Orthopedicians should lens out rare clinical entities when patients present with musculoskeletal complaints of chronicity.
Dr. Madhan Jeyaraman,
Department of Orthopedics, School of Medical Sciences and Research, Sharda University, Greater Noida 201 306, Uttar Pradesh, India. E-mail: madhanjeyaraman@gmail.com
Introduction: Intrasynovial hemangioma, a rare benign vascular lesion of joint cavities, is also called intrasynovial papillary endothelial hyperplasia or Masson’s hemangioma (MH). These lesions are characterized by abnormal proliferation of endothelial cells with mild atypia with an organized thrombus. MH follows an individualized protocol for its management.
Case Report: A 22-year-old male gave a 10-year history of the diagnosed and operated (arthroscopically) case of synovial hemangioma of the left knee and presented with similar complaints. There was no history of trauma or infection over the left knee. The movements of the left knee were normal except with terminal restriction of movements. MRI of the left knee suggested intrasynovial low flow vascular malformation within the substance of the synovium. The patient underwent synovectomy of the hemangiomatous lesion in toto without any recurrence in the follow-up period for 8 months. Histopathology confirmed the diagnosis of intravascular papillary endothelial hyperplasia/MH.
Conclusion: Although intrasynovial hemangioma is a very rare clinical entity, Orthopedic surgeons should have a high index of suspicion for MH, when a long-standing pain and hemarthrosis of the knee joint is being encountered in the clinical practice.
Keywords: Hemangioma, Intrasynovial, MRI, Excision biopsy.
Intrasynovial hemangioma is a rare benign vascular lesion of joint cavities [1, 2, 3]. They contribute 2–7% of all hemangiomas. They are also called intrasynovial papillary endothelial hyperplasia or Masson’s hemangioma (MH) [4, 5, 6]. These lesions are characterized by abnormal proliferation of endothelial cells with mild atypia with an organized thrombus [7]. The diagnosis of MH is a challenging task as they mimic angiosarcoma and Kaposi’s sarcoma. The combination of radiological, histopathological, and histomorphological modalities aid in clinching the appropriate diagnosis of the lesion [4, 8]. After obtaining informed and written consent from the patient, we quote a rare case of MH and its management in a nutshell manner in view of invoking fundamentals and enhancement of knowledge among all the orthopedic practitioners.
A 22-year-old male gave a history of pain and swelling in the left knee before 10 years for which he got radiologically diagnosed as synovial hemangioma and arthroscopic debridement and biopsy was done. Now the patient resulted in similar swelling and pain in the left knee which was dull aching pain and vague. The pain was aggravated on movements and relieved partially by rest and medications. On examination, the left knee swelling was tender, euthermic with boggy consistency, and synovial hypertrophy. The patient had a painful terminal range of movements of his left knee. The plain radiograph of the left knee was shown in (Fig. 1).
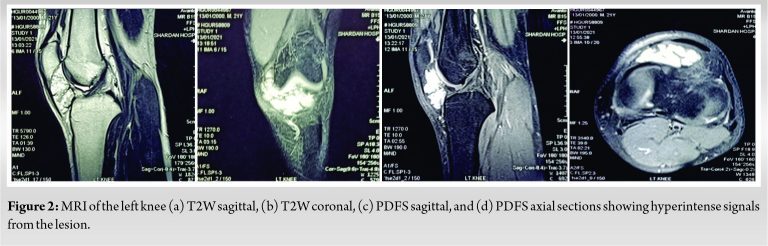
USG of the left knee revealed a well-defined, encapsulated mass of 4.2 × 1.9 cm2 which was hypoechogenic in nature. Color doppler revealed the hypervascular nature of the lesion. MRI scan of the left knee was performed to understand the pathology, which revealed an encapsulated, well-defined, lobulated mass measuring 41 × 23 × 19 mm (approx.) in the substance of synovium lining of the left knee with hyperintense signals on T2W images and hypointense on T1W with a foci of gradient-echo images as shown in (Fig. 2a, 2b). PD-FS sequence shows hyperintense signals from the lesion in the anterior compartment of the left knee as shown in (Fig. 2c, 2d).
Under spinal anesthesia, surgical exploration of the left knee was done and performed excision biopsy of the mass along with the synovium as shown in (Fig. 3a, 3b).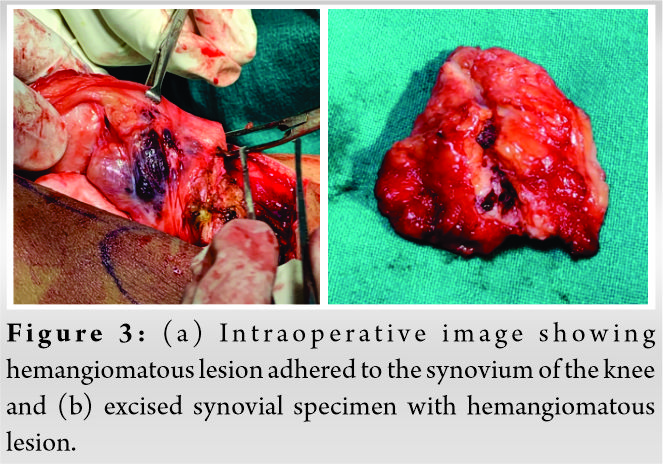
Grossly, the synovium of the left knee was admixed with the vascular tumor measuring 5.8 × 3.2 cm. The cut surface of the mass revealed numerous vascular channels with areas of thrombus and hemorrhages. The histopathological examination of the mass revealed fibrous tissue along with the papillary proliferation of vascular channels and organized thrombus as shown in (Fig. 4a).
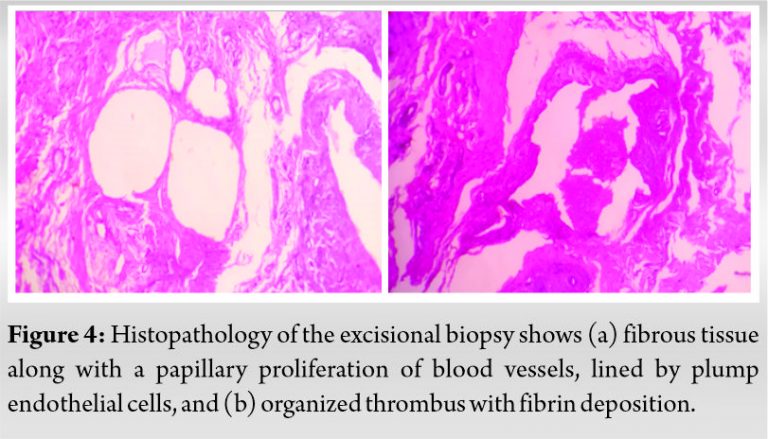
Few cells are dilated and lined by plump endothelial cells which show mild atypia. The histopathology section in (Fig. 4b) shows the papillary proliferation of blood vessels along with deposition of fibrin. Immunohistochemical studies showed positive staining for α-SMA and Ki-67 markers as shown in (Fig. 5a, 5b). The aforementioned findings were suggestive of MH. During the follow-up of 8 months, the patient showed a full range of movements over the left knee without any recurrence of the lesion.
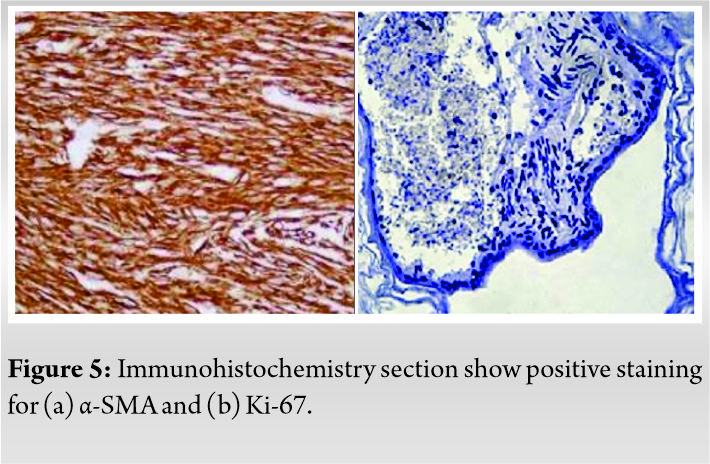
In 1923, Masson’s coined the lesion as “vegetant intravascular hemangioendothelioma” in a patient with ulcerated irreducible hemorrhoids [5, 7, 9]. MH, called intravascular papillary endothelial hyperplasia (IPEH) [6, 10], synovial hemangioma [3], reactive papillary endothelial hyperplasias [8], or intravascular angiomatosis [11, 12], is a rare benign soft tissue lesion of vascular origin which is characterized by hamartomatous proliferation of endothelial cells [7]. Intrasynovial hemangioma contribute for 2–7% of all hemangioams. Hashimoto et al. reported three types of IPEH namely (a) primary IPEH (de novo pattern) (56%), (b) secondary IPEH (secondary to pre-existing vascular lesions) (40%), and (c) extravascular IPEH (secondary to post-traumatic hematoma) (4%) [13]. Although MH lesions are non-neoplastic, they mimic benign or malignant lesions of vascular origin such as pyogenic granuloma, angiosarcoma, and Kaposi’s sarcoma [5, 14, 15]. MH lesions are more commonly seen in females around 30 years of age. They are frequently found in extremities, head and neck, and face but IPEH/MH occurrence in abdominal viscera were reported in the literature [5, 16]. It is hypothesized that IPEH/MH develops when organized thrombus as a result of vascular injury, mediate proliferation of endothelial cellular lining of the vascular channel [17, 18, 19]. There is a temporal association between basic fibroblastic growth factor and the development of IPEH/MH lesion [17, 20]. The molecular pathogenesis in IPEH/MH formation is by the release of endothelial b-FGF by macrophages in the surrounding environment producing a positive feedback loop mechanism of the proliferation of endothelial cells [6, 21]. Various studies have stated that the diagnosis of synovial hemangioma is delayed due to the presence of non-specific symptoms. About 60% of synovial hemangioma occurs in the knee joint usually in the anterior compartment. These hemangiomas present as non-specific synovitis, synovial chondromatosis, giant cell tumor of the tendon sheath, fibrolipoma, lipoma arborescens, or localized villonodular synovitis of the affected knee. The diagnosis of IPEH plays a major diagnostic glitch. A radiograph of the affected knee joint shows no pathological findings other than soft tissue swelling around the knee joint. MRI assists in clenching the diagnosis and aid in planning the management. Sasho et al. studied MRI features of MH/IPEH who described the lesion as iso- or hypointense signals on T1W images and hyperintense signals on T2W images. With gadolinium injection, heterogeneous enhancement of the lesion can be visualized. In our case, MRI of the left knee showed a hyperintense signal from T2W and PD-FS images from the lesion in the anterior compartment of the left knee which was encapsulated, well defined lobulated mass measuring 41 × 23 × 19 mm (approx.) in the substance of synovium lining of the left knee. The diagnosis of IPEH/MH lesions poses a great challenge among pathologists. The biological behavior of IPEH/MH must be differentiated from other lesions of vascular etiology. IPEH/MH is characterized by (a) intraluminal lesion with the intravascular origin, (b) organized thrombus, (c) minimal necrosis with lack of pleomorphism and mitotic figures, and (d) rarely extravascular hematoma [5, 22, 23]. The histomorphological features were consistent with the lack of pleomorphism and mitotic figures, possess mild endothelial cellular atypia, and intraluminal lesion with organized thrombus, which were suggestive of IPEH. Immunohistochemical markers aid in a more precise diagnosis of MH. Akdur et al. demonstrated positive staining for CD-31, -34, and -105, factor 8 and 13a, type 4 collagen, and α-smooth muscle actin in IPEH lesions [17]. In a series of IPEH lesion of the hands, Patai et al. reported positivity for WT1, CD-31, α-SMA, and ERG and negativity for GLUT-1 receptors [24]. In our case, we observed positive staining for ki-67 and α-SMA markers. The management of IPEH/MH depends on the tumor size, extent, behavior, resectability, and the functional need of the individual [25, 26]. They range from intrasynovial instillation of sclerosing agents (sodium tetradecyl sulfate [3% sterol], sodium morrhuate, monoethanolamine oleate, and sodium psylliate) [27], holmium ablation [28], Nd:YAG laser [29], cauterization [30], freezing [30], radiotherapy, embolization [30], and open or arthroscopic synovectomy [31, 32]. In our case, we performed open synovectomy to provide a better functional outcome. The literature stated that inadequate excision poses a recurrence rate of 20–60% [33, 34, 35, 36]. No recurrence was reported in our case in the last 8 months of follow-up.
Although intrasynovial hemangioma is a very rare clinical entity, Orthopedic surgeons should have a high index of suspicion for MH, when a long-standing pain and hemarthrosis of the knee joint is being encountered in the clinical practice. MRI along with the histopathological examination of the biopsied lesion aid in the prompt diagnosis of the MH. Complete excision of MH/IPEH prevents the occurrence of recurrence in the follow-up period.
Orthopedicians should include rare entities under the mindful list of probable diagnosis when patients present with a history of chronic pain in the knee joint with or without hemarthrosis in order to plan best treatment as per patient’s profile and deliver optimum care to subjugate associated complications compromising functional quality of patient’s life.
References
- 1.Mattila KA, Aronniemi J, Salminen P, Rintala RJ, Kyrklund K. Intra-articular venous malformation of the knee in children: Magnetic resonance imaging findings and significance of synovial involvement. Pediatr Radiol 2020;50:509-15. [Google Scholar]
- 2.Arslan H, İslamoğlu N, Akdemir Z, Adanaş C. Synovial hemangioma in the knee: MRI findings. J Clin Imaging Sci 2015;5:23. [Google Scholar]
- 3.Yilmaz E, Karakurt L, Ozdemir H, Serin E, Incesu M. Diffuse synovial hemangioma of the knee: A case report. Acta Orthop Traumatol Turc 2004;38:224-8. [Google Scholar]
- 4.Korkolis DP, Papaevangelou M, Koulaxouzidis G, Zirganos N, Psichogiou H, Vassilopoulos PP. Intravascular papillary endothelial hyperplasia (Masson’s hemangioma) presenting as a soft-tissue sarcoma. Anticancer Res 2005;25:1409-12. [Google Scholar]
- 5.Pountos I, Siddiqui A. Intravascular papillary endothelial hyperplasia (Masson’s tumor) involving the knee synovium. J Orthop Case Rep 2018;8:23-5. [Google Scholar]
- 6.Mahapatra QS, Sahai K, Malik A, Mani NS. Intravascular papillary endothelial hyperplasia: An unusual histopathological entity. Indian Dermatol Online J 2015;6:277-9. [Google Scholar]
- 7.Kumar A, Surowiec S, Nigwekar P, Illig KA. Masson’s intravascular hemangioma masquerading as effort thrombosis. J Vasc Surg 2004;40:812-4. [Google Scholar]
- 8.Guledgud MV, Patil K, Saikrishna D, Madhavan A, Yelamali T. Intravascular papillary endothelial hyperplasia: Diagnostic sequence and literature review of an orofacial lesion. Case Rep Dent 2014;2014:934593. [Google Scholar]
- 9.Kuo T, Sayers CP, Rosai J. Masson’s “vegetant intravascular hemangioendothelioma”: A lesion often mistaken for angiosarcoma: Study of seventeen cases located in the skin and soft tissues. Cancer 1976;38:1227-36. [Google Scholar]
- 10.Boukovalas S, Dillard R, Qiu S, Cole EL. Intravascular papillary endothelial hyperplasia (Masson’s tumor): Diagnosis the plastic surgeon should be aware of. Plast Reconstr Surg Glob Open 2017;5:e1122. [Google Scholar]
- 11.Sasso SE, Naspolini AP, de Boit Milanez T, Suchard G. Masson’s tumor (intravascular papillary endothelial hyperplasia). An Bras Dermatol 2019;94:620-1. [Google Scholar]
- 12.Shah HC, Mittal DH, Shah JK. Intravascular papillary endothelial hyperplasia (Masson’s tumor) of the scalp with intracranial extension. J Pediatr Neurosci 2014;9:260-2. [Google Scholar]
- 13.Hashimoto H, Daimaru Y, Enjoji M. Intravascular papillary endothelial hyperplasia. A clinicopathologic study of 91 cases. Am J Dermatopathol 1983;5:539-46. [Google Scholar]
- 14.Rajagopal S. Intravascular papillary endothelial hyperplasia (Masson’s tumour) of the lower eyelid-a rare tumour of the orbital region. Eur J Plast Surg 2020;43:843-6. [Google Scholar]
- 15.Dunet B, Tournier C, Pallaro J, Boullet F, Fabre T. Arthroscopic treatment of an intra-articular hemangioma in the posterior compartment of the knee. Orthop Traumatol Surg Res 2014;100:337-9. [Google Scholar]
- 16.Gupta A, Chaturvedi S, Jha K, Nazir W. Intravascular papillary endothelial hyperplasia presenting as a cystic mass in the scalp with underlying bone involvement: A rare entity. Int J Appl Basic Med Res 2017;7:269-71. [Google Scholar]
- 17.Akdur NC, Donmez M, Gozel S, Ustun H, Hucumenoglu S. Intravascular papillary endothelial hyperplasia: Histomorphological and immunohistochemical features. Diagn Pathol 2013;8:167. [Google Scholar]
- 18.Han Y, Yun S, Choi IH. Intravascular papillary endothelial hyperplasia occurring in the foot. Medicine (Baltimore) 2019;98:e16391. [Google Scholar]
- 19.D’Aguanno V, Ralli M, de Virgilio A, Greco A, de Vincentiis M. The role of differential diagnosis in intravascular papillary endothelial hyperplasia of the sinonasal cavity mimicking angiosarcoma: A case report. Oncol Lett 2019;17:1253-6. [Google Scholar]
- 20.Lazarous DF, Shou M, Scheinowitz M, Hodge E, Thirumurti V, Kitsiou AN, et al. Comparative effects of basic fibroblast growth factor and vascular endothelial growth factor on coronary collateral development and the arterial response to injury. Circulation 1996;94:1074-82. [Google Scholar]
- 21.Sung KY, Lee S, Jeong Y, Lee SY. Intravascular papillary endothelial hyperplasia of the finger: A case of Masson’s tumor. Case Reports Plast Surg Hand Surg 2021;8:23-6. [Google Scholar]
- 22.Kakhandaki A, Dinesh US, Akash B. Intravascular papillary endothelial hyperplasia as an unusual diagnosis for peri-orbital tumour-a case report. Indian J Ophthalmol 2018;66:163-5. [Google Scholar]
- 23.Milhan Nv, Torquato LC, Costa V, de Marco AC, Carvalho YR, Anbinde AL. A mixed form of intravascular papillary endothelial hyperplasia in an uncommon location: Case and literature review. Dermatol Online J 2018;24(2): :13030/qt2dk039r1. [Google Scholar]
- 24.Patai BB, Peterfy N, Szakacs N, Sapi Z, Hetthessy JR. Papillary endothelial hyperplasia (masson tumor) of the hand. surgical and pathological consideration from seven cases using new vascular markers. Pathol Oncol Res 2020;26:2083-90. [Google Scholar]
- 25.Akgün I, Kesmezacar H, Öğüt T, Dervişoğlu S. Intra-articular hemangioma of the knee. Arthroscopy 2003;19:E17. [Google Scholar]
- 26.Rajni, Khanna G, Gupta A, Gupta V. Synovial hemangioma: A rare benign synovial lesion. Indian J Pathol Microbiol 2008;51:257-8. [Google Scholar]
- 27.Parvathidevi MK, Koppal S, Rukmangada T, Byatnal AR. Management of haemangioma with sclerosing agent: A case report. BMJ Case Rep 2013;2013:bcr2013200660. [Google Scholar]
- 28.Schreiner C, Schleberger R. Synovial hemangioma as a rare differential diagnosis of juvenile hemarthrosis. Therapy by laser ablation. Chirurg 1995;66:1272-4. [Google Scholar]
- 29.Vakil-Adli A, Zandieh S, Hochreiter J, Huber M, Ritschl P. Synovial hemangioma of the knee joint in a 12-year-old boy: A case report. J Med Case Rep 2010;4:105. [Google Scholar]
- 30.Kumar I. Synovial haemangioma of the knee joint diagnosed on magnetic resonance imaging: A rare case report. Menoufia Med J 2019;32:1528-30. Available from: https://www.mmj.eg.net/article.asp?issn=1110-2098;year=2019;volume=32;issue=4;spage=1528;epage=1530;aulast=kumar. [Last accessed on 2021 Apr 11]. [Google Scholar]
- 31.Sanghi AK, Ly JQ, McDermott J, Sorge DG. Synovial hemangioma of the knee: A case report. Radiol Case Rep 2007;2:33-6. [Google Scholar]
- 32.Holzapfel BM, Geitner U, Diebold J, Glaser C, Jansson V, Dürr HR. Synovial hemangioma of the knee joint with cystic invasion of the femur: A case report and review of the literature. Arch Orthop Trauma Surg 2009;129:143-8. [Google Scholar]
- 33.Kumar R, Ranjan R, Jeyaraman M, Chaudhary D, Arora A, Kumar S. Pronator quadratus hemangioma (PQH): A rare case report and review. Indian J Orthop 2021;55:1056-63. [Google Scholar]
- 34.Ly JQ, Sanders TG, Mulloy JP, Soares GM, Beall DP, Parsons TW, et al. Osseous change adjacent to soft-tissue hemangiomas of the extremities: Correlation with lesion size and proximity to bone. Am J Roentgenol 2003;180:1695-700. [Google Scholar]
- 35.Bella GP, Manivel JC, Thompson RC, Clohisy DR, Cheng EY. Intramuscular hemangioma: Recurrence risk related to surgical margins. Clin Orthop Relat Res 2007;459:186-91. [Google Scholar]
- 36.DeGiovanni JC, Simmonds J, Lang-Orsini M, Lee A. Recurrent intramuscular hemangioma (infiltrating angiolipoma) of the lower lip: A case report and review of the literature. Ear Nose Throat J 2020:145561320957759. [Google Scholar]











