With a multidisciplinary approach, timely surgical intervention along with antitubercular therapy can lead to complete neurological recovery while allowing the pregnancy to continue safely.
Hamza Shaikh, Spine Surgery Unit, Manipal Hospital Dwarka Sector, Dwarka, Delhi, India. E-mail: hamza.shaikh@manipalhospitals.com
Introduction: Spinal tuberculosis is primarily managed with antitubercular therapy, while surgical intervention is reserved for cases with neurological compromise or spinal instability. The occurrence of Pott’s paraplegia during pregnancy is rare and presents unique diagnostic and therapeutic challenges due to concerns regarding fetal safety, imaging limitations, and surgical risks. Management during the second trimester is particularly complex, as options, such as medical termination or delivery are not feasible. Only a limited number of case reports describing Pott’s paraplegia during pregnancy exist in the literature, with very few addressing surgical management during the second trimester while continuing the pregnancy.
Case Report: We report the case of a 18 year-old woman of Indian origin, primigravida at 26 weeks of gestation, who presented with acute-onset paraplegia preceded by mild upper thoracic back pain. Neurological examination revealed spastic paraplegia with sensory deficits below the T3 level. Magnetic resonance imaging of the thoracic spine demonstrated pathological collapse of the T2 vertebra with an epidural abscess causing spinal cord compression. In view of acute neurological deterioration, urgent posterior decompression and stabilization were performed with meticulous obstetric and anesthetic pre-cautions to ensure fetal safety. Histopathological examination and Gene Xpert confirmed tuberculous infection, and antitubercular therapy was initiated post-operatively. Neurological recovery was progressive, and the pregnancy was successfully continued. The patient delivered a healthy neonate by cesarean section at 36 weeks of gestation. At 1-year follow-up, the patient had complete neurological recovery with radiological evidence of spinal healing, and the child demonstrated normal developmental milestones.
Conclusion: This case highlights that timely surgical decompression and stabilization for Pott’s paraplegia during the second trimester of pregnancy can be safely performed with a multidisciplinary approach, allowing for neurological recovery and successful continuation of pregnancy. This manuscript is an original case report of particular relevance to orthopedic spine surgery, neurosurgery, obstetrics, and anesthesiology, while also carrying broader clinical significance across medicine.
Keywords: Antitubercle therapy, Pott’s paraplegia, pregnancy, spinal tuberculosis, thoracic spine infection.
The management of tuberculous spine primarily includes a course of anti-tubercular drugs (ATT) for a period of 9–12 months, and surgery is reserved for patients with compromised spine stability or neurological compression [1]. Tuberculous spondylodiscitis during pregnancy complicates the situation; the infection itself and ATT as well have possible adverse effects on the growing fetus and on maternal health, and surgery is to be avoided as far as possible. Moreover, the occurrence of Pott’s paraplegia in pregnancy is rare and only a few cases have been reported in the literature [2,3,4,5,6]. In the majority of the reported cases, paraplegia occurred either in the first or third trimester, which were managed by surgery before or after a medical termination of pregnancy (MTP) or induced delivery or cesarean section, respectively, depending upon the trimester. Reports on the surgical management of Pott’s paraplegia in second-trimester pregnancy are not readily described in the literature. Moreover, the options of either legal MTP or induced labor or cesarean section when the fetus is non-viable cannot be opted during the second trimester. This case report describes the surgical management of a Pott’s paraplegia in a 2nd trimester pregnant lady along with the successful maintenance of the pregnancy. The patient was informed that a clinical summary concerning her case would be submitted for publication, and she provided consent.
A 18-year old primary gravida, with 26 weeks of pregnancy, presented to the emergency department with complaints of mild upper back pain for 3 months and sudden onset of weakness of both legs, making her bedridden for 2 days. The sensations in both the legs and the bladder and bowel control were intact. History of recent trauma, fever, or other constitutional symptoms was absent. On examination, there was no swelling, sinus, or deformity of the spine; however, tenderness was present at the upper dorsal spine. Spasticity was present in both legs. Motor power in both legs was 0/5 Medical Research Council grade, and sensations were reduced bilaterally from D3 level onward. Deep tendon reflexes were exaggerated, with the ankle and patellar clonus bilaterally elicited. Babinski’s sign was positive bilaterally and the abdominal reflex was absent in all four quadrants. An X-ray was not done to avoid radiation exposure. Magnetic resonance imaging (MRI) of the thoracic spine revealed pathological collapse of D2 vertebra and an abscess collection anterior to the vertebral body and in the epidural space, compressing the spinal cord (Fig. 1).
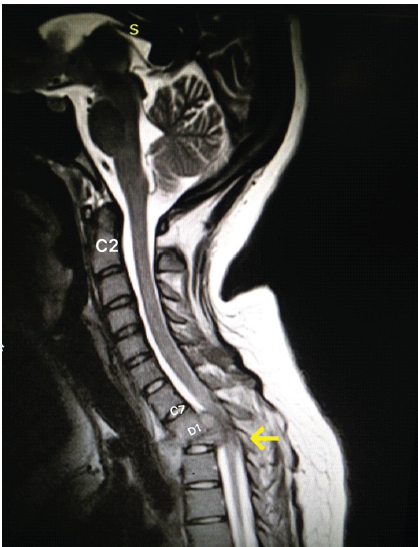
Figure 1: T2W section of magnetic resonance imaging (MRI) thoracic spine. T2 section of MRI thoracic spine revealed pathological collapse of D2 vertebra and abscess collection (marked with arrow) anterior to vertebral body and in the epidural space compressing the spinal cord.
The total counts, erythrocyte sedimentation rate (ESR), and C-reactive protein were significantly raised (Table 1).
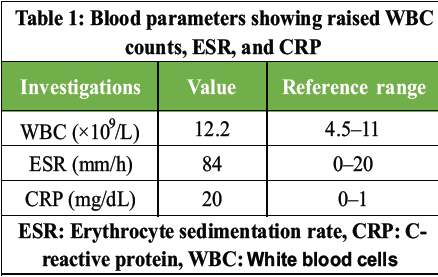
Table 1: Blood parameters showing raised WBC counts, ESR, and CRP
At this stage, clinical diagnosis of thoracic compressive myelopathy due to a pathological collapse of D2 vertebra with epidural abscess was made. In view of the acute onset of paraplegia, urgent decompression and stabilization surgery was planned. The evaluation y Obstetrician revealed a live and healthy fetus. After discussion and opinion from the obstetrician, pediatrician, and anesthesiologist, the decision to continue the pregnancy was made. Before proceeding for surgery, the patient and family were explained about the risks to the fetus, including the possibility of spontaneous abortion. Continuous intraoperative electronic fetal monitoring method was not possible in the second trimester; hence, pre- and post-surgery Doppler was used for fetal heart rate (FHR) monitoring. Surgery was started in the presence of a team of obstetricians and pediatricians. Special consideration was given for the prone positioning of the patient. Big and heavy bolsters were used and kept as apart as possible to avoid any compression over the abdomen. A lead apron was used for covering the lower back and abdomen to protect the fetus from the radiation exposure. Surgery was contemplated through the posterior approach. Pedicle screws were inserted in D1 and D3 vertebrae bilaterally using a free-hand technique. Only two C-arm shots were used during instrumentation, one for checking the level and the other after putting the screws. D2 complete laminectomy and costotransversectomy were performed to facilitate better anterior decompression of the cord. The organized pus and granulation tissue were evacuated and sent for aerobic culture, Gene Xpert, and histopathological study. The patient tolerated the surgery well, and the repeat Doppler revealed a live fetus with a normal heart rate. On the 1st post-operative day, her neurological status was improved, and she was able to do voluntary toe flickering, and sensations in both legs were also improved. She was put on anti-tuberculosis (TB) drugs after confirmation of tuberculous infection on Gene Xpert and histopathology. For the first 3 months, she took four first-line drugs (Rifampicin, Isoniazid, Pyrazinamide, and Ethambutol) followed by two drugs (Rifampicin and Isoniazid) for the next 9 months. At regular follow-up (every 4–6 weeks), ESR and liver function tests were assessed for checking the compliance of treatment (Table 2).
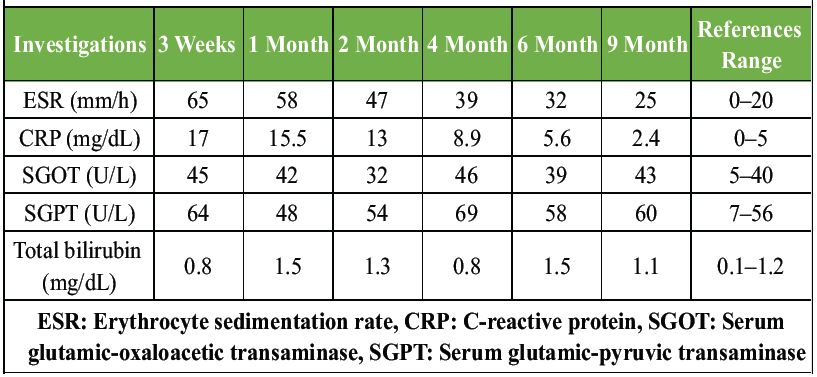
Table 2: Sequential changes in ESR, CRP, and liver function tests during treatment
At 36 weeks of gestation, she had pre-mature rupture of membranes and had difficulty in perceiving the labor contractions; hence, she underwent emergency cesarean section. She delivered a healthy male child with a birth weight of 2.8 kg and was kept in a neonatal intensive care unit for a couple of days for monitoring. She was able to stand and walk with walker support after 3 months and became an independent ambulator by 5 months post-surgery. At the past follow-up, 12 months post-surgery; she has regained full motor power in her legs and is able to walk independently. Repeat MRI dorsal spine showed good bony healing and no neural compression. Her baby was also healthy and had normal developmental milestones till date. Post-operative X-rays (Fig. 2a and b), and computed tomography scans (Fig. 3a and b) were done after delivery.
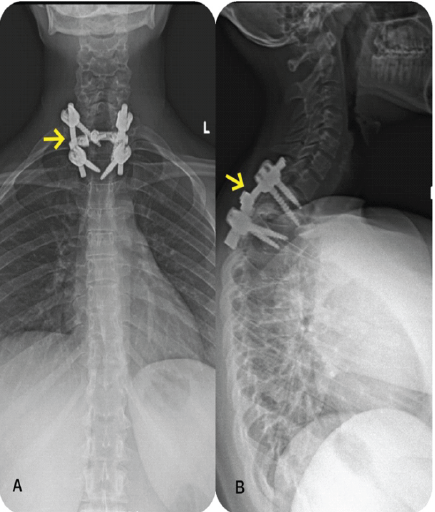
Figure 2: (a and b) Post-operative anteroposterior (a) and lateral, (b) X-rays of cervicodorsolumbar spine showing pedicle screw fixation. Arrowed areas show pedicle screw fixation from D1-D3.
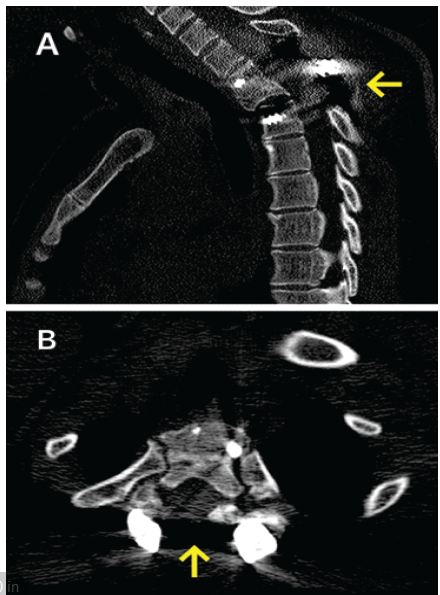
Figure 3: Post-operative computed tomography (CT) saggital and axial cuts. (a) Non-contrast sagittal CT scan of dorsal spine, (b) non-contrast axial CT scan of dorsal spine at screw level. Arrowed areas show a laminectomy with pedicle screw fixation.
TB is one of the top causes of death globally in women of reproductive age (15–45 years), a leading non-obstetric cause of maternal mortality [7]. The prevalence is even higher in human immunodeficiency virus-positive patients [8,9,10,11]. Tuberculous spondylodiscitis accounts for 50% of cases of all musculoskeletal TB and neurological involvement is seen in 20–40% of cases of Pott’s spine. However, the exact data on Pott’s paraplegia complicating the pregnancy is very limited; hence, the treatment protocol is also not clearly defined [2,3,4,5,6]. Pregnancy is a relatively immunocompromised state due to the lower level of the T-helper 1 (Th1), which may mask symptoms while increasing susceptibility to new infection or the reactivation of TB. After delivery, exacerbation of symptoms can be seen due to the effect of the reversion of Th1 suppression [12]. Therefore, rapid progression and destruction of the spine are seen during pregnancy, and it needs to control the disease at the earliest [13]. TB infection poses a risk of spontaneous abortion. Vertical transmission of infection from mother to fetus can occur through the hematogenous route or by aspiration of amniotic fluid during delivery, or respiratory droplets postpartum, which can increase the risk of fetal distress, low birth weight, pre-maturity, and infant mortality [14,15,16]. Early evaluation and diagnosis of tuberculous spondylodiscitis is often overlooked since non-specific back pain commonly occurs during pregnancy, and constitutional symptoms, such as loss of weight and fever, are often masked by the physiological weight gain during the pregnancy [17]. X-rays of the spine are avoided due to the risk of radiation exposure on developing fetus. MRI is an investigation of choice for evaluation of infective spondylodiscitis, especially in pregnant women. It is a non-ionizing modality and has been shown to have no adverse effect on the growing fetus or on pregnancy. However, Gadolinium-enhanced MRI at any time of pregnancy should be avoided as it is associated with rheumatological, inflammatory, or infiltrative skin conditions and risk of stillbirth or neonatal death [18]. Even after the radiological diagnosis of infective spondylodiscitis, knowledge of histological diagnosis is of prime importance for starting of the correct antibiotic treatment. Molecular testing methods, such as polymerase chain reaction or Interferon-gamma release assay (tests) have a low sensitivity in diagnosing musculoskeletal TB and their results are not widely validated in pregnant women [19]. In our case, MRI was done to ascertain the cause of paraplegia, and surgery was performed to decompress the spinal cord. At the time of surgery, the abscess and infected granulation tissue were sent for the Gene Xpert, aerobic culture, and histological study. ATT was started only after confirmation on the Gene Xpert and the presence of granuloma on the histological study. Medical management of spinal TB consists of a course of ATT. All first-line ATT drugs, Isoniazid, Rifampicin, Pyrazinamide, and Ethambutol, except Streptomycin, are safe for maternal-fetal health and do not require any dose adjustment in pregnancy. ATT is given in two phases: The initial phase consists of all four drugs for 2 months, followed by a continuation phase of Isoniazid and Rifampicin for 6–9 months [13]. Although the total duration of ATT for spinal TB is a matter of controversy, and the commonly followed regimes are for 9–12 months [1]. In cases of multidrug-resistant-TB, 2nd line ATT regimen is to be followed, better under the care of an infection specialist. Our patient took first-line ATT for a total duration of 12 months: Isoniazid, Rifampicin, Pyrazinamide, and Ethambutol for 2 months, followed by Isoniazid and Rifampicin for the next 10 months. Patients with severe or progressive neurologic deficits or with severe mechanical instability pain may require surgical management. However, the protocol on the surgical management of Pott’s paraplegia in pregnancy is not clearly described in the literature. Some authors, based on their experience with limited cases, have suggested for abortion followed by surgical management for the first-trimester pregnancy and combined cesarean section and spine surgery for the third-trimester pregnancy [2,3,19]. Spinal TB with paraplegia during second-trimester pregnancy is very rare, and the knowledge about the management is guided by limited case reports. Vaidya et al., managed Pott’s paraplegia surgically after the induced delivery at 24 weeks of gestation, while Han et al., [19] managed T3-T5 Pott’s paraplegia in 16-week pregnant women, by MTP and spinal fusion surgery [3]. On the other hand, Govender et al., delayed the surgical management in 24-week pregnant women till the term delivery [2]. We have performed the surgery in the prone position, although some authors have recommended against the prone position after the first trimester, to avoid aortocaval compression by the gravid uterus [20]. Special big and heavy bolsters were used and kept them as apart as possible to avoid any compression over the abdomen. A lead apron was used for covering the lower back and abdomen to protect the fetus from the radiation exposure.
Anesthesia and obstetric considerations during surgical management in pregnant women [20].
Pregnant women should not be denied any indicated non-obstetric surgery regardless of trimester, as currently used anesthetic agents are not shown to have any teratogenic effects in humans when used in standard concentrations at any gestational age. Whenever possible, any elective surgery should be avoided or at least postponed till the second trimester when pre-term contractions and spontaneous abortion are least likely to occur. Non-obstetric surgery should be performed in the presence of an obstetrician and at an institution, fully equipped with neonatal and pediatric care. Fetal monitoring should be done before and after the procedure. Intraoperative electronic fetal monitoring is appropriate and possible when a fetus is viable; however, until the age of viability, a Doppler machine can be used for FHR monitoring. Another important aspect in planning a non-obstetric surgery is to obtain informed consent from the patient and family for an emergency cesarean delivery
MRI is safe and the diagnosis of choice during pregnancy for the evaluation of spine pathology.
- ATT are safe during pregnancy and cause no adverse effect on the developing fetus
- ATT treatment should be started as soon as the diagnosis is made, as the tubercular infection is known to cause growth retardation of the fetus or spontaneous abortion
- A pregnant patient should not be denied the indicated surgical management of Pott’s paraplegia, irrespective of the trimester of pregnancy
- Prone position for surgery can be utilized for addressing the pathology at upper thoracic or cervical levels; however, care should be taken to keep the abdomen free from any compression
- Surgery should be performed in the presence of an obstetrician and at an institution, fully equipped with neonatal and pediatric care.
Pott’s paraplegia presenting during the second trimester of pregnancy requires careful and thorough neurological assessment and should not be managed conservatively when acute deficits are present. Adequate surgical decompression and stabilization, when performed timely with a multidisciplinary obstetric and anesthetic approach, can yield good neurological recovery without compromising fetal outcomes.
References
- 1. Nene AM, Pawar U. Tuberculosis of the spine 2011 update. ArgoSpine News J 2011;23:105-9. [Google Scholar] [PubMed]
- 2. Govender S, Moodley SC, Grootboom MJ. Tuberculous paraplegia during pregnancy. A report of 4 cases. S Afr Med J 1989;75:190-2. [Google Scholar] [PubMed]
- 3. Vaidya MK, Shah GV, Bharucha KE. Pregnancy and its outcome in quadriplegia due to Pott’s spine. Int J Gynaecol Obstet 1995;49:319-21. [Google Scholar] [PubMed]
- 4. Badve SA, Ghate SD, Badve MS, Rustagi T, Macchiwala T, Parekh AN, et al. Tuberculosis of spine with neurological deficit in advanced pregnancy: A report of three cases. Spine J 2011;11:e9-16. [Google Scholar] [PubMed]
- 5. Goel P, Gupta R, Devi K, Aggarwal A, Bahadur R, Malhotra S. Pregnancy complicated by paraplegia due to Pott’s spine. J Indian Med Assoc 2004;102:508, 518. [Google Scholar] [PubMed]
- 6. Alajaji NM, Sallout B, Baradwan S. A 27-year-old woman diagnosed with tuberculous spondylitis, or pott disease, during pregnancy: A case report. Am J Case Rep 2022;23:e936583. [Google Scholar] [PubMed]
- 7. Morton AJ, Roddy Mitchell A, Melville RE, Hui L, Tong SY, Dunstan SJ, et al. Mycobacterium tuberculosis infection in pregnancy: A systematic review. PLOS Glob Public Health 2024;4:e0003578. [Google Scholar] [PubMed]
- 8. Gupta A, Nayak U, Ram M, Bhosale R, Patil S, Basavraj A, et al. Postpartum tuberculosis incidence and mortality among HIV-infected women and their infants in Pune, India, 2002-2005. Clin Infect Dis 2007;45:241-9. [Google Scholar] [PubMed]
- 9. Jonnalagadda S, Lohman Payne B, Brown E, Wamalwa D, Maleche Obimbo E, Majiwa M, et al. Latent tuberculosis detection by interferon γ release assay during pregnancy predicts active tuberculosis and mortality in human immunodeficiency virus type 1-infected women and their children. J Infect Dis 2010;202:1826-35. [Google Scholar] [PubMed]
- 10. Margono F, Mroueh J, Garely A, White D, Duerr A, Minkoff HL. Resurgence of active tuberculosis among pregnant women. Obstet Gynecol 1994;83:911-4. [Google Scholar] [PubMed]
- 11. Kothari A, Mahadevan N, Girling J. Tuberculosis and pregnancy–results of a study in a high prevalence area in London. Eur J Obstet Gynecol Reprod Biol 2006;126:48-55. [Google Scholar] [PubMed]
- 12. Wilsher ML, Hagan C, Prestidge R, Wells AU, Murison G. Human in vitro immune responses to Mycobacterium tuberculosis. Tuber Lung Dis 1999;79:371-7. [Google Scholar] [PubMed]
- 13. Mathad JS, Gupta A. Tuberculosis in pregnant and postpartum women: Epidemiology, management, and research gaps. Clin Infect Dis 2012;55:1532-49. [Google Scholar] [PubMed]
- 14. Pillay T, Sturm AW, Khan M, Adhikari M, Moodley J, Connolly C, et al. Vertical transmission of Mycobacterium tuberculosis in KwaZulu Natal: Impact of HIV-1 co-infection. Int J Tuberc Lung Dis 2004;8:59-69. [Google Scholar] [PubMed]
- 15. Jana N, Vasishta K, Jindal SK, Khunnu B, Ghosh K. Perinatal outcome in pregnancies complicated by pulmonary tuberculosis. Int J Gynaecol Obstet 1994;44:119-24. [Google Scholar] [PubMed]
- 16. Jana N, Vasishta K, Saha SC, Ghosh K. Obstetrical outcomes among women with extrapulmonary tuberculosis. N Engl J Med 1999;341:645-9. [Google Scholar] [PubMed]
- 17. Robertson DN. Pregnancy and labour in the paraplegic. Paraplegia 1972;10:209-12. [Google Scholar] [PubMed]
- 18. Ray JG, Vermeulen MJ, Bharatha A, Montanera WJ, Park AL. Association between MRI exposure during pregnancy and fetal and childhood outcomes. JAMA 2016;316:952-61. [Google Scholar] [PubMed]
- 19. Han IH, Kuh SU, Kim JH, Chin DK, Kim KS, Yoon YS, et al. Clinical approach and surgical strategy for spinal diseases in pregnant women: A report of ten cases. Spine (Phila Pa 1976) 2008;33:E614-9. [Google Scholar] [PubMed]
- 20. Committee opinion No.696. Nonobstetric surgery during pregnancy. Obstet and Gynecol 2017;129:777-8. [Google Scholar] [PubMed]







