Innovative, minimally invasive method of internal fixation in a tumor cavity filled with sandwich method.
Dr. Rachit Sekhrajka, Department of Orthopaedics, Seth G.S. Medical College and King Edward VII Memorial Hospital, Mumbai, Maharashtra, India. E-mail: rsekhrajka@gmail.com
Introduction: Giant cell tumor (GCT) of bone is a benign yet locally aggressive tumor that predominantly affects the epiphyseal/metaphyseal regions of long bones. While GCTs frequently occur around the knee or sacrum, their presentation in the proximal humerus is rare. Management of Campanacci Grade 3 GCTs poses challenges in achieving tumor control while preserving joint function. This case highlights a successful, minimally invasive approach to treating a proximal humerus GCT in an immunocompromised young patient.
Case Report: A 30-year-old male presented with pain and restricted right shoulder movement following trivial trauma. Examination revealed bony-hard swelling and a restricted range of motion without neurovascular deficits. Imaging showed an expansile, lytic lesion in the proximal humerus with a cortical breach (Campanacci grade 3). Histopathological evaluation confirmed the diagnosis of GCT. The patient underwent intralesional curettage, reconstruction using a sandwich technique with sub-chondral bone graft, interdigitating cancellous screws in a cross-beam construct, and bone cement for filling the cavity. Adjuvants, including hydrogen peroxide and high-speed burring, were used to achieve negative margins and further reduce the risk of recurrence, given that intra-lesional curettage was used ahead of wide resection. The cross-beam construct of screws within the cavity not helped decrease surgical time and morbidity but provided a similar level of stability and decreased the risk of post-operative fractures. Postoperatively, the patient was immobilized for 6 weeks, followed by progressive mobilization. At 1.5 years follow-up, imaging showed no recurrence, and the patient achieved a painless, acceptable range of motion.
Conclusion: This case underscores the potential of joint-preserving surgery for Campanacci grade 3 GCTs, particularly in younger patients. The combination of intralesional curettage, sandwich reconstruction, and interdigitating screws provided stable fixation with minimal morbidity. This approach preserved shoulder function, avoided the need for arthroplasty, and minimized surgical complications, even in a patient with compromised immunity. The findings support the use of innovative reconstruction techniques for aggressive GCTs, offering improved functional outcomes and reduced recurrence risk.
Keywords: Proximal humerus, giant cell tumor, reconstruction, screw interdigitation method, minimally invasive.
Giant cell tumor (GCT), also known as osteoclastoma, is one of the most common bone tumors. GCT of bone is usually solitary and benign but locally aggressive, with 5% risk of metastasis and 1–3% incidence of malignant transformation [1]. It has a predilection for the epiphyseal/metaphyseal region of the long bones [2] and commonly affects people in third and fourth decades of their life, with a higher incidence in females. The most common sites for this tumor include bones around the knee, distal radius, and sacrum [3]. Proximal humerus is a relatively rare location. The recurrence rates calculated postoperatively regardless of the surgical procedure performed is as high as 50% [4]. Campanacci classified giant cells tumors based on their radiological appearance into three types:
- One with an intact cortex, second with cortical expansion, and third with a cortical breach. Campanacci classification guided treatment options stating that Type 3 requires wide resection and reconstruction as compared to intralesional curettage for the former 2 types [5].
We present a case of a 30-year-old male who presented to the hospital with complaints of pain and restricted range of movement in the right shoulder since 3 months following a trivial trauma. Examination revealed bony-hard swelling, tenderness over the lateral aspect of shoulder, no signs of inflammation with restricted shoulder range of motion. No distal neurovascular deficit was found. Initial radiograph at local hospital revealed an incidental finding of an expansile, lytic lesion in epiphyseo-metaphyseal region of the right proximal humerus. Further radiological investigation in the form of magnetic resonance imaging revealed a Campanacci Grade 3 osteoclastoma with an evident cortical breach (Fig. 1). Patient was planned for a biopsy of the lesion, and histopathologic investigation confirmed the diagnosis. Blood investigations revealed the patient was seropositive for Hepatitis B, but were otherwise non-contributory.
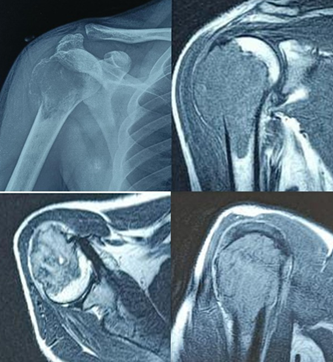
Figure 1: Pre-operative scan showing an expansile lytic lesion with a characteristic soap bubble appearance, with a cortical breach and tumor extending into the deltoid muscle.
Patient was then posted for a planned, methodical extended curettage of the proximal humerus GCT along with reconstruction using the bone graft, bone cement, and interdigitating screws. In beach-chair position, using a deltoid splitting approach with the incision being inclusive of the previous biopsy site (Fig. 2).
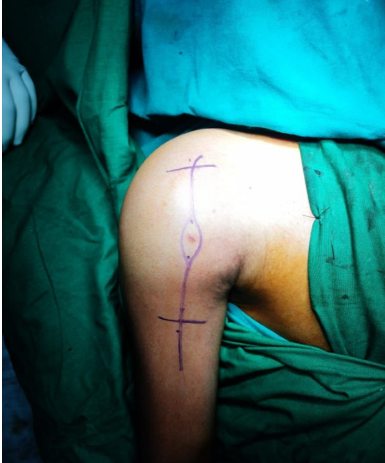
Figure 2: Deltoid splitting approach with incision including the previous biopsy site.
Soft-tissue dissection included carefully incising the sinus tract created during the biopsy through its entire length. The surgical field was circumferentially lined with hydrogen-peroxide soaked mops as a form of chemical cauterization, and two separate trolleys were maintained – one for instruments contaminated with tumor material and the other for sterile instruments. After adequate exposure, tumor material was scooped out with a curette, with the tumor cells carefully removed using a high- speed burr to achieve negative margins. Utmost care was taken to not breach the anterior or medial wall of the proximal humerus while using the burr under fluoroscopic guidance. A final hydrogen-peroxide wash was given. Reconstruction of the void started with a basal layer of tri-cortical iliac crest bone graft. Under fluoroscopic guidance, 4 fully-threaded cancellous screws were then inserted in a pre-determined sequence of different orthogonal plains to create a scaffold (2 in rostro-caudal manner, 1 antero-posterior, and 1 transverse) (Fig.3) .
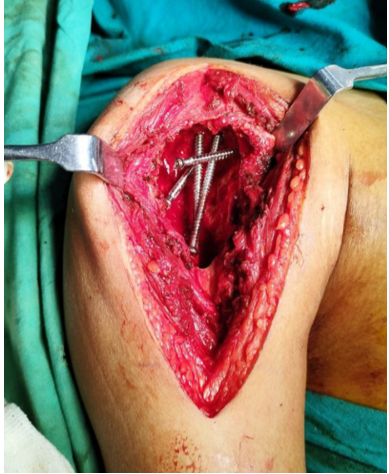
Figure 3: Cancellous screw scaffold created in the tumor void.
Low-viscosity bone cement was prepared and poured over the scaffold (Fig.4).
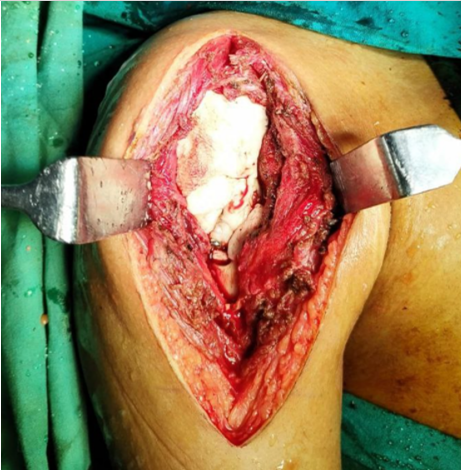
Figure 4: Tumor void filled with allograft, screw scaffold, and bone cement.
Besides it is pleiotropic tumoricidal effect, low viscosity has a working advantage of it increasing in size being inserted and easier to handle as it flows easily and has more working time before it solidifies. Intra-op range was checked, and the construct was found to be stable. Closure was achieved, and post-operative patient was kept in a universal shoulder immobilizer for 6 weeks. Mobilization was started with pendulum exercises, followed by the shoulder range of motion as tolerated by the patient. 1.5-year follow-up imaging reveals no obvious tumor recurrence. Range of motion is of acceptable range and painless.
The treatment modalities for management of GCT’s include chemotherapy, radiotherapy, embolization, cryosurgery, and surgery. Among all treatment methods, surgery remains the only effective treatment for GCT, in most cases. The operative method is determined by the surgical stage of the giant-cell tumor, its size and location, age of the patient, and whether it is a primary tumor or a recurrence [6]. Nonetheless, the ideal form of surgery for this tumor, particularly when arising near joints or when present in weight-bearing bones, remains controversial [7], especially in Campanacci Grade III giant cell tumors, as their massive sizes provide an uphill task in reconstruction and demands case-specific planning from the surgeons. Surgical options include intralesional curettage with bone grafting and/or cement or segmental resection and reconstruction with allograft or prosthesis. The goal of surgery in current times is not only to prevent recurrence by completely removing the tumor but also to maintain adjacent joint function. The evaluation of function, described by Musculoskeletal Tumor Society score (MSTS) adequately gauges the patient’s acceptability of a given procedure [8]. While wide resection and reconstruction minimize tumor recurrence, it fails to provide acceptable functional outcomes, especially in the shoulder joint after treatment of proximal humerus lesions. Intralesional curettage has a higher reported recurrence rate but performs much better on the acceptability scale. Therefore, joint-sparing surgery and careful curettage with the use of adjuvants should be the treatment of choice whenever possible [9]. Grade III osteoclastoma can be treated by intralesional excision, provided the articular surfaces and part of the metaphysis are intact [8]. In younger patients, intra-lesional curettage and reconstruction provide optimum outcomes with minimal chances of recurrence and higher MSTS scores [10]. This has been a major shift in management as high-grade GCT’s were conventionally treated with wide resection due to fear of recurrence [11]. This shift can be attributed to adoption of combined cauterization methods – mechanical (high speed burr), chemical (use of adjuvants like phenol, Hydrogen peroxide, ethanol, and zinc chloride) [12], and thermal (use of bone cement in reconstruction of the cavity) [13], to attenuate the risk of recurrence. Reconstruction using the sandwich technique – in which allograft is placed in the subchondral region and is overlaid with a layer of gelfoam and the rest of the cavity is filled with cement [14] Sandwich technique has been highly popular since first described by Campanacci as it circumvents the disadvantages of both, the bone cement and bone graft when used alone [14,15]. While bone cement helps with immediate stability and early post-operative mobilization, tumoricidal effect via heat generation and easier detection of recurrence as it is easily visualized at the bone-cement interface [16]. It also provides a technical advantage as the plasticity of cement during polymerization allows complete filling of a tumor cavity with irregular confines, and the polymer can be firmly molded around metallic implants [17]. However, it may result in but subchondral articular cartilage damage and joint destruction, and hence, increases the risk of requiring arthroplasty in the long term [13]. The shortcomings are tackled by the layer of subchondral bone graft, as it limits the thermal destruction of the cartilage, as well as provides structural support to the joint. Another advantage that using bone graft provides is a permanent reconstruction of the joint once the graft has completely incorporated into the native bone. The two components of the sandwich technique have a synergistic effect of reducing post-operative complications without increasing the rates of recurrence [18]. However, the vulnerability of this technique of curettage and cementation lies in the high fracture risk, which is caused due to the early loading of the bone and sub-optimal fixation of the cement in the cavity [19]. To alleviate the risk of post-operative fractures, various methods of internal fixation have been tried. These include, in increasing order of stability, Steinmann pins, interdigitating screws [17,20], and locking plates [21]. While inter-digitating screws were relatively less stability as compared to locking plates, they compensated in terms of technical feasibility as they could be inserted directly under vision [17], reduction in operative time, and minimal dissection was required further than what was already done for the curettage, all of which the author believed to be pivotal in the outcome for this patient with a deficient immune system [20]. Toy et al., in a biomechanical study of distal femur defects, furthered the case for this system. They proved that since the screws crossed the midline and had purchase on healthy bone on the opposite side, they allowed the cement mantle to remain stable, thus transferring most of the load to the bone away from the defect and therefore, if a fracture were to occur after this reconstruction, it would most likely be an fracture away from the cavity, which would not only be easier to treat but also would not necessitate any removal of implant. Even though the findings of this study cannot be directly applied to other anatomical sites, the author believes that the principles of augmenting the cementation with screws in a cross-beam construct can be adopted and applied to create a configuration custom to the area of the lesion, as it is the configuration of the screws and not the screws themselves which helps provide the stability. This could help surgeons circumvent the longer incisions and use of excessive hardware while achieving a similar level of stability and ease of future implant removal. The author used an interdigitating cancellous screw in the cavity of the proximal humerus in a pre-determined sequence to cater to the needs of the shoulder joint during its movements, protecting the soft tissue envelope. The screws helped create a scaffold and increase the core strength of the construct. Complimenting this with bone cement and a layer of sub-chondral bone graft to fill the cavity helped us achieve a stable construct in a minimally invasive way.(Fig.5)
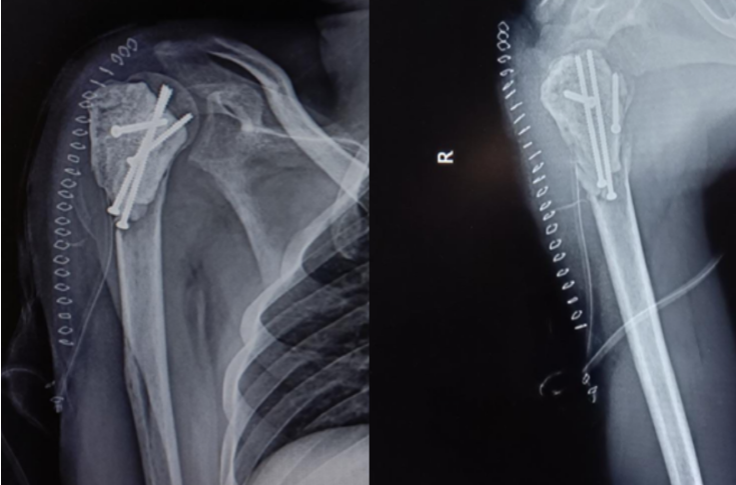
Figure 5: Fixation using 4 fully threaded cancellous screws and bone cement.
The case under discussion was a young individual with a lesser capacity to handle surgical stress due to the deficient immune system, with the expectations of considerable joint function and avoiding replacement surgery. This scenario adjudicated the use of intra-lesional curettage in place of wide resection, the sandwich technique for reconstruction and interdigitating screws in a cross-beam construct instead of conventional mode of locking plates as the mode of internal fixation despite the lack of literature, risk of recurrence, and less than highest possible level of stability.
This case report highlights the role of single-stage management of a proximal humerus Campanacci Grade 3 osteoclastoma, which was managed with curettage and screw interdigitation, and cement spacer. Patients with giant cell tumors of the bone have good life expectancy and low recurrence rates if managed carefully and meticulously; hence, author presents the use of screw interdigitation and cement spacer as a less morbid procedure, stable fixation with minimal damage to the natural anatomy and musculature in a case of a large locally advanced tumor with maximum return to complete range of motion and functional restoration of the shoulder joint.
This case demonstrates a minimally invasive, joint-preserving approach for treating a Campanacci Grade 3 GCT by combining intralesional curettage, sandwich reconstruction, and interdigitating screws. Tumor control was achieved while preserving shoulder function and minimizing recurrence risk. This method, backed by biomechanical studies, can be further explored for similar situations in other patients.
References
- 1. Thomas DM, Skubitz KM. Giant cell tumour of bone. Curr Opin Oncol 2009;21:338-44. [Google Scholar] [PubMed]
- 2. Werner M. Giant cell tumour of bone: Morphological, biological and histogenetical aspects. Int Orthop 2006;30:484-9. [Google Scholar] [PubMed]
- 3. Mavrogenis AF, Igoumenou VG, Megaloikonomos PD, Panagopoulos GN, Papagelopoulos PJ, Soucacos PN. Giant cell tumor of bone revisited. SICOT J 2017;3:54. [Google Scholar] [PubMed]
- 4. McDonald DJ, Sim FH, McLeod RA, Dahlin DC. Giant-cell tumor of bone. J Bone Joint Surg Am 1986;68:235-42. [Google Scholar] [PubMed]
- 5. Campanacci M, Baldini N, Boriani S, Sudanese A. Giant-cell tumor of bone. J Bone Joint Surg Am 1987;69:106-14. [Google Scholar] [PubMed]
- 6. Szendröi M, Czitrom AA, Szendro M. Experience with giant cell tumors. Magy Traumatol Orthop Helyreallito Seb 1989;32:151-9. [Google Scholar] [PubMed]
- 7. Singh S, Singh M, Mak I, Ghert M. Giant cell tumor of bone. Open Orthop J 2013;7:476-84. [Google Scholar] [PubMed]
- 8. Rooney RJ, Asirvatham R, Lifeso RM, Ali MA, Parikh S. Giant cell tumour of bone. A surgical approach to grade III tumours. Int Orthop 1993;17:87-92. [Google Scholar] [PubMed]
- 9. Szendröi M, Kiss J, Antal I. Surgical treatment and prognostic factors in giant-cell tumor of bone. Acta Chir Orthop Traumatol Cech 2003;70:142-50. [Google Scholar] [PubMed]
- 10. Lackman RD, Crawford EA, King JJ, Ogilvie CM. Conservative treatment of Campanacci grade III proximal humerus giant cell tumors. Clin Orthop Relat Res 2009;467:1355-9. [Google Scholar] [PubMed]
- 11. Knochentumoren A, Becker WT, Dohle J, Bernd L, Braun A, Cserhati M, et al. Local recurrence of giant cell tumor of bone after intralesional treatment with and without adjuvant therapy. J Bone Joint Surg Am 2008;90:1060-7. [Google Scholar] [PubMed]
- 12. Machak GN, Snetkov AI. The impact of curettage technique on local control in giant cell tumour of bone. Int Orthop 2021;45:779-89. [Google Scholar] [PubMed]
- 13. Gaston CL, Bhumbra R, Watanuki M, Abudu AT, Carter SR, Jeys LM, et al. Does the addition of cement improve the rate of local recurrence after curettage of giant cell tumours in bone? J Bone Joint Surg Br 2011;93:1665-9. [Google Scholar] [PubMed]
- 14. Saibaba B, Chouhan DK, Kumar V, Dhillon MS, Rajoli SR. Curettage and reconstruction by the sandwich technique for giant cell tumours around the knee. J Orthop Surg (Hong Kong) 2014;22:351-5. [Google Scholar] [PubMed]
- 15. Campanacci M, Capanna R, Fabbri N, Bettelli G. Curettage of giant cell tumor of bone. Reconstruction with subchondral grafts and cement. Chir Organi Mov 1990;75:212-3. [Google Scholar] [PubMed]
- 16. Segura J, Albareda J, Bueno AL, Nuez A, Palanca D, Seral F. The treatment of giant cell tumors by curettage and filling with acrylic cement. Long-term functional results. Chir Organi Mov 1997;82:373-80. [Google Scholar] [PubMed]
- 17. Toy PC, France J, Randall RL, Neel MD, Shorr RI, Heck RK. Reconstruction of noncontained distal femoral defects with polymethylmethacrylate and crossed-screw augmentation: A biomechanical study. Clin Orthop Relat Res 2006;452:167-73. [Google Scholar] [PubMed]
- 18. Benevenia J, Rivero SM, Moore J, Ippolito JA, Siegerman DA, Beebe KS, et al. Supplemental bone grafting in giant cell tumor of the extremity reduces nononcologic complications. Clin Orthop Relat Res 2017;475:776-83. [Google Scholar] [PubMed]
- 19. Pattijn V. Medical image-based, preformed titanium membranes for bone reconstructions: Design study and first clinical evaluation. J Biomech 2002;35:1200-8. [Google Scholar] [PubMed]
- 20. Toy PC, Arthur S, Brown D, Heck RK. Reconstruction of noncontained proximal tibial defects with divergent screws and cement. Clin Orthop Relat Res 2007;459:167-73. [Google Scholar] [PubMed]
- 21. Uglialoro AD, Maceroli M, Beebe KS, Benevenia J, Patterson FR. Distal femur defects reconstructed with polymethylmethacrylate and internal fixation devices: A biomechanical study. Orthopedics 2009;32:561-7. [Google Scholar] [PubMed]









