B, Graf J, Schneider U, Niethard FU. The blood supply of the Achilles tendon. Int Orthop 1992;16:29-31. [Google Scholar] [PubMed]
- 5. Salcido RS. Necrotizing fasciitis: Reviewing the causes and treatment strategies. Adv Skin Wound Care 2007;20:288-93; quiz 294-5. [Google Scholar] [PubMed]
- 6. Angoules AG, Kontakis G, Drakoulakis E, Vrentzos G, Granick MS, Giannoudis PV. Necrotising fasciitis of upper and lower limb: A systematic review. Injury 2007;38:S19-26. [Google Scholar] [PubMed]
- 7. Um JH, Jo DI, Kim SH. New proposal for skin grafts on tendon-exposed wounds. Arch Plast Surg 2022;49:86-90. [Google Scholar] [PubMed]
- 8. Wollina U, Meseg A, Weber A. Use of a collagen-elastin matrix for hard to treat soft tissue defects. Int Wound J 2011;8:291-6. [Google Scholar] [PubMed]
- 9. Samuel J, Gharde P, Surya D, Durge S, Gopalan V. A comparative review of meshed versus unmeshed grafts in split-thickness skin grafting: Clinical implications and outcomes. Cureus 2024;16:e69606. [Google Scholar] [PubMed]
- 10. Cainzos M, Gonzalez-Rodriguez FJ. Necrotizing soft tissue infections. Curr Opin Crit Care 2007;13:433-9. [Google Scholar] [PubMed]