Escherichia hermannii, though rare, can cause implant-associated infections and biofilm formation even in immunocompetent patients, leading to non-union. Early culture-based diagnosis, awareness of possible antibiotic resistance, and prompt surgical debridement with targeted antimicrobial therapy are essential for successful management.
Dr. C R Jithin, Department of Orthopedics, Baby Memorial Hospital, Kannur, Kerala, India. E-mail: drjithincradhakrishnan@gmail.com
Introduction: Escherichia hermannii is an infrequent monomicrobial pathogen in human infections. Notably, it has never been documented as the sole isolate in cases of infected radial shaft fractures.
Case Report: This report presents a case of a non-union in a distal third radius shaft fracture associated with an E. hermannii-infected implant that remained in situ. The patient, who suffered a crush injury 3 months prior, initially underwent open reduction and internal fixation using a fibula strut graft secured with a titanium plate, along with primary wound closure. However, after 12 weeks, he developed an infection at the implant site, accompanied by a sinus tract. To address this complication, a repeat debridement was performed, and a retention strategy was implemented, allowing the original implant to be preserved.
Conclusion: The patient was treated with intravenous meropenem for 7 days, followed by an 11-week course of oral ciprofloxacin. At the conclusion of treatment, he remained free of infection, asymptomatic, and was able to continue working with excellent functional outcomes.
Keywords: Escherichia hermannii, radial shaft fracture, implant-associated infection, nonunion, biofilm.
Escherichia hermannii, a member of the Enterobacteriaceae family, was initially described in 1982 and later classified as a distinct species within the Escherichia genus due to identified biochemical and genomic differences from Escherichia coli. Characterized as a Gram-negative, rod-shaped bacterium, E. hermannii was first noted in 2008, involving a case of purulent conjunctivitis in a patient with no prior antibiotic treatment [1,2]. Although E. hermannii has been associated with infections over the past few decades – most commonly in cases of bloodstream and urinary tract infections – it has not previously been documented as the primary pathogen in cases of infected implants in radial shaft fractures [3]. This case contributes to the expanding body of literature that highlights E. hermannii as a potentially pathogenic and virulent organism capable of causing monomicrobial infections. Understanding its mechanisms of infection and the conditions under which it can act as a primary pathogen is crucial for enhancing diagnostic accuracy and treatment strategies. The findings suggest that there must be heightened awareness of E. hermannii in clinical practice, particularly in complex cases involving implant-related infections.
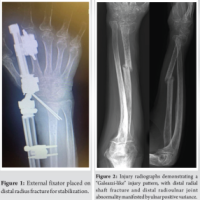
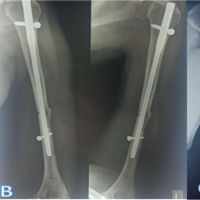
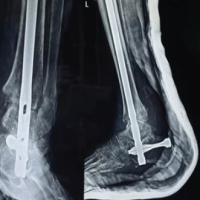
A 52-year-old man was referred to our tertiary care center for a chronic draining wound at the site of a prior open radial shaft fracture. He is a known case of Type 2 diabetes mellitus and is on treatment for the same. The patient initially had a crush injury to the left forearm, which resulted in an absent distal pulse. He was planned for a staged procedure given the Gustilo Type 3C open fracture to achieve an optimal clinical outcome. An external fixator responded by performing a thorough wound debridement, exploration, and bone stabilization. A revascularization procedure was performed by a plastic surgeon on the left forearm, involving primary reconstruction of the left radial and ulnar arteries along with the cephalic vein using a massive saphenous vein graft. Subsequently, a repair of the left median nerve using a cable graft was performed in the initial stage as per damage control orthopedic protocols. Stay sutures were put in, but the defect was not covered. A week later, a groin flap was utilized for coverage of the left forearm. One month after the flap procedure, there was satisfactory flap take-up and no clinical signs of infection; hence, the left groin flap was divided, and the left forearm wound was further debrided and sutured. During this stage, he was planned for definitive fixation of bone; hence, the external fixator was removed. His distal 1/3rd radius fracture with 3 cm bone loss (Fig. 1a and b) was addressed with the volar Henry approach without disturbing the flap extending from the lateral aspect of the radial side to dorsally. He underwent an open reduction internal fixation using a titanium dynamic compression plate, where the 3 cm bone loss was addressed using a fibula strut graft taken from the ipsilateral side (Fig. 2a, b, c).
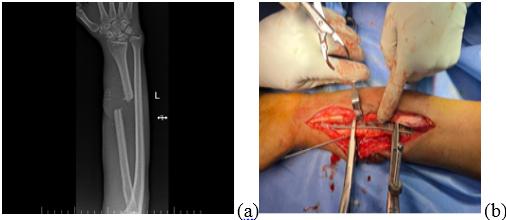
Figure 1: Pre-operative X-rays of distal 1/3rd radius fracture with 3 cm bone loss.
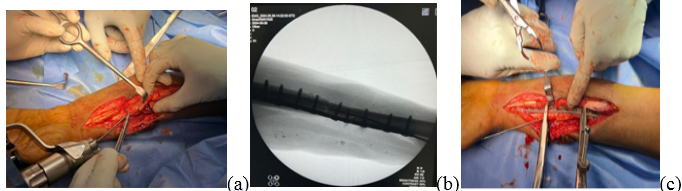
Figure 2: (a, b, c) Intraoperative images of open reduction internal fixation using fibula strut and primary wound closure.
Regular wound inspection was done, and he was on intravenous cefuroxime 1.5 g twice daily for 5 days and discharged on the 5th post-operative day. Regular follow-ups were conducted at 2-week intervals, and during these visits, there were no clinical signs of infection, and the surgical site was healthy. During the patient’s initial visit, laboratory tests showed a C-reactive protein level of 6.94 mg/dL (normal range 0–6 mg/dL) and a white blood cell count of 4.83 × 10⁹/L (normal range 4–11 × 10⁹/L). Three months after the initial surgery, the patient presented with a resurfacing raw area and a discharging sinus. On physical examination, he had a left distal radial wound that was 1 × 1 cm, with a sinus. There were blebs with discharge on the surgical site (Fig. 3). The decision was made to perform wound debridement, and the radius fracture site was exposed by our plastic surgery colleague, with careful attention to the radius fracture area.
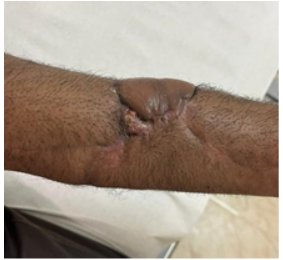
Figure 3: Presented with a resurfacing raw area and a discharging sinus.
Multiple samples were taken from both the wound site and the implant for culture and sensitivity testing. After thorough wound irrigation, the implant was retained, and the wound was closed in layers. (Table 1)

Table 1: Microbiological findings – organism and source
The culture results revealed E. hermannii as the sole pathogen, with no other organisms detected. Based on the sensitivity report, the patient was treated with intravenous meropenem for one week and subsequently placed on oral ciprofloxacin for 11 weeks. (Table 2 and Fig. 4)
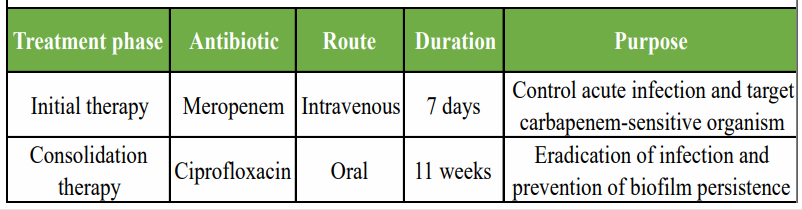
Table 2: Suggested antibiotic treatment protocol used in the case
At the end of antimicrobial therapy, the surgical site was healthy, there were no signs of infection, and the supervised rehabilitation resulted in full movement at the elbow and wrist.
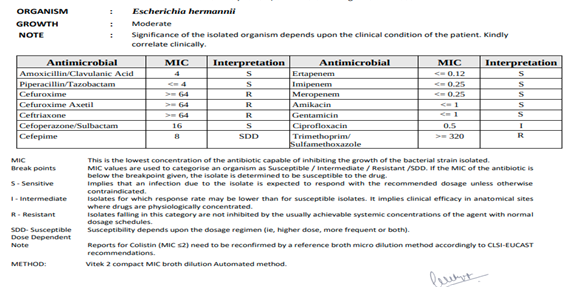
Figure 4: Culture and sensitivity report.
In this report, we describe a case involving a patient with an open radial shaft fracture that progressed to non-union due to infection by E. hermannii. Originally thought to be part of an E. coli-like biogroup, E. hermannii was identified as a distinct species in 1982 and belongs to the Enterobacteriaceae family [4]. It is notable for producing a characteristic yellow pigment and shares only 35% to 45% genetic similarity with E. coli. This organism is typically isolated from environmental sources but is occasionally found in wound, respiratory, urinary, and stool samples [5]. There is increasing evidence in the literature that E. hermannii infections are becoming more frequent. This pathogen has been reported as the sole cause of infections such as urinary tract infections [6], central nervous system infections, abdominal infections, and soft-tissue infections in 17 patients and in a case of open tibial fracture [1,7]. However, there are no cases of crush injury-nonunion radial shaft fracture reported for the same after an extensive search in the literature. A systematic review of patients with monomicrobial E. hermannii infections revealed that treatment primarily involved cephalosporins, aminoglycosides, quinolones, and piperacillin-tazobactam [1]. Treatment also included surgical drainage and removal of central venous catheters when present. Many affected patients were immunocompromised, suffering from conditions such as chronic kidney disease on dialysis, organ transplants, Acquired Immunodeficiency Syndrome, malignancies on chemotherapy, or had central venous catheters, all of which predisposed them to infection [8]. What makes this case unique is that it involves an immunocompetent patient who developed osteomyelitis due to an open fracture, despite not having any of the typical risk factors. The patient most likely contracted this organism from environmental exposure.
The patient’s treatment included surgical debridement and hardware removal to reduce biofilm burden. In this case, only surgical debridement was performed, and the hardware was left in place. Biofilm formation, particularly on orthopedic devices, has been discussed in the literature as a key factor in the persistence of infections, making eradication challenging [6,9]. Biofilm formation on medical devices can impair their function and negatively affect biological processes like osseointegration and healing [10]. This organism is inherently resistant to penicillin, ampicillin, and carbenicillin due to its beta-lactamase production. In addition to surgical management, the patient received a 6-week course of intravenous ceftriaxone, which successfully resolved the infection [11]. However, in our case, culture and sensitivity came back: cephalosporin resistant and meropenem sensitive. Hence, the patient received 1 week of intravenous meropenem followed by 11 weeks of oral ciprofloxacin, which resolved the infection.
This case demonstrates that E. hermannii, although rare, should be recognized as a potential cause of implant-associated infections in orthopedic trauma. Prompt identification through culture, awareness of its resistance profile, and timely surgical and targeted antibiotic management can lead to complete resolution and good functional outcomes, even with implant retention.
Orthopedic implant-associated infections can occasionally be caused by uncommon organisms such as E. hermannii, even in patients without immunocompromising conditions. Failure to recognize these rare pathogens may delay appropriate treatment and compromise outcomes. Clinicians should maintain a high index of suspicion for unusual bacteria in cases of persistent infection or non-union, especially when standard empirical therapy fails. Comprehensive culture and sensitivity testing are critical to identify the causative organism and its resistance pattern, as E. hermannii may show resistance to commonly used antibiotics like cephalosporins. Management should involve timely and thorough surgical debridement to reduce biofilm burden, along with a tailored antibiotic regimen guided by sensitivity results. With prompt, pathogen-specific intervention, complete infection resolution and favorable functional recovery are achievable, even when implant retention is necessary.
References
- 1. Ioannou P. Escherichia hermannii Infections in Humans: A Systematic Review. Trop Med Infect Dis. 2019 Jan 21;4(1):17. doi: 10.3390/tropicalmed4010017. PMID: 30669559; PMCID: PMC6473853. [Google Scholar] [PubMed] [CrossRef]
- 2. Brenner D.J., Davis B.R., Steigerwalt A.G., Riddle C.F., McWhorter A.C., Allen S.D., Farmer J.J., Saitoh Y., Fanning G.R. Atypical biogroups of Escherichia coli found in clinical specimens and description of Escherichia hermannii sp. nov. J. Clin. Microbiol. 1982;15:703–713. doi: 10.1128/jcm.15.4.703-713.1982 [Google Scholar] [PubMed] [CrossRef]
- 3. Timsit JF, Ruppé E, Barbier F, Tabah A, Bassetti M. Bloodstream infections in critically ill patients: an expert statement. Intensive Care Med. 2020 Feb;46(2):266-284. doi: 10.1007/s00134-020-05950-6. Epub 2020 Feb 11. PMID: 32047941; PMCID: PMC7223992. [Google Scholar] [PubMed] [CrossRef]
- 4. F.D., Shrum S., Swenson J.M., Hill B.C., Thornsberry C., Farmer J.J. Colonization of human wounds by Escherichia vulneris and Escherichia hermannii. J. Clin. Microbiol. 1985;22:283–285. doi: 10.1128/jcm.22.2.283-285.1985. [Google Scholar] [PubMed] [CrossRef]
- 5. Dahl K.M., Barry J., DeBiasi R.L. Escherichia hermannii infection of a cephalohematoma: case report, review of the literature, and description of a novel invasive pathogen. Clin Infect Dis. 2002;35:e96–e98. doi: 10.1086/342304. [Google Scholar] [PubMed] [CrossRef]
- 6. Haasdijk RA, van Ingen J. Escherichia hermannii as the sole pathogen in urosepsis: case report. New Microbes New Infect. 2017 Nov 22;21:100-101. doi: 10.1016/j.nmni.2017.11.005. PMID: 29379606; PMCID: PMC5785909. [Google Scholar] [PubMed] [CrossRef]
- 7. Compton J, Wynn M, Willey MC, Sekar P. Escherichia hermannii as the sole cause of osteomyelitis in a patient with an open tibial shaft fracture. BMJ Case Rep. 2019 Nov 26;12(11):e231206. doi: 10.1136/bcr-2019-231206. PMID: 31776149; PMCID: PMC6887438. [Google Scholar] [PubMed] [CrossRef]
- 8. Sedlock C, Tokarczyk M, Sternlieb M, Flomenberg P. PICC-associated infection with Escherichia hermannii: a case report and review of the literature. IDCases. 2018;13:e00444. 10.1016/j.idcr.2018.e00444 [Google Scholar] [PubMed] [CrossRef]
- 9. Rank CU, Lommer Kristensen P, Schrøder Hansen D, et al. . Catheter related Escherichia hermannii sepsis in a haemodialysis patient. Open Microbiol J 2016;10:1–3. 10.2174/1874285801610010001 [Google Scholar] [PubMed] [CrossRef]
- 10. Veerachamy S, Yarlagadda T, Manivasagam G, et al. . Bacterial adherence and biofilm formation on medical implants: a review. Proc Inst Mech Eng H 2014;228:1083–99. 10.1177/0954411914556137 [Google Scholar] [PubMed] [CrossRef]
- 11. Choudhury S, Seet C. Escherichia hermannii bloodstream infection in a long-term haemodialysis patient. Pathology 2013;45:531 10.1097/PAT.0b013e3283633fd7 [Google Scholar] [PubMed] [CrossRef]