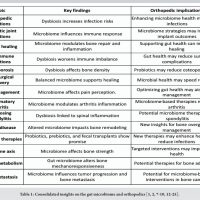
Emerging evidence highlights a strong association between gut microbiome dysbiosis and fibromyalgia, suggesting microbial alterations may contribute to pain modulation, immune dysregulation, and central sensitization.
Dr. Madhan Jeyaraman, Department of Orthopaedics, ACS Medical College and Hospital, Dr. MGR Educational and Research Institute, Chennai, Tamil Nadu, India & Department of Regenerative Medicine, Agathisha Institute of Stemcell and Regenerative Medicine (AISRM), Chennai, Tamil Nadu, India. E-mail: madhanjeyaraman@gmail.com
Introduction: Fibromyalgia (FM) is a chronic pain syndrome marked by widespread musculoskeletal pain, fatigue, sleep disturbances, and cognitive dysfunction. Despite extensive research, its pathophysiology remains unclear. Emerging evidence implicates the gut microbiome (GMB) in FM through mechanisms involving pain modulation, immune dysregulation, and neuroinflammation. This review explores the role of gut dysbiosis in FM pathogenesis, focusing on microbial alterations, immune interactions, intestinal permeability, and neurochemical pathways.
Materials and Methods: A systematic search of PubMed, Scopus, and Web of Science was conducted to identify studies published in the last two decades examining the relationship between GMB and FM. Inclusion criteria encompassed original research, systematic reviews, and meta-analyses addressing microbial dysbiosis, immune modulation, and neurochemical alterations in FM. Studies focused solely on treatment interventions were excluded. A narrative synthesis approach was used to integrate findings and highlight mechanistic insights.
Results: FM patients exhibit significant gut microbial dysbiosis, including reduced butyrate-producing bacteria and increased pro-inflammatory species. These alterations are associated with compromised intestinal barrier integrity, systemic immune activation, and elevated pro-inflammatory cytokines. Neurochemical disruptions include serotonin deficiency, gamma-aminobutyric acid/glutamate imbalance, and reduced short-chain fatty acids, contributing to central sensitization and neuroinflammation. Dysregulation of the gut–brain axis and microbial metabolite pathways further exacerbate FM symptoms.
Conclusion: GMB dysbiosis plays a pivotal role in FM pathogenesis through immune activation, intestinal permeability changes, and neurochemical modulation. Understanding these mechanisms may inform future research into microbiome-based biomarkers and therapeutic strategies. While treatment implications are beyond the scope of this review, the findings underscore the potential of targeting microbial pathways in FM management.
Keywords: Fibromyalgia, gut microbiome, dysbiosis, microbial metabolites.
Fibromyalgia (FM) is a chronic and debilitating disorder characterized by widespread musculoskeletal pain, persistent fatigue, cognitive disturbances (commonly referred to as “fibro fog”), and sleep disorders. These symptoms significantly impact the quality of life, and their underlying pathophysiology remains poorly understood. Despite extensive research, no single etiology has been identified, suggesting a complex, multifactorial origin involving genetic predisposition, environmental triggers, central and peripheral nervous system alterations, and immune dysfunction [1]. Recent studies have highlighted the potential involvement of the gut microbiome (GMB) in FM pathogenesis through various biological pathways. GMB, a diverse community of microorganisms residing in the gastrointestinal tract, plays a crucial role in maintaining metabolic homeostasis, immune regulation, and neural communication. Emerging evidence suggests that microbial dysbiosis – characterized by alterations in microbial composition and diversity – may contribute to systemic inflammation, heightened pain sensitivity, and neuroimmune interactions observed in FM [2,3,4]. Furthermore, disturbances in gut–brain communication through the vagus nerve and microbial metabolites such as short-chain fatty acids (SCFAs) and tryptophan derivatives may influence central sensitization, a hallmark feature of FM. Immune activation driven by microbial translocation and increased intestinal permeability, commonly referred to as “leaky gut,” has also been implicated in FM pathophysiology [5,6,7]. This review synthesizes current literature on the GMB’s role in FM, with an emphasis on its pathomechanical aspects. By elucidating the connections between gut dysbiosis, immune modulation, and neurochemical alterations, this review aims to provide a comprehensive understanding of how microbial factors contribute to FM, ultimately paving the way for future research into novel diagnostic and preventive strategies [8,9,10].
A systematic search of electronic databases, including PubMed, Scopus, and Web of Science, was conducted to identify relevant articles published in the last two decades. The search strategy was designed to encompass a comprehensive range of studies exploring the relationship between the GMB and FM. Keywords and medical subject headings terms related to “fibromyalgia,” “gut microbiome,” “microbial dysbiosis,” “immune modulation,” “intestinal permeability,” “short-chain fatty acids,” “neurotransmitter alterations,” and “gut–brain axis” were utilized to ensure a broad and inclusive retrieval of literature. The inclusion criteria encompassed original research articles, systematic reviews, and meta-analyses that focused on the GMB, microbial metabolites, immune modulation, and neurochemical alterations in FM patients. Studies utilizing animal models or human cohorts that provided insights into gut dysbiosis, immune-inflammatory markers, microbial metabolic byproducts, and neurochemical interactions relevant to FM were included [11]. Exclusion criteria included studies primarily discussing treatment interventions such as probiotics, antibiotics, or dietary modifications without a significant focus on underlying microbial and immunological mechanisms. Additionally, case reports, conference abstracts, and articles not published in peer-reviewed journals were excluded to maintain the rigor and reliability of the findings [12]. The selected studies were assessed for methodological quality, including sample size, study design, statistical robustness, and reproducibility of findings. A narrative synthesis approach was employed to integrate the existing knowledge, identify gaps in the literature, and highlight key mechanistic insights into the role of gut dysbiosis in FM pathogenesis.
GMB Alterations in FM
Microbial dysbiosis
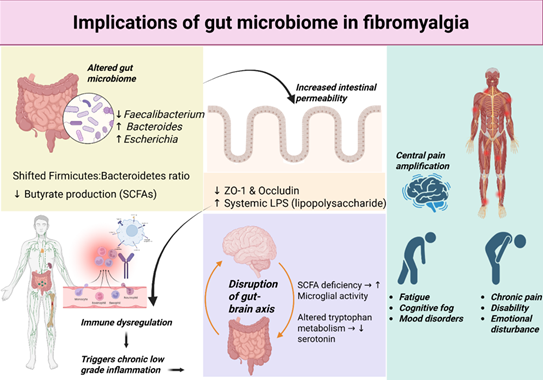
Figure 1: Implications of gut microbiome in fibromyalgia.
Recent studies indicate significant differences in gut microbial composition between FM patients and healthy controls, suggesting that dysbiosis may play a critical role in disease pathogenesis (Fig. 1). Notable findings include:
- Decreased levels of butyrate-producing bacteria (e.g., Faecalibacterium prausnitzii, Roseburia spp.), which are essential for maintaining intestinal barrier integrity, reducing oxidative stress, and modulating systemic inflammation. The reduction in these beneficial bacteria may contribute to increased intestinal permeability, allowing bacterial endotoxins to enter circulation and trigger immune responses [11,12,13].
- Increased abundance of pro-inflammatory bacteria such as Bacteroides and Escherichia species, which are known to produce endotoxins like lipopolysaccharides (LPS). These endotoxins can activate toll-like receptors (TLR4) on immune cells, leading to systemic inflammation, neuroimmune activation, and heightened pain perception – a hallmark of FM pathophysiology [14,15,16,17,18].
- Altered microbial diversity and metabolic function, with shifts in the Firmicutes-to-Bacteroidetes ratio, which has been associated with metabolic dysfunction and immune dysregulation in other chronic pain conditions. These changes may disrupt the production of key microbial metabolites such as SCFAs, neurotransmitters, and bioactive compounds involved in pain modulation [19,20,21].
- Potential involvement of gut-derived neurotoxins, which may influence central sensitization in FM through the gut–brain axis. Dysbiosis-related disruptions in tryptophan metabolism and serotonin synthesis could contribute to mood disturbances and pain hypersensitivity observed in FM patients [22,23,24].
Intestinal barrier dysfunction and immune activation
GMB plays a crucial role in maintaining intestinal barrier integrity by regulating mucosal immunity and epithelial tight junctions. Dysbiosis, characterized by an imbalance in microbial composition, can compromise this barrier function, leading to increased intestinal permeability, commonly referred to as “leaky gut.” This disruption allows the translocation of bacterial endotoxins, such as LPS, into the systemic circulation, initiating immune activation and inflammatory cascades [25]. Key mechanisms involved in this process include:
- Systemic immune activation through LPS, which interacts with TLR4 on immune cells, triggers the release of pro-inflammatory mediators and perpetuates chronic low-grade inflammation. This sustained immune response may contribute to heightened pain sensitivity and fatigue commonly observed in FM patients.
- Elevated levels of pro-inflammatory cytokines, including tumor necrosis factor-alpha, interleukin-6 (IL-6), and IL-1 beta. These cytokines have been found to correlate with pain hypersensitivity, neuroinflammation, and central sensitization – key hallmarks of FM pathophysiology [26].
- Disruption of tight junction proteins, such as zonula occludens-1 and occludin, which regulate epithelial barrier integrity. A compromised gut barrier leads to the influx of microbial antigens, further exacerbating immune dysregulation and systemic inflammation [27].
- Microglial activation in the central nervous system, which may be driven by peripheral inflammatory signals originating from gut-derived endotoxins. This neuroimmune crosstalk could enhance pain processing and contribute to cognitive dysfunction in FM [28].
Gut–brain axis and neurochemical alterations
Neurotransmitter dysregulation
GMB influences neurotransmitter synthesis and metabolism, playing a vital role in modulating pain perception, mood, and cognitive function. In FM, gut dysbiosis has been linked to disruptions in key neurochemical pathways, contributing to central sensitization and widespread pain [29]. Key mechanisms include:
- Serotonin deficiency: Approximately 90% of serotonin (5-HT) is synthesized in the gut, where microbial metabolites influence its production. Reduced levels of gut-derived serotonin may contribute to mood disorders, sleep disturbances, and heightened pain sensitivity in FM patients. Alterations in tryptophan metabolism, a serotonin precursor, may further exacerbate neurotransmitter imbalances [22,30,31,32,33].
- Gamma-aminobutyric acid (GABA) and glutamate imbalance: The gut microbiota regulates the balance between inhibitory and excitatory neurotransmitters, such as GABA and glutamate. Dysbiosis-related alterations in GABAergic and glutamatergic signaling may contribute to hyperalgesia, anxiety, and impaired pain modulation commonly observed in FM [17,24].
- SCFAs: SCFAs, such as butyrate, propionate, and acetate, are microbial metabolites with neuroactive properties. Butyrate, in particular, has been shown to modulate neuroinflammation, glial cell activation, and blood–brain barrier integrity. Reduced levels of SCFAs may contribute to neuroinflammation and altered central pain processing in FM [14].
- Dysregulation of the gut–brain axis: The vagus nerve serves as a critical communication pathway between the gut and the brain. Altered microbial composition and metabolite production may disrupt vagal signaling, further contributing to autonomic dysfunction, mood disturbances, and pain hypersensitivity in FM patients [29,34].
These findings suggest that GMB alterations play a pivotal role in neurotransmitter dysregulation and central sensitization in FM. Understanding these mechanisms may provide insights into novel diagnostic and therapeutic targets for FM management [19].
Microbial metabolites and neuroinflammation
Microbial metabolites, including SCFAs, tryptophan derivatives, and bile acids, play essential roles in neuroimmune modulation and homeostasis. Dysregulation of these metabolites in FM patients has been implicated in systemic inflammation and neurological dysfunction [15,35]. The following flowchart illustrates the pathological process linking GMB dysbiosis to FM. It depicts key steps including gut permeability changes, systemic inflammation, neuroinflammation, neurotransmitter imbalance, and central sensitization, forming a self-perpetuating cycle. Key mechanisms include:
- Increased microglial activation and neuroinflammation: SCFAs, particularly butyrate, possess anti-inflammatory properties and help maintain blood–brain barrier integrity. A reduction in SCFAs may lead to excessive microglial activation, promoting neuroinflammation and heightened pain sensitivity in FM patients [36].
- Dysregulated hypothalamic-pituitary-adrenal (HPA) axis function: The gut microbiota influences stress response pathways by modulating cortisol production through the HPA axis. Dysbiosis-associated disruptions in microbial metabolites can lead to an exaggerated HPA axis response, contributing to increased stress susceptibility and symptom exacerbation in FM [3].
- Altered tryptophan metabolism: Gut bacteria play a crucial role in metabolizing tryptophan into bioactive compounds, including serotonin and kynurenine. Dysregulation of this pathway in FM may contribute to neuroinflammation, mood disturbances, and central sensitization [37].
- Bile acid imbalance and neuroimmune crosstalk: Bile acids act as signaling molecules influencing gut-liver-brain communication. Altered bile acid metabolism in FM patients may disrupt immune homeostasis, further perpetuating chronic pain and fatigue [28,38].
These findings underscore the significance of microbial metabolites in FM pathophysiology, highlighting potential mechanistic links between gut dysbiosis, neuroinflammation, and systemic dysregulation.
GMB plays a pivotal role in FM pathogenesis through microbial dysbiosis, immune activation, and neurochemical modulation. The interplay between microbial alterations, systemic inflammation, and neurotransmitter imbalances suggests a complex disease mechanism that warrants further exploration. While this review does not address treatment implications, understanding these pathophysiological processes lays a critical foundation for future research into diagnostic and preventive strategies. Identifying microbiome-based biomarkers may aid in early detection and risk assessment, while further studies are needed to establish causal relationships between gut dysbiosis and FM symptoms. Expanding our knowledge of gut microbial contributions to FM could potentially pave the way for novel therapeutic approaches, improving disease management and patient outcomes.
• FM patients exhibit reduced beneficial gut bacteria and increased pro-inflammatory species, which may drive systemic inflammation and pain hypersensitivity.
• Alterations in microbial metabolites influence neurotransmitter balance, contributing to neuroinflammation, mood disturbances, and heightened pain sensitivity.
• Increased intestinal permeability allows bacterial endotoxins to trigger immune-mediated inflammation, perpetuating chronic pain and fatigue in FM.
References
- 1. Häuser W, Ablin J, Fitzcharles MA, Littlejohn G, Luciano JV, Usui C, et al. Fibromyalgia. Nat Rev Dis Primers 2015;1:15022. [Google Scholar] [PubMed]
- 2. Clauw DJ. Fibromyalgia: A clinical review. JAMA 2014;311:1547-55. [Google Scholar] [PubMed]
- 3. Minerbi A, Gonzalez E, Brereton NJ, Anjarkouchian A, Dewar K, Fitzcharles MA, et al. Altered microbiome composition in individuals with fibromyalgia. Pain 2019;160:2589-602. [Google Scholar] [PubMed]
- 4. Clos-Garcia M, Andrés-Marin N, Fernández-Eulate G, Abecia L, Lavín JL, Van Liempd S, et al. Gut microbiome and serum metabolome analyses identify molecular biomarkers and altered glutamate metabolism in fibromyalgia. EBioMedicine 2019;46:499-511. [Google Scholar] [PubMed]
- 5. Cryan JF, O’Riordan KJ, Cowan CS, Sandhu KV, Bastiaanssen TF, Boehme M, et al. The microbiota-gut-brain axis. Physiol Rev 2019;99:1877-2013. [Google Scholar] [PubMed]
- 6. Cerdá B, Pérez M, Pérez-Santiago JD, Tornero-Aguilera JF, González-Soltero R, Larrosa M. Gut microbiota modification: Another piece in the puzzle of the benefits of physical exercise in health? Front Physiol 2016;7:51. [Google Scholar] [PubMed]
- 7. Liu L, Wu Q, Chen Y, Ren H, Zhang Q, Yang H, et al. Gut microbiota in chronic pain: Novel insights into mechanisms and promising therapeutic strategies. Int Immunopharmacol 2023;115:109685. [Google Scholar] [PubMed]
- 8. O’Mahony SM, Clarke G, Borre YE, Dinan TG, Cryan JF. Serotonin, tryptophan metabolism and the brain-gut-microbiome axis. Behav Brain Res 2015;277:32-48. [Google Scholar] [PubMed]
- 9. Wang Z, Jiang D, Zhang M, Teng Y, Huang Y. Causal association between gut microbiota and fibromyalgia: A Mendelian randomization study. Front Microbiol 2024;14:1305361. [Google Scholar] [PubMed]
- 10. Garofalo C, Cristiani CM, Ilari S, Passacatini LC, Malafoglia V, Viglietto G, et al. Fibromyalgia and irritable bowel syndrome interaction: A possible role for gut microbiota and gut-brain axis. Biomedicines 2023;11:1701. [Google Scholar] [PubMed]
- 11. Kelly JR, Kennedy PJ, Cryan JF, Dinan TG, Clarke G, Hyland NP. Breaking down the barriers: The gut microbiome, intestinal permeability and stress-related psychiatric disorders. Front Cell Neurosci 2015;9:392. [Google Scholar] [PubMed]
- 12. Fasano A. Leaky gut and autoimmune diseases. Clin Rev Allergy Immunol 2012;42:71-8. [Google Scholar] [PubMed]
- 13. Vujkovic-Cvijin I, Dunham RM, Iwai S, Maher MC, Albright RG, Broadhurst MJ, et al. Dysbiosis of the gut microbiota is associated with HIV disease progression and tryptophan catabolism. Sci Transl Med 2013;5:193ra91. [Google Scholar] [PubMed]
- 14. Rea K, Dinan TG, Cryan JF. The microbiome: A key regulator of stress and neuroinflammation. Neurobiol Stress 2016;4:23-33. [Google Scholar] [PubMed]
- 15. Dinan TG, Cryan JF. Brain-gut-microbiota axis and mental health. Psychosom Med 2017;79:920-6. [Google Scholar] [PubMed]
- 16. Minerbi A, Khoutorsky A, Shir Y. Decoding the connection: Unraveling the role of gut microbiome in fibromyalgia. Pain Rep 2024;10:e1224. [Google Scholar] [PubMed]
- 17. Forsythe P, Kunze WA, Bienenstock J. On communication between gut microbes and the brain. Curr Opin Gastroenterol 2012;28:557-62. [Google Scholar] [PubMed]
- 18. Piccirillo CA. Regulatory T cells in health and disease. Cytokine 2008;43:395-401. [Google Scholar] [PubMed]
- 19. Haroon E, Raison CL, Miller AH. Psychoneuroimmunology meets neuropsychopharmacology: Translational implications of the impact of inflammation on behavior. Neuropsychopharmacology 2012;37:137-62. [Google Scholar] [PubMed]
- 20. D’Mello C, Swain MG. Immune-to-brain communication pathways in inflammation-associated sickness and depression. Curr Top Behav Neurosci 2017;31:73-94. [Google Scholar] [PubMed]
- 21. Erny D, Hrabě De Angelis AL, Prinz M. Communicating systems in the body: How microbiota and microglia cooperate. Immunology 2017;150:7-15. [Google Scholar] [PubMed]
- 22. Cani PD, Everard A, Duparc T. Gut microbiota, enteroendocrine functions and metabolism. Curr Opin Pharmacol 2013;13:935-40. [Google Scholar] [PubMed]
- 23. Van De Wouw M, Schellekens H, Dinan TG, Cryan JF. Microbiota-gut-brain axis: Modulator of host metabolism and appetite. J Nutr 2017;147:727-45. [Google Scholar] [PubMed]
- 24. Braniste V, Al-Asmakh M, Kowal C, Anuar F, Abbaspour A, Tóth M, et al. The gut microbiota influences blood-brain barrier permeability in mice. Sci Transl Med 2014;6:263ra158. [Google Scholar] [PubMed]
- 25. Sharon G, Garg N, Debelius J, Knight R, Dorrestein PC, Mazmanian SK. Specialized metabolites from the microbiome in health and disease. Cell Metab 2014;20:719-30. [Google Scholar] [PubMed]
- 26. Waterman A, Doumas SA, Fischer M, Mattar M, Charbel S, Jennings J, et al. Uncovering the hidden link between the aberrant intestinal microbiome and fibromyalgia. Gastroenterol Hepatol (N Y) 2025;21:111-21. [Google Scholar] [PubMed]
- 27. Janssen AW, Kersten S. The role of the gut microbiota in metabolic health. FASEB J 2015;29:3111-23. [Google Scholar] [PubMed]
- 28. Enaud R, Prevel R, Ciarlo E, Beaufils F, Wieërs G, Guery B, et al. The gut-lung axis in health and respiratory diseases: A place for inter-organ and inter-kingdom crosstalks. Front Cell Infect Microbiol 2020;10:9. [Google Scholar] [PubMed]
- 29. Mayer EA, Tillisch K, Gupta A. Gut/brain axis and the microbiota. J Clin Invest 2015;125:926-38. [Google Scholar] [PubMed]
- 30. Zhao Y, Yu YB. Intestinal microbiota and chronic constipation. SpringerPlus 2016;5:1130. [Google Scholar] [PubMed]
- 31. Carding S, Verbeke K, Vipond DT, Corfe BM, Owen LJ. Dysbiosis of the gut microbiota in disease. Microb Ecol Health Dis 2015;26:26191. [Google Scholar] [PubMed]
- 32. Cryan JF, Dinan TG. Mind-altering microorganisms: The impact of the gut microbiota on brain and behaviour. Nat Rev Neurosci 2012;13:701-12. [Google Scholar] [PubMed]
- 33. Luo C, Jian C, Liao Y, Huang Q, Wu Y, Liu X, et al. The role of microglia in multiple sclerosis. Neuropsychiatr Dis Treat 2017;13:1661-7. [Google Scholar] [PubMed]
- 34. Collins SM, Surette M, Bercik P. The interplay between the intestinal microbiota and the brain. Nat Rev Microbiol 2012;10:735-42. [Google Scholar] [PubMed]
- 35. Alroqi FJ, Chatila TA. T regulatory cell biology in health and disease. Curr Allergy Asthma Rep 2016;16:27. [Google Scholar] [PubMed]
- 36. Antonelli A, Bianchi M, Fear E, Giorgi L, Rossi L. Management of fibromyalgia: Novel nutraceutical therapies beyond traditional pharmaceuticals. Nutrients 2025;17:530. [Google Scholar] [PubMed]
- 37. Minerbi A, Fitzcharles MA. Gut microbiome: Pertinence in fibromyalgia. Clin Exp Rheumatol 2020;38 Suppl 123:99-104. [Google Scholar] [PubMed]
- 38. Alveirinho M, Freitas P, Faleiro ML. Role of gut microbiota in metabolic syndrome: A review of recent evidence. Porto Biomed J 2020;5:e105. [Google Scholar] [PubMed]