This case highlights that rare opportunistic pathogens, such as Campylobacter fetus, should be considered in the differential diagnosis of periprosthetic joint infections – especially in immunocompromised patients or atypical presentations – as prompt identification is essential to prevent limb- or life-threatening complications.
Dr. Anna Hunkár, Department of Orthopaedics, Semmelweis University, Budapest, Hungary. E-mail: anna.hunkar@gmail.com
Introduction: Periprosthetic joint infection (PJI) is a serious complication following total joint arthroplasty. It can be potentially limb- or life-threatening, often necessitating complex surgical intervention and prolonged antimicrobial therapy. While Staphylococcus aureus and coagulase-negative staphylococci are the most commonly implicated pathogens, rare organisms, such as Campylobacter fetus, can occasionally be involved, particularly in immunocompromised patients or those with specific dietary exposures. Due to its low virulence and atypical clinical presentation, infections caused by C. fetus may lead to diagnostic delays.
Case Report: We present the case of an 80-year-old woman with a history of total knee arthroplasty, who developed a C. fetus PJI complicated by a draining sinus tract.
Conclusion: This case underscores the diagnostic complexity of atypical PJI. It highlights the importance of repeated aspirations and comprehensive clinical evaluation. Additionally, it reveals that joint aspiration can yield false-negative results if performed during or shortly after an antibiotic course and contributes to the limited literature on C. fetus as a causative organism in PJI.
Keywords: Periprosthetic joint infection, Campylobacter fetus, atypical pathogen.
Periprosthetic joint infection (PJI) remains one of the most serious complications following total joint arthroplasty, often associated with prolonged hospitalization, and the need for complex surgical management. The condition is typically caused by high-virulence organisms such as Staphylococcus aureus or coagulase-negative staphylococci, which usually present with acute inflammation, local signs of infection, and elevated inflammatory markers. In contrast, atypical or low-virulence pathogens may lead to more insidious disease courses, resulting in delayed diagnosis and treatment. Campylobacter fetus is a rare but clinically significant zoonotic pathogen. Well known for causing systemic infections in immunocompromised hosts, but only sporadically reported as a cause of PJI. Its unusual clinical presentation, lack of specific diagnostic markers, and limited culture yield further complicate recognition. Existing literature over the past three decades documents only a small number of C. fetus PJIs, highlighting the exceptional rarity of this condition and the absence of standardized treatment recommendations. This case contributes to the existing body of knowledge by presenting a PJI caused by C. fetus in a patient without obvious immunosuppression, illustrating the diagnostic challenges and emphasizing the importance of repeated joint aspiration.
Our patient is an 80-year-old woman who underwent right total knee arthroplasty 15 years ago. Her medical history includes breast cancer, for which she underwent mastectomy, sentinel lymph node removal, and subsequent radiation therapy due to metastatic disease. She also has a history of asthma and severe polydiscopathy, both of which have been treated with corticosteroids. Additional comorbidities include hypertension, ischemic heart disease, gastroesophageal reflux disease, dyspepsia, and thrombocytopenia. The patient has a documented penicillin allergy. Initially presented with right knee pain in July 2024 and over the following 3 months, she underwent outpatient investigations. As no surgical intervention was deemed necessary, conservative treatment was initiated, including non-steroidal anti-inflammatory drugs, ice application, rest, and physical therapy. By October 2024, her symptoms progressed, and she developed swelling and erythema around the right knee and intermittent fever. Fig. 1 illustrates the clinical appearance of her knee during this initial evaluation phase.
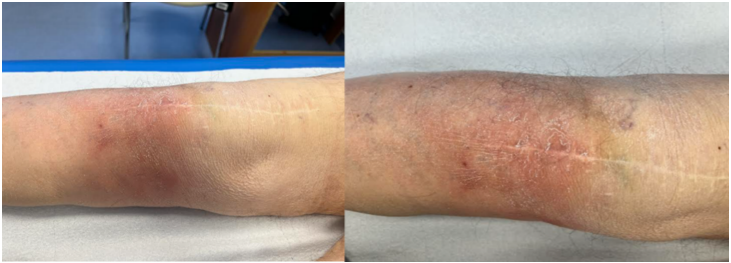
Figure 1: Images of the patient’s right knee. Hyperemia and moderate swelling can be observed around the distal region of the surgical scar.
Therefore, 4 months after the onset of her symptoms. In October, the patient presented to the emergency department, where deep vein thrombosis was ruled out. She was diagnosed with erysipelas and started on oral clindamycin. As her symptoms persisted, she was re-evaluated in the orthopedic outpatient clinic, where joint aspiration was performed. The result was negative, leading to a preliminary diagnosis of aseptic loosening. She was subsequently referred to our department in November 2024, where a diagnosis of polyethylene wear was considered. Radiographs taken at that time (Fig. 2) revealed lytic lesions and significant bone resorption around both the femoral and tibial components.
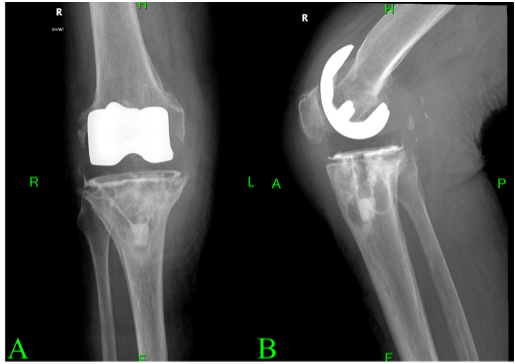
Figure 2: Prominent resorptive changes are observed of the femoral component. Lytic lesions are also noted along the tray of the tibial component. The image in panel A (a) shows an anteroposterior view, while panel B (b) presents a lateral view of the radiograph.
Despite the previous negative aspiration result, the clinical presentation – along with the macroscopic changes depicted in Fig. 1 – continued to raise suspicion for a PJI. As a result, repeated aspiration was performed in November 2024, which yielded a positive result for C. fetus. The patient was subsequently scheduled for a two-stage revision procedure. A few weeks later, a draining sinus tract developed at the surgical site, as shown in Fig. 3.
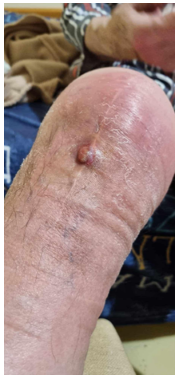
Figure 3: Photograph of the patient’s right knee taken on December 2024, showing the draining sinus tract at the surgical site.
According to the 2018 International Consensus Meeting criteria, the presence of a sinus tract constitutes a major criterion for PJI. Consequently, the first stage of the planned two-stage revision was performed ahead of schedule. In January 2025, the infected prosthesis was explanted, and an antibiotic (vancomycin and gentamycin) elution handmade bone cement mobile spacer was implanted. Follow-up radiographs taken 6 weeks postoperatively are shown in Fig. 4.
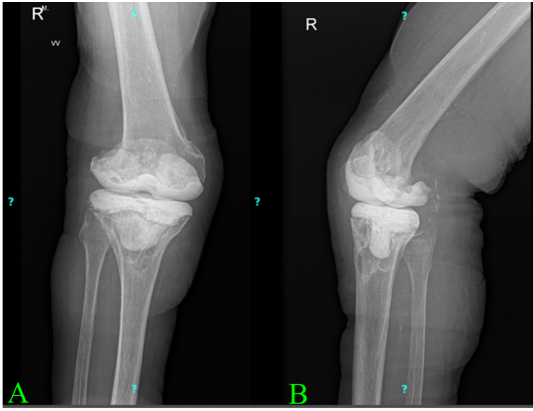
Figure 4: Anteroposterior and lateral radiographs of the right knee following spacer implantation, taken 6 weeks postoperatively. The image in panel A (a) shows the anteroposterior view, and image in panel B (b) shows the lateral view.
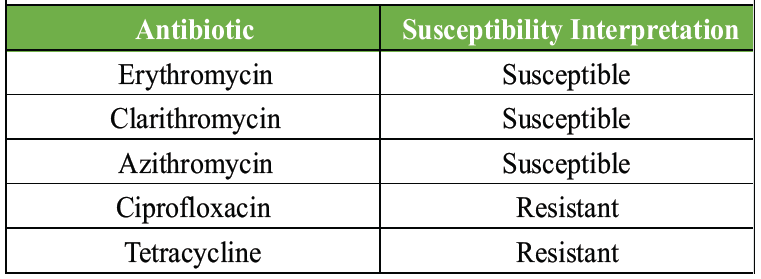
Table 1: The antibiotic susceptibility profile of Campylobacter fetus cultured from intraoperative microbiological samples
Intraoperative microbiological cultures also confirmed the presence of C. fetus. On retrospective questioning, the patient reported consuming goat cheese before the onset of symptoms, which could be the most likely source of the infection. Susceptibility testing was initially performed by the Kirby-Bauer disk diffusion method to ciprofloxacin, tetracycline, and erythromycin; unfortunately, the isolate was non-viable in subculture, and further susceptibility testing could not be performed. Erythromycin susceptibility was interpreted based on breakpoints of C. coli and C. jejuni defined by EUCAST (European Committee on Antimicrobial Susceptibility Testing) [1]. Based on the susceptibility profile (Table 1) provided by a microbiology laboratory, a multidisciplinary team, including orthopedic surgeon microbiologist and infectious disease specialists, designed the antimicrobial regimen. Initially, an intravenous combination therapy of gentamicin and vancomycin was administered, which was later switched to meropenem and vancomycin. This was followed by a 10-week course of intravenous meropenem monotherapy, as oral treatment options were limited. Although azithromycin was a viable candidate based on sensitivity testing, there was insufficient experience regarding its long-term use for this indication. The patient spent 3 weeks in the orthopedic ward, and intravenous antibiotic therapy continued during rehabilitation for a total of 12 weeks. During the follow-up visits in April and June 2025, the patient showed clinical improvement, and her inflammatory markers were within normal limits without the need for antibiotic therapy. As a result, reimplantation of the prosthesis was carried out in June. The patient received antibiotic prophylaxis appropriate for a general incision for the prosthesis reimplantation surgery. Due to a confirmed penicillin allergy, the prophylaxis consisted of clindamycin 3 times daily. Clindamycin therapy continued postoperatively until the surgical culture results became available. As all intraoperative cultures returned negative, the antibiotic treatment was discontinued on post-operative day 10, considering the sterile results. In our clinic, during two-stage revision procedures, we usually administer prolonged antibiotic therapy for 6–12 weeks after prosthesis reimplantation, based on the sensitivity of the pathogen identified during the first stage. In this case, due to the negative culture results and the fact that prolonged treatment could only have been provided intravenously, we decided – after evaluating the risk-benefit ratio – to discontinue parenteral antibiotic therapy and terminate the antibiotic treatment. In the early post-operative period, the patient experienced severe pain, for which an epidural catheter was placed to provide continuous analgesia. However, this intervention proved to be ineffective. Following the removal of the catheter, the patient developed peroneal nerve paresis. A magnetic resonance imaging of the lumbosacral spine revealed a hematoma compressing the exiting nerve root. The hematoma was surgically evacuated by the neurosurgical team. Following the intervention, the patient was admitted to a rehabilitation facility. At the rehabilitation facility, 2 months after prosthesis reimplantation, the patient developed warmth and swelling of the operated knee following physical exertion and a possible “cracking sensation,” which was associated with a transient fever of 38.1°C. Laboratory tests showed mildly elevated inflammatory markers. Bacteriuria was identified and successfully treated with fosfomycin, resulting in resolution of fever and normalization of urine findings. Physical examination revealed a persistent joint effusion; aspiration yielded 25 mL of blood-stained fluid, which was submitted for microbiological culture. Prophylactic meropenem therapy and radiographic assessment were initiated due to hemarthrosis. The culture was negative for bacterial growth, radiographs suggested possible tibial component loosening, which was not supported clinically. Inflammatory markers gradually normalized. At the 3-month follow-up, the patient was asymptomatic, and inflammatory markers were within normal range.
Campylobacter species are Gram-negative, curved bacteria primarily known for causing gastrointestinal infections [2]. C. fetus is mainly a zoonotic pathogen, with infection typically occurring after the consumption of raw or undercooked meat, contaminated water, or unpasteurized dairy products. While C. jejuni and Campylobacter coli are the most common causes of intestinal campylobacteriosis, C. fetus is the species most frequently associated with Campylobacter bacteremia. Although rare, the infection can become systemic, especially in immunocompromised individuals [2]. This group is more likely to include individuals with underlying conditions such as human immunodeficiency virus infection, chronic liver disease, diabetes mellitus, cardiovascular disease, or the presence of implanted medical devices. Immunosuppressive states due to chemotherapy, immunotherapy, or long-term corticosteroid use are also recognized risk factors. However, healthy individuals may also be affected, particularly those with occupational exposure to animals, such as farm or abattoir workers. C. fetus can colonize the oral cavity, and dental disease may lead to the hematogenous spread of the bacteria. In susceptible individuals, C. fetus usually spreads to the bloodstream from the gastrointestinal tract, with bacteremia often preceded or accompanied by episodes of diarrhea [3]. In our case, this was not observed; the patient denied experiencing any gastrointestinal symptoms either before or concurrently with their complaints. However, the patient did report consuming homemade goat cheese, which could be a potential source of the infection. The treatment of invasive C. fetus infections necessitates parenteral antibiotic therapy, but there are no established guidelines regarding the optimal antimicrobial regimen, and there are no EUCAST breakpoints for the interpretation of the antibiotic susceptibility of this species. A limited number of studies show that C. fetus strains have low minimum inhibitory concentrations for meropenem and gentamicin (under one mg/L each, respectively), indicating that treatment with these antibiotics is probably effective. Susceptibility to other antibiotics, such as ampicillin or ciprofloxacin, varies by strain, but may be valid therapeutic options following susceptibility testing [4]. Systemic infections have been successfully treated with imipenem, meropenem, ampicillin, gentamicin, and chloramphenicol [5,6]. Previous reports have documented resistance to ciprofloxacin, doxycycline, and erythromycin [7]. In our case, the antibiogram demonstrated resistance to ciprofloxacin and tetracycline; therefore, meropenem was chosen as the parenteral treatment after the removal of the prosthesis for over 12 weeks. Following the second surgery, the patient received clindamycin for 1 week. C. fetus bacteremia may lead to the infection of implanted medical devices, such as biological or mechanical prosthetic heart valves, internal cardioverter defibrillator devices, breast implants, and hip or knee joint prostheses [8,9,10]. Over the past three decades, a total of 18 cases of C. fetus PJI have been reported (Table 2).
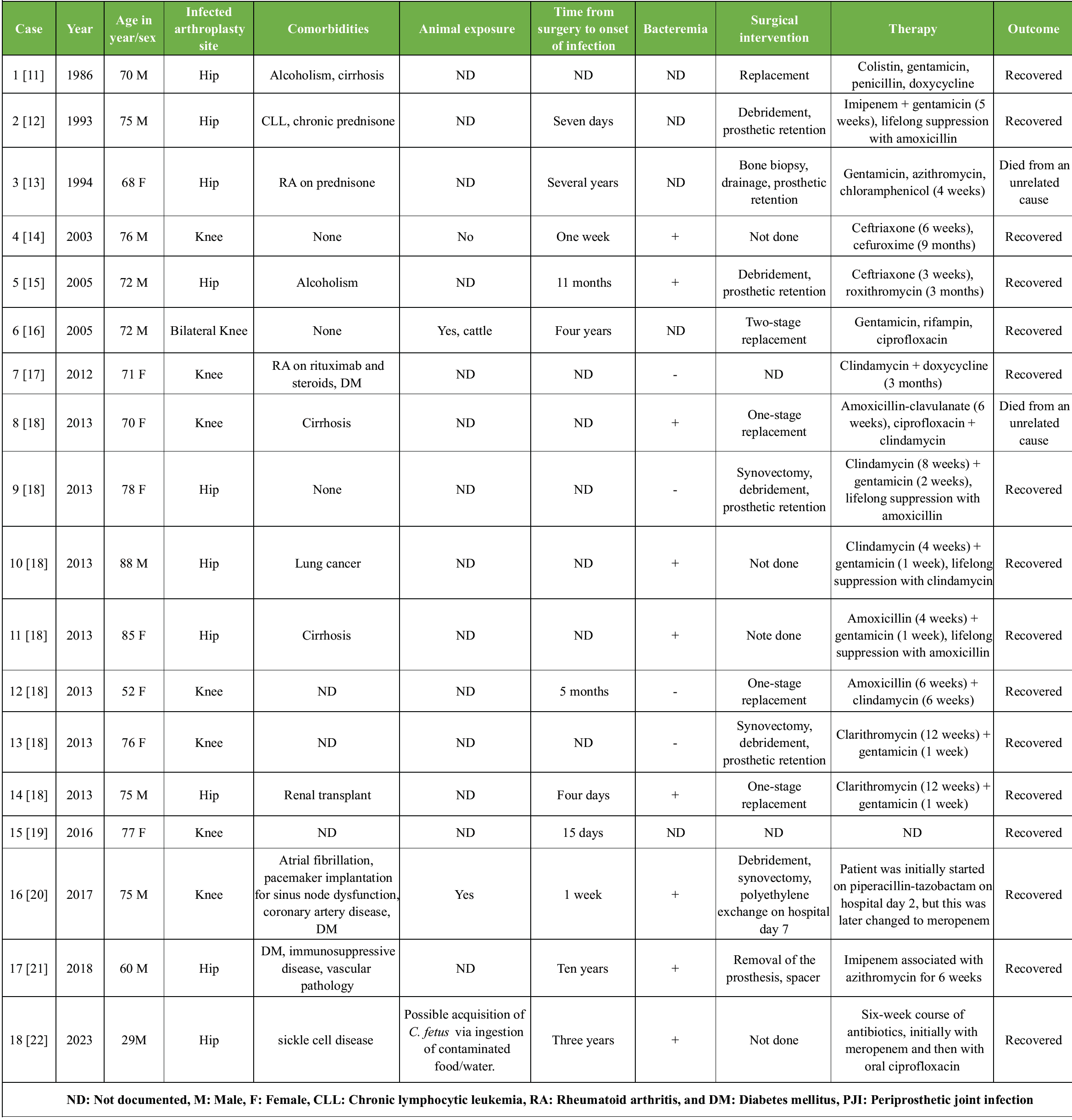
Table 2: Patient demographics, clinical presentations, and treatment outcomes in published cases of Campylobacter fetus PJIs
Bilateral involvement was described in only one case. Approximately 50% of the infections affected hip prostheses, while the remaining 50% involved knee prostheses. All patients were aged 60 years or older, and the majority had notable underlying comorbidities or conditions associated with immunosuppression. In most cases, antimicrobial therapy was combined with surgical intervention. The most notable difference between global trends and our chosen approach lies in the treatment strategy. In the majority of published cases, clinicians opted to retain the original prosthesis or perform a one-stage revision. The outcomes were favorable; except for two patients who died from unrelated causes, all others had recovered from the infection by the time of their last follow-up. Another difference is that, in our case, the patient cannot be clearly classified as immunosuppressed, whereas in the literature, nearly all reported patients had some identifiable immunocompromising factor. Based on this, the pathogen should not be considered capable of causing this condition exclusively in immunosuppressed individuals.
C. fetus is a frequently overlooked and underreported pathogen that can lead to severe, invasive, and occasionally fatal infections in humans. With the increasing number of prosthetic joint surgeries and associated infections, as well as the growing population of elderly individuals and patients with immunocompromising conditions such as chronic illnesses, malignancies, or those undergoing immunosuppressive therapies, the incidence of C. fetus-related PJI may rise. It is crucial to recognize C. fetus as a true pathogen in cases of confirmed joint infections.
This case highlights that C. fetus, though rare, should be considered a potential pathogen in PJI, even in patients without clear immunosuppression. Early recognition and repeated aspirations can prevent diagnostic delay and significantly improve outcomes.
References
- 1. Sifré E, Salha BA, Ducournau A, Floch P, Chardon H, Mégraud F, et al. EUCAST recommendations for antimicrobial susceptibility testing applied to the three main Campylobacter species isolated in humans. J Microbiol Methods 2015;119:206-13. [Google Scholar] [PubMed]
- 2. Wagenaar JA, Van Bergen MA, Blaser MJ, Tauxe RV, Newell DG, Van Putten JP. Campylobacter fetus infections in humans: Exposure and disease. Clin Infect Dis 2014;58:1579-86. [Google Scholar] [PubMed]
- 3. Blaser MJ. Campylobacter fetus–emerging infection and model system for bacterial pathogenesis at mucosal surfaces. Clin Infect Dis 1998;27:256-8. [Google Scholar] [PubMed]
- 4. Zhang MM, Melton TA, Akhtar H, Shah A, Schuetz AN, Clement J, et al. Antimicrobial susceptibilities of Campylobacter fetus: Report from a reference laboratory. J Clin Microbiol 2024;62:e0144723. [Google Scholar] [PubMed]
- 5. Tremblay C, Gaudreau C, Lorange M. Epidemiology and antimicrobial susceptibilities of 111 Campylobacter fetus subsp. Fetus strains isolated in Québec, Canada, from 1983 to 2000. J Clin Microbiol 2003;41:463-6. [Google Scholar] [PubMed]
- 6. Fujihara N, Takakura S, Saito T, Iinuma Y, Ichiyama S. A case of perinatal sepsis by Campylobacter fetus subsp. Fetus infection successfully treated with carbapenem–case report and literature review. J Infect 2006;53:e199-202. [Google Scholar] [PubMed]
- 7. Van Der Graaf-Van Bloois L, Duim B, Looft T, Veldman KT, Zomer AL, Wagenaar JA. Antimicrobial resistance in Campylobacter fetus: Emergence and genomic evolution. Microb Genom 2023;9:mgen000934. [Google Scholar] [PubMed]
- 8. Peetermans WE, De Man F, Moerman P, Van De Werf F. Fatal prosthetic valve endocarditis due to Campylobacter fetus. J Infect 2000;41:180-2. [Google Scholar] [PubMed]
- 9. Ahmar W, Johnson D, Richards M, Strathmore N. Campylobacter fetus infection of an internal cardioverter defibrillator. Pacing Clin Electrophysiol 2008;31:258-9. [Google Scholar] [PubMed]
- 10. Nishikubo M, Nasu S, Maruoka H, Kawabata T, Ikeda M, Nishioka H. Sequential breast implant infections due to Campylobacter fetus subsp. Fetus. J Infect Chemother 2021;27:1080-3. [Google Scholar] [PubMed]
- 11. Joly P, Boissonnas A, Fournier R, Khalifa P, Vedel G, Cremer GA, et al. Septic arthritis caused by Campylobacter fetus. Rev Rhum Mal Osteoartic 1986;53:223-6. [Google Scholar] [PubMed]
- 12. Yao JD, Ng HM, Campbell I. Prosthetic hip joint infection due to Campylobacter fetus. J Clin Microbiol 1993;31:3323-4. [Google Scholar] [PubMed]
- 13. Bates CJ, Clarke TC, Spencer RC. Prosthetic hip joint infection due to Campylobacter fetus. J Clin Microbiol 1994;32:2037. [Google Scholar] [PubMed]
- 14. Cone LA, Dreisbach PB, Hirschberg J, Shekar C, Dreisbach LP, Salatich W. Cellulitis and septic arthritis caused by Campylobacter fetus and Campylobacter jejuni: Report of 2 cases and review of the literature. J Clin Rheumatol 2003;9:362-9. [Google Scholar] [PubMed]
- 15. Chambers ST, Morpeth SC, Laird HM. Campylobacter fetus prosthetic hip joint infection: Successful management with device retention and review. J Infect 2005;50:258-61. [Google Scholar] [PubMed]
- 16. David J, Nasser RM, Goldberg JW, Reed KD, Earll MD. Bilateral prosthetic knee infection by Campylobacter fetus. J Arthroplasty 2005;20:401-5. [Google Scholar] [PubMed]
- 17. Meyer A, Theulin A, Chatelus E, Argemi X, Sordet C, Javier RM, et al. Campylobacter fetus infection in three rheumatoid arthritis patients treated with rituximab. Ann Rheum Dis 2012;71:1094-5. [Google Scholar] [PubMed]
- 18. Prendki V, Marmor S, Zeller V, Lhotellier L, Mégraud F, Desplaces N. Campylobacter infection after prosthetic joint surgery. Scand J Infect Dis 2013;45:706-10. [Google Scholar] [PubMed]
- 19. Yahia SA, Zeller V, Desplaces N, Chazerain P, Lhotellier L, Marmor S, et al. Crystal-induced arthritis after arthroplasty: 7 cases. Joint Bone Spine 2016;83:559-62. [Google Scholar] [PubMed]
- 20. Dumic I, Sengodan M, Franson JJ, Zea D, Ramanan P. Early onset prosthetic joint infection and bacteremia due to Campylobacter fetus subspecies fetus. Case Rep Infect Dis 2017;2017:5892846. [Google Scholar] [PubMed]
- 21. Zamora-López MJ, Álvarez-García P, García-Campello M. Prosthetic hip joint infection caused by Campylobacter fetus: A case report and literature review. Rev Esp Quimioter 2018;31:53-7. [Google Scholar] [PubMed]
- 22. Azar SS, Simeone FJ, Jarolimova J, Nussbaum EZ. Case 37-2023: A 29-year-old man with sickle cell disease and right hip pain. N Engl J Med 2023;389:2088-96. [Google Scholar] [PubMed]