To study the role of neoadjuvant denosumab therapy along with conventional operative procedure in the recession and recurrence of giant cell tumor.
Dr. Ninad Kishor Honwadkar, Department of Orthopaedics, Grant Government Medical College and Sir JJ Group of Hospitals, Mumbai, Maharashtra, India. E-mail: drninadhonwadkar@gmail.com
Introduction: Giant cell tumor of bone (GCTB) is an uncommon benign primary bone tumor that mainly affects the long bones. Its occurrence is most frequent in patients between 30 and 40 years old. Curettage and bone grafting mainstay of treatment but lesions with large extent and unclear margins require meticulous planning and comparatively morbid procedures. A locally aggressive primary bone tumor that contains numerous osteoclasts formed from marrow-derived precursors through receptor activator of nuclear factor κ-B ligand (RANKL). Denosumab is immunoglobulin G2 monoclonal antibody which stops differentiation of osteoclast-like giant cells and consequent osteolytic damage by binding RANK-L.
Case Report: A 26-year-old female with complaints of pain in the right foot for 1 year, swelling in the right foot for 6 months, unable to bear weight on the affected foot with tender hard swelling present over the dorsomedial aspect of the right foot with inversion and eversion of the foot painful. Computed tomography scan and magnetic resonance imaging modalities revealed an expansile lesion in the navicular bone lobulated extension of lesion on the plantar aspect and adjacent tarsal bones and intertarsal joints. Histopathological report of the lesion determined giant cell tumor (GCT). After diagnosis of GCT established plan for excision of the lesion using curettage and chemical cauterization taken. More stable fixation; fill the void of excision bone grafting along with plate (3.5 mm combihole plate) fixation done Neo-adjuvant denosumab therapy given intraoperatively + regular intervals (denosumab 120 mg on day 0, 8, 15, 28). With informed consent patient was regularly followed annually, half year analysis of the patient reported a significant reduction in pain and no new increase of size in swelling or any new swelling. Adverse effect: Muscular pain and fatigue often encountered with denosumab therapy not seen in the patient.
Conclusion: Denosumab leads to downsizing of the tumor and leads to fibrosis of the margin and inhibiting the progress of the disease. Planned resection may become less morbid when preoperative denosumab is administered. En bloc resection of GCTB, especially in the case of soft-tissue involvement, can be facilitated by a neoadjuvant denosumab regimen. Strong evidence for the effect of denosumab for non-resectable GCTB. Optimal treatment duration and maintenance dose and interval of denosumab in unresectable disease are still under analysis.
Keywords: Navicular, giant cell tumor, curettage, denosumab, fixation.
Giant cell tumor of bone (GCTB) is an uncommon benign primary bone tumor that mainly affects the long bones. Their occurrence is most frequent in patients between 30 and 40 years old. It is a locally aggressive primary bone tumor that contains numerous osteoclasts formed from marrow-derived precursors through receptor activator of nuclear factor κ-B ligand (RANKL) [1], an osteoclast differentiation factor expressed in neoplastic cells of GCTB which rarely transforms in malignant form. Curettage and bone grafting are the mainstay of treatment [2], but lesions with a large extent and unclear margins require meticulous planning and comparatively morbid procedures [3]. Denosumab is an immunoglobulin G2 monoclonal antibody which stops the differentiation of osteoclast-like giant cells and consequent osteolytic damage by binding RANK-L [4]. The main objective of this case report is to study the functional outcome of combined operative and neoadjuvant therapy with follow-up to analyze about recurrence.
A Fig. 1 the arrow head indicating the swelling Fig. 1 the arrow head indicating the swelling 6 year old female came with complaints of pain in right foot since 1 year, swelling in the right foot since 6 months. Patient is unable to bear weight on the affected foot.
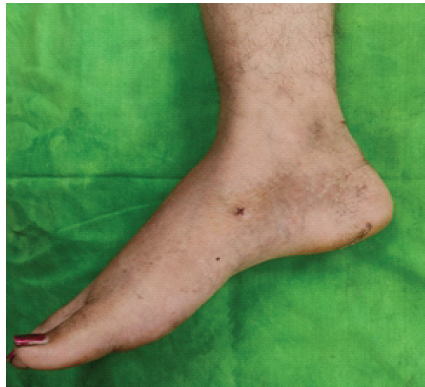
Figure 1: The arrow head indicating the swelling.
Swelling present over the dorsomedial aspect of right foot 3 × 3 × 1 cm in size, hard in consistency, skin over the swelling is free.
Tenderness present over swelling, inversion, and eversion of the foot painful.
Assessment
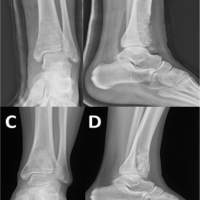
Routine radiological investigations revealed a large globular lesion in the navicular bone (Fig. 2). Computed tomography scan (Fig. 3, 4) and magnetic resonance imaging (Fig. 5) modalities revealed an expansile lesion in the navicular bone lobulated extension of lesion on the plantar aspect and adjacent tarsal bones (cuneiform, cuboid) and intertarsal joints. We thus had a suspicion of a malignant lesion? Round cell tumour? Ewing’s sarcoma. On following up with histopathology, the histopathological report of lesion determined giant cell tumor (GCT).
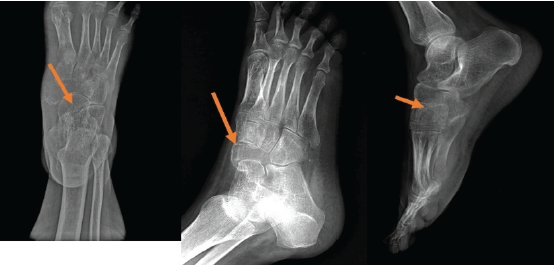
Figure 2: Routine X-rays.
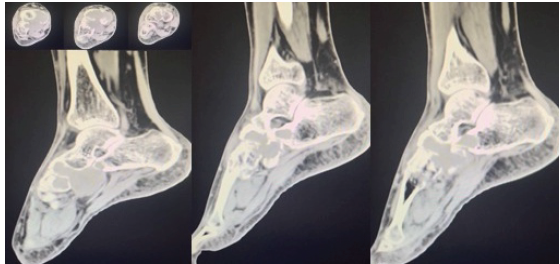
Figure 3: Computed tomography scan shows expansile lesion.
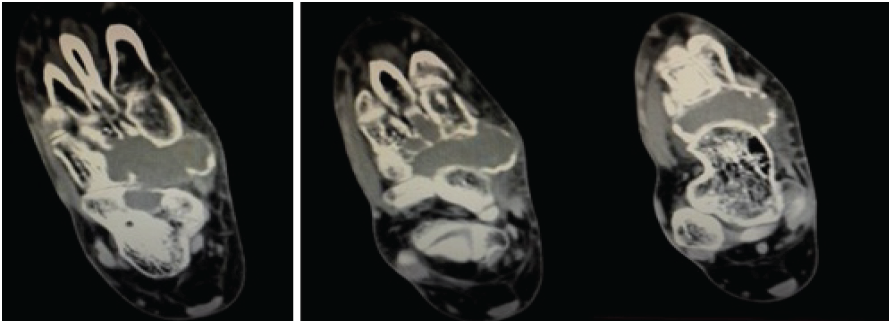
Figure 4: Computed tomography scan shows expansile lesion.
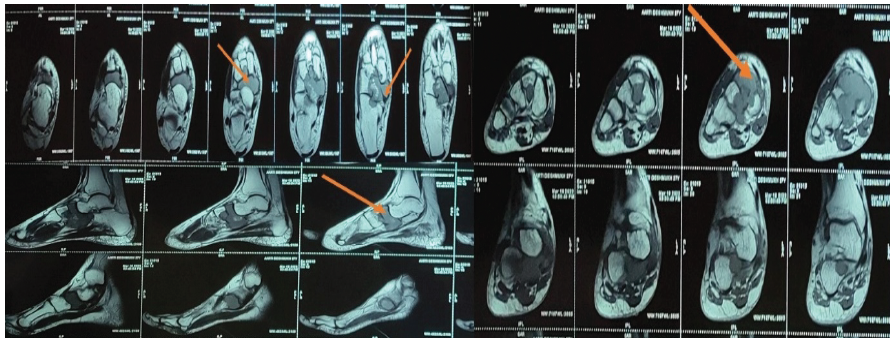
Figure 5: Magnetic resonance imaging scans indicate hypointense lesion in the navicular bone.
Management
After the diagnosis of GCT was established, the patient was prepared for excision of the lesion using curettage and chemical cauterization. To impart more stable fixation and to fill the void of excision, bone grafting along with plate (3.5 mm combihole plate) fixation was done (Fig. 6, 7).
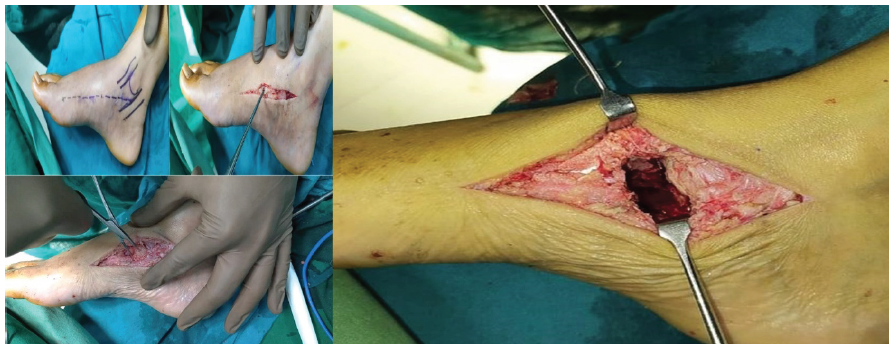
Figure 6: Intraoperative images.
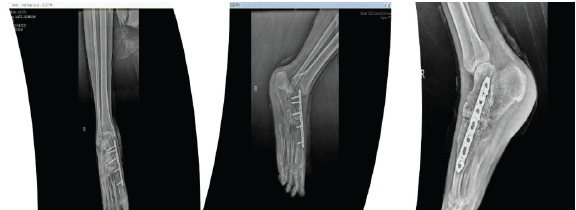
Figure 7: Immediate post-operative X-rays.
Neo adjuvant denosumab therapy was given pre and intraoperatively and at regular intervals. (denosumab 120 mg on day 0, 8, 15, 28) [5].
Denosumab leads to downsizing of the tumor and leads to fibrosis of the margin and inhibiting progress of the disease [4,5]; hence, a planned resection may become less morbid when preoperative denosumab is administered [6]. There is no confirmation of improved local control post-surgery, but more importantly, no increase in recurrence rates has been confirmed either [7]. En-bloc resection of GCTB, especially in the case of soft-tissue involvement, can be facilitated by a neoadjuvant denosumab regimen. The optimal treatment duration and maintenance dose and interval of denosumab in unresectable disease are still under analysis [8,9]. After stopping denosumab, the incidence was increased which was noted in few studies [10] (Fig. 8, 9). 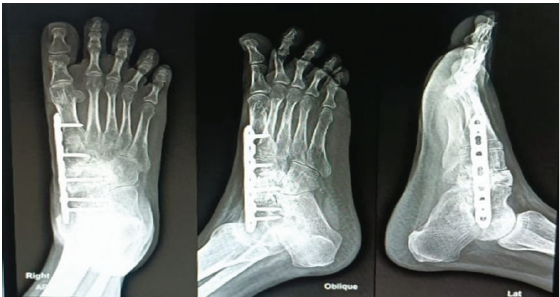
Figure 8: 8-month follow-up X-rays.
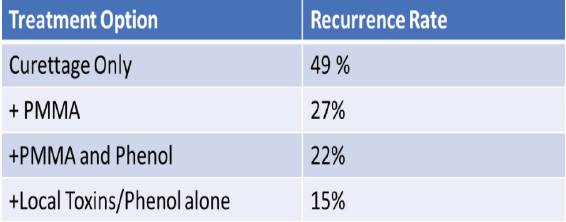
Figure 9: Comparative analysis of different modalities of management.
With proper written informed consent, the patient was on regular quarterly follow-up schedules for 1 year. At the half-year analysis, the patient reported a significant reduction in pain and no new increase of size in swelling or any new swelling. Adverse effects such as muscular pain and fatigue often encountered with denosumab therapy were not seen in this patient.
At present, the role of neoadjuvant denosumab in operable GCTB is limited to selected cases in which diffuse reactive bone formation and peripheral ossification can make an easier surgical procedure, for example, in tumors with a large soft tissue component. Evidence has been found that pre-operative denosumab therapy helps in downsizing the tumor with an indirect action on the tumor vasculature, leading to fibrosis of the soft tissue component of the tumor, leading to enhancing the margins for efficient resection. Adequate resection with cauterization with chemical components such as phenol, polymethyl methacrylate eventually reduces the possibility of recurrence, while denosumab plays an RANK-L ligand inhibiting role in recurrence.
References
- 1. Singh VA, Puri A. The current standing on the use of denosumab in giant cell tumour of the bone. J Orthop Surg (Hong Kong) 2020;28:2309499020979750. [Google Scholar] [PubMed]
- 2. Palmerini E, Staals EL, Jones LB, Donati DM, Longhi A, Randall RL. Role of (Neo)adjuvant denosumab for giant cell tumor of bone. Curr Treat Options Oncol 2020;21:68. [Google Scholar] [PubMed]
- 3. Alothman M, Althobaity W, Asiri Y, Alreshoodi S, Alismail K, Alshaalan M. Giant cell tumor of bone following denosumab treatment: Assessment of tumor response using various imaging modalities. Insights Imaging 2020;11:41. [Google Scholar] [PubMed]
- 4. Ahn JH, Choy WS, Kim HY, Kim HS. Midfoot reconstruction for a large, aggressive giant-cell tumor: A case report. J Bone Joint Surg Am 2011;93:e133. [Google Scholar] [PubMed]
- 5. Li H, Gao J, Gao Y, Lin N, Zheng M, Ye Z. Denosumab in giant cell tumor of bone: current status and pitfalls. Front Oncol 2020;10:580605. [Google Scholar] [PubMed]
- 6. Agarwal MG, Gundavda MK, Gupta R, Reddy R. Does denosumab change the giant cell tumor treatment strategy? Lessons learned from early experience. Clin Orthop Relat Res 2018;476:1773-82. [Google Scholar] [PubMed]
- 7. Cowan RW, Singh G. Giant cell tumor of bone: A basic science perspective. Bone 2013;52:238-46. [Google Scholar] [PubMed]
- 8. Chawla S, Blay JY, Rutkowski P, Le Cesne A, Reichardt P, Gelderblom H, et al. Denosumab in patients with giant-cell tumour of bone: A multicentre, open-label, phase 2 study. Lancet Oncol 2019;20:1719-29. [Google Scholar] [PubMed]
- 9. Chawla S, Henshaw R, Seeger L, Choy E, Blay JY, Ferrari S, et al. Safety and efficacy of denosumab for adults and skeletally mature adolescents with giant cell tumour of bone: Interim analysis of an open-label, parallel-group, phase 2 study. Lancet Oncol 2013;14:901-8. [Google Scholar] [PubMed]
- 10. Knochentumoren A, Becker WT, Dohle J, Bernd L, Braun A, Cserhati M, et al. Local recurrence of giant cell tumor of bone after intralesional treatment with and without adjuvant therapy. J Bone Joint Surg Am 2008;90:1060-7. [Google Scholar] [PubMed]
- 11. Deveci MA, Paydaş S, Gönlüşen G, Özkan C, Biçer ÖS, Tekin M. Clinical and pathological results of denosumab treatment for giant cell tumors of bone: Prospective study of 14 cases. Acta Orthop Traumatol Turc 2017;51:1-6. [Google Scholar] [PubMed]