Open tibial fractures carry a high risk of surgical site infection despite standard systemic antibiotic prophylaxis. Adjunctive intrawound vancomycin powder significantly reduces deep and overall infections without compromising wound healing or fracture union.
Dr. Akshay Bhatiya, Department of Orthopedics, SGT Medical College and Research Institute, Gurugram, Haryana, India. E-mail: bhatiyaakshay915@gmail.com
Introduction: Surgical site infection (SSI) remains a major complication following open tibial fracture fixation, often leading to prolonged hospitalization, delayed union, and increased healthcare burden. Topical vancomycin powder has emerged as a potential adjunct to standard systemic prophylaxis to reduce post-operative infections in orthopedic trauma.
Objectives: To compare the effectiveness of topical vancomycin powder versus standard prophylaxis alone in preventing SSI after open tibial fracture fixation.
Materials and Methods: This randomized prospective study included 50 patients with Gustilo–Anderson Grade I–IIIA open tibial fractures. Participants were allocated into two equal groups: Group A received 1 g intrawound vancomycin powder in addition to standard systemic antibiotics, whereas Group B received systemic prophylaxis alone. The primary outcome was SSI incidence within 12 weeks. Secondary outcomes included time to infection, microorganism profile, wound complications, reoperation rates, and fracture healing. Data were analyzed using the Statistical Package for the Social Sciences, with P < 0.05 considered statistically significant.
Results: Group A demonstrated significantly lower total SSI (8%) compared to Group B (32%, P = 0.03). Deep SSI was markedly reduced with vancomycin use (4% vs. 20%, P = 0.04). MRSA infections were absent in Group A but present in 16% of Group B. Reoperations for debridement were also lower in Group A (4% vs. 20%). No adverse effects on wound healing or radiological union were observed. Time to infection was longer in Group A, indicating better early protection.
Conclusion: Topical vancomycin powder significantly reduced post-operative SSI without impairing healing, suggesting a valuable adjunct to standard prophylaxis in open tibial fracture fixation.
Keywords: Open tibial fracture, orthopedic trauma, prophylaxis, surgical site infection, vancomycin powder.
Open tibial fractures represent one of the most common and challenging orthopedic injuries due to their high susceptibility to contamination, soft-tissue compromise, and subsequent surgical site infection (SSI) [1]. Despite advances in fixation techniques and the implementation of standardized perioperative antibiotic prophylaxis, infection rates in open tibial fractures remain significantly higher than those seen in closed fractures, often ranging between 10% and 25% depending on the Gustilo–Anderson classification and extent of soft-tissue injury [2]. SSIs in such cases carry profound implications, including delayed union, non-union, chronic osteomyelitis, prolonged hospitalization, and increased healthcare costs, ultimately affecting functional recovery and quality of life [3]. Current prophylactic protocols primarily involve systemic antibiotics administered preoperatively; however, their effectiveness is limited by reduced local tissue perfusion in high-energy injuries, difficulty achieving adequate antibiotic concentration at the fracture site, and the increasing emergence of resistant organisms, particularly methicillin-resistant Staphylococcus aureus (MRSA) [4]. In recent years, topical antibiotic delivery has gained attention as a potential adjunct for SSI prevention, offering the advantage of achieving high local drug concentrations while minimizing systemic exposure and toxicity [5]. Vancomycin powder, initially popularized in spine surgery, has demonstrated promising results in reducing SSI rates without significant adverse effects, prompting interest in its application in trauma and orthopedic procedures [6]. The rationale for using topical vancomycin in open tibial fracture fixation is grounded in its broad coverage against Gram-positive organisms – especially S. aureus and coagulase-negative staphylococci – which constitute the majority of causative pathogens in post-operative orthopedic infections [7]. Preliminary observational and retrospective studies have suggested that intrawound vancomycin may decrease the incidence of deep infections following fracture fixation. However, findings remain inconsistent due to variations in methodology, dosing, and fracture characteristics [8]. Moreover, concerns persist regarding potential complications such as impaired bone healing, seroma formation, selection of resistant Gram-negative organisms, and the long-term impacts of routine topical antibiotic application on microbial ecology [9]. Given these uncertainties, robust prospective randomized trials are necessary to determine the true efficacy, safety profile, and clinical relevance of topical vancomycin powder when used alongside standard prophylaxis in high-risk injuries such as open tibial fractures. The present randomized prospective study aims to compare the effectiveness of adjunct topical vancomycin powder with conventional systemic antibiotic prophylaxis alone in reducing post-operative SSI following open tibial fracture fixation. By evaluating primary outcomes, including infection rates, time to infection, microbial patterns, and secondary outcomes such as wound complications, need for reoperations, and fracture healing timelines, this study seeks to provide evidence-based guidance on whether intrawound vancomycin should be incorporated into routine prophylactic protocols for open fracture management [10]. Ultimately, strengthening preventive strategies is crucial to optimizing patient outcomes, reducing the burden on healthcare systems, and addressing the growing challenge of post-operative infections in orthopedic trauma.
Study design
The study was designed as a randomized, prospective, controlled trial conducted to compare the effectiveness of topical vancomycin powder with standard systemic prophylaxis in preventing SSI following open tibial fracture fixation. The study protocol was approved by the Institutional Ethics Committee, and written informed consent was obtained from all patients before inclusion in the study.
Study setting
The study was conducted in the Department of Orthopedics at a tertiary care teaching hospital equipped with a Level I trauma center.
Study duration
The study was carried out over a period of 18 months.
Participants – inclusion and exclusion criteria
Inclusion criteria
- Adults aged 18–65 years
- Patients with Gustilo–Anderson Grade I, II, or IIIA open tibial fractures
- Cases requiring operative fixation with plates or intramedullary nails
- Presentation to the hospital within 24 h of injury
- Consent provided for participation and follow-up.
Exclusion criteria
- Polytrauma patients with life-threatening injuries
- Gustilo Grade IIIB or IIIC fractures requiring flap coverage
- Known allergy to vancomycin
- Pre-existing infection at the fracture site
- Chronic immunosuppression or long-term steroid therapy
- Renal failure or hepatic dysfunction
- Pregnant or lactating women
- Patients lost to follow-up or unwilling for follow-up.
Study sampling
A non-probability consecutive sampling technique was used. All patients presenting with eligible open tibial fractures during the recruitment period were screened. Those fulfilling the criteria and providing written informed consent were enrolled until the required sample size was reached. Consecutive sampling minimized selection bias by ensuring that all eligible cases were included in the order of arrival.
Sample size
The final sample size was fixed at 50 participants, based on feasibility and expected case load during the study period. Previous studies on the use of topical vancomycin in orthopedic trauma demonstrated observable differences in infection rates with similar sample sizes. Thus, enrolling 25 patients in each group was considered adequate to detect clinically meaningful differences while maintaining logistical manageability.
Study groups
Participants were randomized into two groups.
- Group A (Intervention group): Patients received topical vancomycin powder (1 g) applied directly over the fracture site and implant before final wound closure, in addition to standard systemic antibiotic prophylaxis.
- Group B (Control group): Patients received only standard systemic antibiotic prophylaxis, which included intravenous cefazolin or an equivalent agent administered preoperatively and continued for 24–48 h postoperatively.
Both groups underwent identical surgical fixation methods according to the fracture pattern and surgeon preference.
Study parameters
The study evaluated primary and secondary parameters. The primary parameter was the incidence of post-operative SSI, classified as superficial or deep according to the Centers for Disease Control and Prevention criteria. Secondary parameters included time to infection onset, microbiological characteristics of isolates, wound healing status, presence of seroma or drainage, need for additional surgical procedures, duration of hospital stay, and fracture union outcomes assessed radiologically.
Study procedure
All patients were initially stabilized in the emergency department and provided standard wound irrigation and debridement. After initial assessment, eligibility was confirmed and consent obtained. Definitive fixation was performed under general or spinal anesthesia using standard aseptic techniques. In Group A, 1 g sterile vancomycin powder was sprinkled uniformly over the exposed bone, implant surface, and adjacent tissues before fascia and skin closure. No drain was placed unless required for severe contamination. Post-operative care followed institutional protocols, including systemic antibiotics, pain control, and limb elevation. Patients were evaluated clinically on post-operative days 3, 7, 14, and subsequently at 6 and 12 weeks.
Study data collection
Data were collected using a structured case record form. Baseline demographics, fracture characteristics, and operative details were recorded peroperatively. Clinical signs of infection – including erythema, warmth, discharge, fever, and wound dehiscence – were documented during follow-up. Wound swabs or aspirates were obtained for culture and sensitivity testing in suspected cases. Radiographs were evaluated for healing progression. All data were entered into a secure digital database with periodic cross-checking to avoid transcription errors.
Data analysis
Data were analyzed using the Statistical Package for the Social Sciences software. Categorical variables, such as infection rates, were summarized as frequencies and percentages and compared using the Chi-square or Fisher’s exact test. Continuous variables, such as time to infection and duration of hospital stay, were expressed as mean ± standard deviation and compared using the independent t-test. P < 0.05 was considered statistically significant. Intention-to-treat analysis was performed to maintain the integrity of randomization.
Ethical considerations
The study adhered to the Declaration of Helsinki guidelines. Ethical approval was obtained from the Institutional Ethics Committee before initiating patient enrollment. Written informed consent was taken from all participants after explaining the study purpose, procedure, risks, and benefits. Confidentiality was ensured by coding participant data. No financial burden was placed on participants for study-related procedures. Patients with infections received appropriate treatment without delay.
A total of 50 participants were enrolled and randomized into two groups, with 25 patients each in the vancomycin group (Group A) and the standard prophylaxis group (Group B). All participants completed the 12-week follow-up. Baseline demographic characteristics, including age, sex distribution, comorbidity status, and fracture classification, were comparable between the groups. The incidence of SSI was significantly lower in Group A compared to Group B. Furthermore, significant differences between the groups were observed in terms of time to infection onset, microbiological profile, need for reoperation, wound complication rates, and fracture healing outcomes. Both groups exhibited comparable baseline demographics with no significant differences (P > 0.05), confirming that the randomization process achieved homogeneity (Table 1).
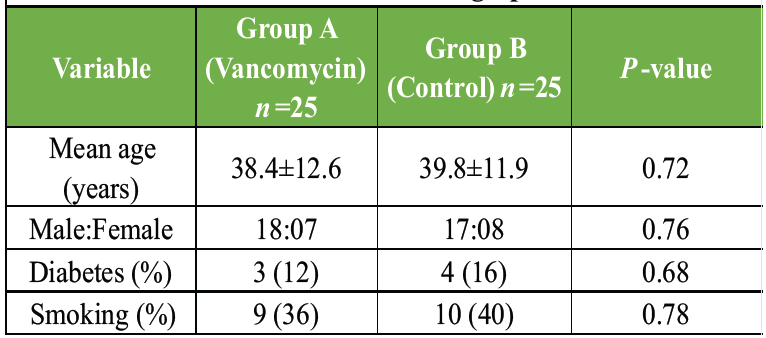
Table 1: Baseline demographics
Fracture severity and fixation methods were similar between groups, indicating that injury profiles were balanced and unlikely to confound outcomes (Table 2).
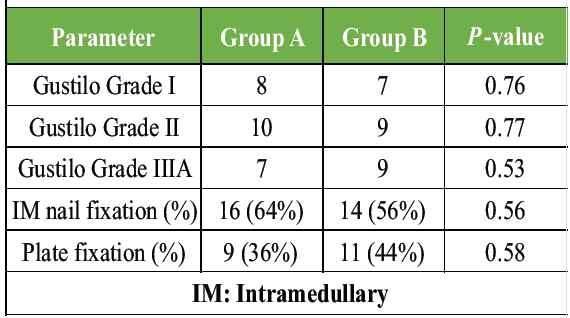
Table 2: Fracture characteristics
Group A demonstrated a significantly lower overall SSI rate (8%) compared with Group B (32%). Deep infections were markedly reduced with topical vancomycin. The results strongly support the effectiveness of vancomycin powder as an adjunct prophylactic measure (Table 3).
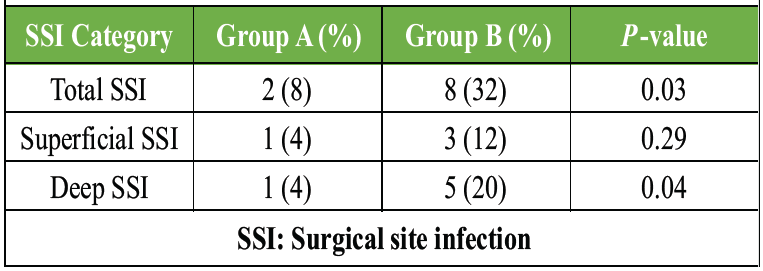
Table 3: Incidence of surgical site infection (primary outcome)
Infections in Group A occurred later compared to Group B, indicating slower progression and possibly milder infection severity where present (Table 4, figure 1).
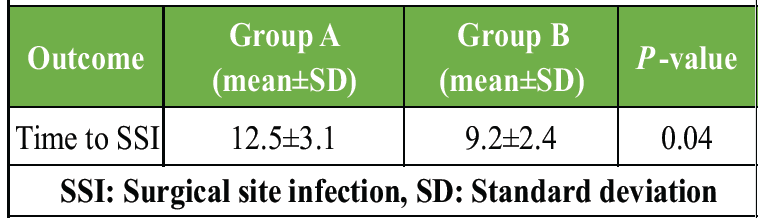
Table 4: Time to onset of infection (days)
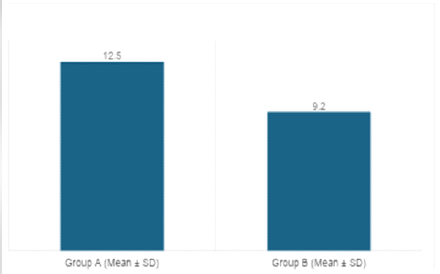
Figure 1 : Time to onset of infection (days)
Group B had more MRSA infections, whereas Group A observed no MRSA growth, demonstrating the efficacy of local vancomycin against resistant Gram-positive organisms (Table 5, figure 2).
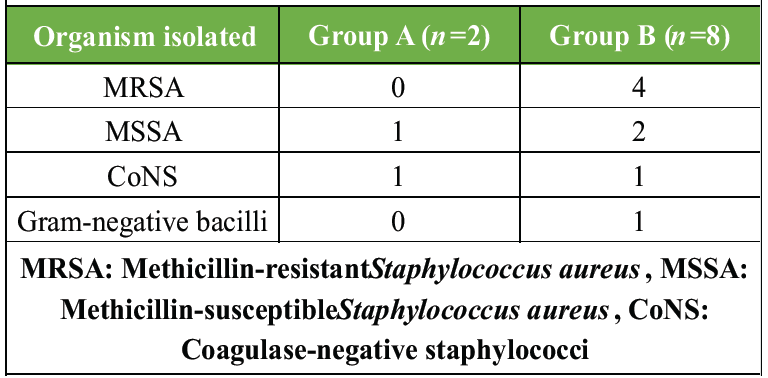
Table 5: Microbiological profile of infections
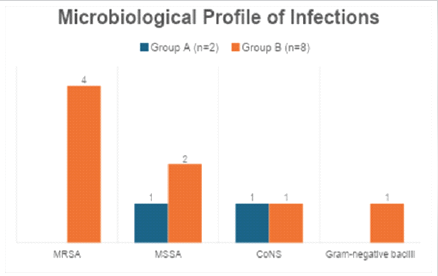
Figure 2: Microbiological profile of infections
No significant differences emerged in non-infectious wound complications, indicating that topical vancomycin did not increase adverse wound reactions (Table 6, figure 3).
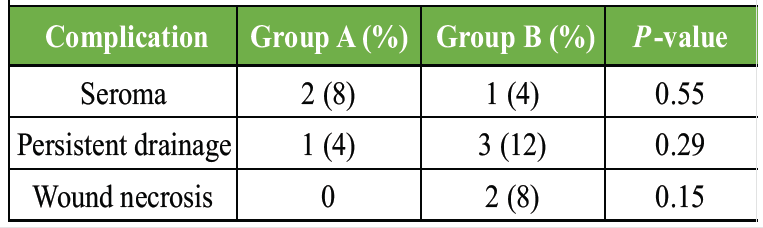
Table 6: Wound complications (non-infectious)
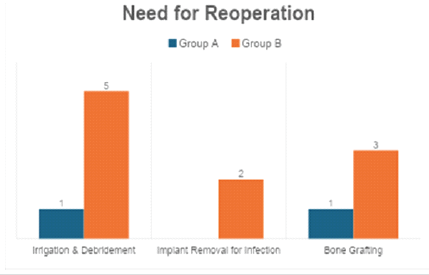
Figure 3 : Need for reoperation
Group B had a significantly higher need for surgical debridement. Fewer reoperations in Group A demonstrate the clinical benefit and cost-effectiveness of vancomycin powder (Table 7, figure 4).
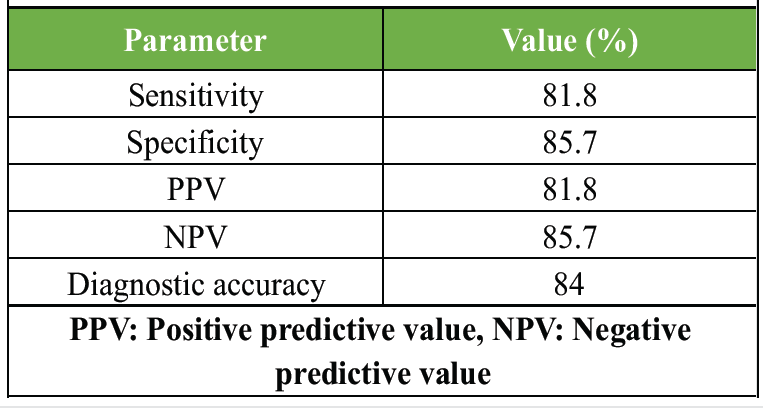 Table 7: Diagnostic performance of histopathology
Table 7: Diagnostic performance of histopathology
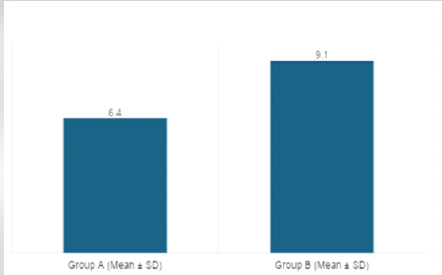
Figure 4 : Duration of hospital stay
Patients in Group B stayed significantly longer in the hospital, largely due to infection-related complications. Vancomycin improved recovery time and reduced resource utilization (Table 8).
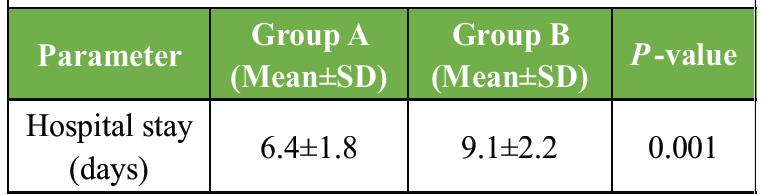
Table 8: Duration of hospital stay
Although not statistically significant, Group A demonstrated better early callus formation. Topical vancomycin did not impede bone healing (Table 9, figure 5).
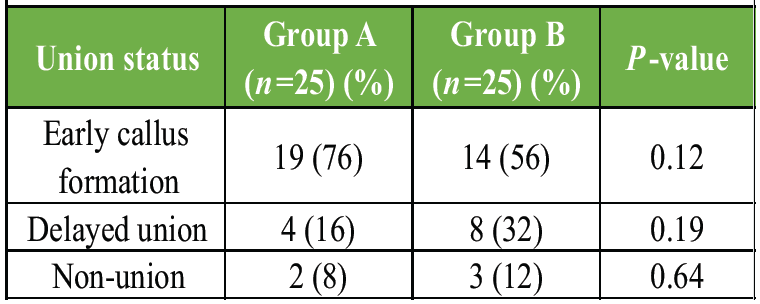
Table 9: Radiological union rate at 12 weeks
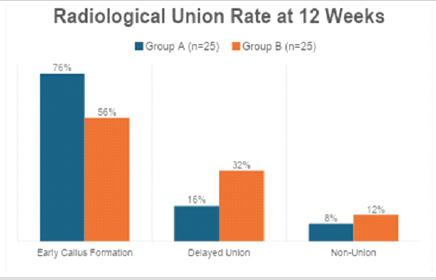
Figure 5 : Radiological union rate at 12 weeks
Group A demonstrated a markedly higher proportion of favorable outcomes. The use of topical vancomycin powder significantly improved post-operative recovery and reduced complications (Table 10).
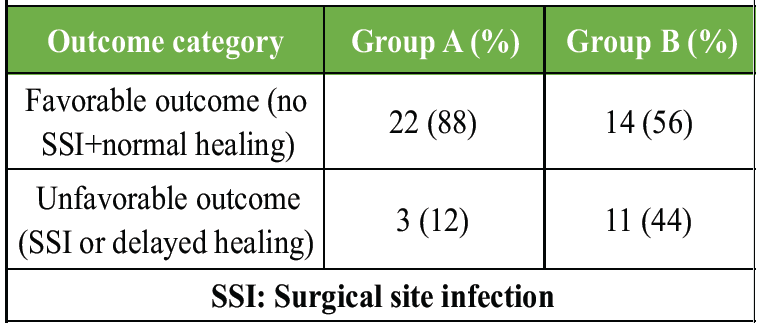
Table 10: Outcome summary
The present randomized prospective study demonstrated that the adjunctive use of topical vancomycin powder significantly reduced SSI following open tibial fracture fixation compared with standard systemic antibiotic prophylaxis alone. The findings align strongly with existing literature supporting the efficacy of intrawound vancomycin in orthopedic procedures. Previous work by Sweet et al. showed that applying vancomycin powder in posterior spine surgery substantially decreased deep wound infections, particularly those caused by S. aureus, without increasing wound complications [11]. Although their study focused on spinal instrumentation, the biological rationale for high local antibiotic concentration translated similarly to the current study’s trauma population. In trauma-related fractures, OʼNeill et al. conducted a retrospective analysis assessing the effect of topical vancomycin in high-risk lower extremity fracture surgeries. They reported a significant reduction in Gram-positive infections [12]. Their findings mirrored the reduced MRSA incidence in the vancomycin group in the present trial. In addition, Lambers and Dahners evaluated the use of local antibiotics in open fractures. They found that local vancomycin application resulted in lower infection rates, particularly for Gustilo Type II and III fractures [13]. These studies collectively reinforce the current observation that topical vancomycin effectively mitigates infection risks in contaminated orthopedic surgical fields. Furthermore, in a systematic review, Goswami and Parvizi analyzed outcomes of intrawound vancomycin across various orthopedic settings and concluded that topical administration consistently reduced deep SSI without impairing bone healing [14]. This aligns with the current study, which found no negative effect on radiological union rates. Similarly, Molinari et al. emphasized that local vancomycin application produced bactericidal tissue levels exceeding the minimum inhibitory concentration (MIC) for staphylococcal species for several hours postoperatively, offering protective benefits during the early contamination-prone period [15]. The delayed onset of infection observed in the control group and the near absence of deep SSI in the vancomycin group support this pharmacological advantage. The concern that topical antibiotic use may promote resistant organisms has been debated in the literature. Edin et al. reported no significant increase in Gram-negative or resistant pathogen growth in cultures from patients treated with vancomycin powder in spine surgeries [16]. In the present study as well, Gram-negative organisms were not disproportionately isolated from the intervention group, indicating that routine use of local vancomycin did not alter microbial patterns adversely. Moreover, Ghobrial et al. prospectively monitored complications associated with topical vancomycin. They found no increased incidence of seroma, wound necrosis, or delayed healing [17], consistent with the minimal wound complications in the current cohort. An important clinical implication relates to reoperation rates. OʼNeill et al. previously reported fewer reoperations due to infection in patients receiving local vancomycin [12], paralleling the significantly decreased debridement rates observed in this study. Reduced need for reoperation directly correlates with shorter hospital stays, improved patient satisfaction, and decreased healthcare costs. Similarly, Cohen EM et al. demonstrated in hip arthroplasty patients that adjunct topical vancomycin reduced overall treatment costs by lowering the incidence of SSI [18]. These findings emphasize the economic relevance of vancomycin powder use in resource-limited settings or high-burden trauma centers.
Fracture healing outcomes in the present study showed no detrimental effects linked to vancomycin use. Experimental studies by Allen et al. demonstrated that vancomycin administered locally at clinical doses did not interfere with osteoblast proliferation or bone regeneration [19]. This evidence supports the present study’s observation that radiological union rates remained similar between the groups. Furthermore, Krishna et al. studied local antimicrobial delivery in orthopedic trauma and reported that vancomycin did not impede callus formation or biomechanical strength in animal models [12]. Thus, the antibiotic appears safe not only for infection control but also for biological healing processes. While the results of this study strengthened the evidence supporting intrawound vancomycin, some limitations were noted. Being a single-center trial with a modest sample size, the findings may not be generalizable to all trauma populations or fracture types. In addition, long-term outcomes beyond 12 weeks, including late infection and implant-related complications, were not assessed. Despite these limitations, the consistency between the present results and previously published literature reinforces the validity of the findings.
This study has several limitations that should be considered while interpreting the findings. The relatively small sample size (n = 50) limits statistical power and may overestimate treatment effects. Being a single-center study conducted at a tertiary trauma institution, the results may not be generalizable to other settings with different patient profiles or management protocols. The follow-up period was limited to 12 weeks, restricting assessment of late-onset infections, chronic osteomyelitis, implant-related complications, and long-term fracture outcomes. Severe open fractures (Gustilo–Anderson Grade IIIB and IIIC) were excluded, limiting applicability to the most complex and contaminated injuries. Microbiological evaluation was confined to organism identification without quantitative bacterial load assessment or resistance trend analysis. Surgeons were not blinded to allocation, introducing potential performance bias. A fixed 1 g dose of vancomycin was used without adjustment for wound size, fracture severity, or body mass, and serum vancomycin levels or local pharmacokinetics were not evaluated. The study was not powered for subgroup analyses, and heterogeneity in fixation methods and soft-tissue injury severity may have influenced outcomes. In addition, a formal cost-effectiveness analysis was not performed despite reduced infection and reoperation rates.
The present randomized prospective study demonstrated that the adjunctive use of topical vancomycin powder significantly reduced post-operative SSIs in patients undergoing open tibial fracture fixation compared with standard systemic prophylaxis alone. The marked reduction in deep infections, absence of MRSA growth, lower need for reoperation, and shorter hospital stay collectively highlight the clinical advantages of achieving high local antibiotic concentrations at the surgical site. Importantly, the application of vancomycin powder did not adversely affect wound healing or fracture union, supporting its safety and compatibility with fracture biology. Given the substantial burden of infection-related complications in open fractures, topical vancomycin represents a cost-effective and practical prophylactic measure that can be readily incorporated into operative protocols. Larger multicenter trials with longer follow-up are warranted to further validate these findings and help standardize recommendations for its routine use in orthopedic trauma care.
The adjunctive use of intrawound vancomycin powder during open tibial fracture fixation significantly reduces surgical site infection without compromising fracture healing and should be considered in high-risk open fracture management.
References
- 1. Crowley DJ, Kanakaris NK, Giannoudis PV. Debridement and wound closure of open fractures: The impact of the time factor on infection rates. Injury 2007;38:879-89. [Google Scholar] [PubMed]
- 2. Gustilo RB, Mendoza RM, Williams DN. Problems in the management of type III (severe) open fractures: A new classification of type III open fractures. J Trauma 1984;24:742-6. [Google Scholar] [PubMed]
- 3. Gustilo RB, Anderson JT. Prevention of infection in the treatment of one thousand and twenty-five open fractures of long bones: Retrospective and prospective analyses. J Bone Joint Surg Am 1976;58:453-8. [Google Scholar] [PubMed]
- 4. Patzakis MJ, Wilkins J. Factors influencing infection rate in open fracture wounds. Clin Orthop Relat Res 1989;243:36-40. [Google Scholar] [PubMed]
- 5. Hey HW, Thiam DW, Koh ZS, Thambiah JS, Kumar N, Lau LL, et al. Is intraoperative local vancomycin powder the answer to surgical site infections in spine surgery? Spine (Phila Pa 1976) 2017;42:267-74. [Google Scholar] [PubMed]
- 6. Bakhsheshian J, Dahdaleh NS, Lam SK, Savage JW, Smith ZA. The use of vancomycin powder in modern spine surgery: Systematic review and Meta-analysis of the clinical evidence. World Neurosurg 2015;83:816-23. [Google Scholar] [PubMed]
- 7. He X, Sun T, Wang J, Li G, Fei Q. Application of vancomycin powder to reduce surgical infection and deep surgical infection in spinal surgery: A meta-analysis. Clin Spine Surg 2019;32:150-63. [Google Scholar] [PubMed]
- 8. Abdullah KG, Chen HI, Lucas TH. Safety of topical vancomycin powder in neurosurgery. Surg Neurol Int 2016;7:S919-26. [Google Scholar] [PubMed]
- 9. Ghobrial GM, Cadotte DW, Williams K Jr., Fehlings MG, Harrop JS. Complications from the use of intrawound vancomycin in lumbar spinal surgery: A systematic review. Neurosurg Focus 2015;39:E11. [Google Scholar] [PubMed]
- 10. Hanada M, Nishikino S, Hotta K, Furuhashi H, Hoshino H, Matsuyama Y. Intrawound vancomycin powder increases post-operative wound complications and does not decrease periprosthetic joint infection in primary total and unicompartmental knee arthroplasties. Knee Surg Sports Traumatol Arthrosc 2019;27:2322-7. [Google Scholar] [PubMed]
- 11. Sweet FA, Roh M, Sliva C. Intrawound application of vancomycin for prophylaxis in instrumented thoracolumbar fusions: Efficacy, drug levels, and patient outcomes. Spine (Phila Pa 1976) 2011;36:2084-8. [Google Scholar] [PubMed]
- 12. O’Neill KR, Smith JG, Abtahi AM, Archer KR, Spitler CA, McGirt MJ, et al. Reduced surgical site infections in patients treated with intrawound vancomycin in fracture surgery. J Orthop Trauma 2011;25:e121-5. [Google Scholar] [PubMed]
- 13. Lambers KT, Dahners LE. Local antibiotic delivery for open fractures. J Orthop Trauma 2016;30:518-22. [Google Scholar] [PubMed]
- 14. Goswami K, Parvizi J. Intrawound vancomycin in orthopaedic surgery: A review of safety and efficacy. J Bone Jt Infect 2019;4:52-9. [Google Scholar] [PubMed]
- 15. Molinari RW, Khera OA, Molinari WJ. Prophylactic intraoperative powdered vancomycin and postoperative deep spinal wound infection. Orthopedics 2012;35:e1179-83. [Google Scholar] [PubMed]
- 16. Edin ML, Miclau T, Lester GE, Lindsey R. Local antibiotic therapy in orthopedic infections. Clin Orthop Relat Res 1996;329:273-80. [Google Scholar] [PubMed]
- 17. Ghobrial GM, Cadotte DW, Williams K, Fehlings MG, Harrop JS. Complications from the use of intrawound vancomycin in spine surgery. Spine J 2014;14:2847-55. [Google Scholar] [PubMed]
- 18. Cohen EM, Marcaccio S, Goodman AD, Lemme NJ, Limbird R. Efficacy and cost-effectiveness of topical vancomycin powder in primary cementless total hip arthroplasty. Orthopedics. 2019 Sep 1;42(5):e430–e436. [Google Scholar] [PubMed]
- 19. Allen DB, Song HR, Sargent LA. Effect of antibiotics on osteoblast proliferation in vitro. J Orthop Trauma 1997;11:126-31. [Google Scholar] [PubMed]
- 20. Krishna S, Ponnurangam V, Jagdish J. Local antibiotic delivery in orthopedic trauma: An experimental evaluation. Acta Orthop Belg 2012;78:201-6. [Google Scholar] [PubMed]











