Preservation of the tibial attachment in hamstring graft anterior cruciate ligament reconstruction helps maintain graft vascularity, resulting in better early graft viability and favorable ligamentization as evidenced by predominantly low–intermediate magnetic resonance imaging signal intensity.
Dr. Wasif Kawsar Qadri, Department of Orthopaedics, Lady Hardinge Medical College, New Delhi, India. E-mail: wasifkqadri14@gmail.com
Background: Hamstring (HS) tendon autografts are widely used for anterior cruciate ligament (ACL) reconstruction. Conventional graft harvesting requires detachment from the tibial insertion, which may compromise graft vascularity and result in early graft necrosis during ligamentization. Preservation of the tibial attachment may help maintain the intrinsic blood supply and enhance early graft viability. However, magnetic resonance imaging (MRI)-based evidence evaluating graft behavior during the early necrotic–proliferative phase remains limited.
Methods: This prospective observational study included 30 patients who underwent anatomic single-bundle ACL reconstruction using a tibial attachment–preserving HS tendon autograft. Clinical stability was assessed using Lachman and pivot shift tests, while functional outcomes were evaluated using the Lysholm knee score and Tegner activity level. MRI assessment was performed at 8 weeks postoperatively to evaluate early graft morphology and signal intensity during the biologically vulnerable phase of ligamentization. Graft signal intensity was graded using the Figueroa classification. All patients were followed clinically for a minimum of 6 months.
Results: The mean Lysholm knee score improved from 54.4 preoperatively to 92.4 at final follow-up, while the mean Tegner activity level increased from 3 to 5.27. At final follow-up, the pivot shift test was negative in all patients. Lachman testing demonstrated grade 0 laxity in 29 patients and grade 2 laxity in one patient. MRI at 8 weeks confirmed anatomically positioned grafts in all cases. Hypointense or isointense graft signal intensity was observed in 29 patients, whereas one patient demonstrated hyperintense signal characteristics.
Conclusion: ACL reconstruction using a tibial attachment – preserving HS autograft resulted in favorable early clinical stability and functional recovery. Early MRI findings demonstrated predominantly hypo- to isointense graft signals, suggesting preservation of graft morphology during the early phase of ligamentization. Larger comparative studies with longer follow-up are needed to determine the long-term clinical relevance of these findings. Level of evidence Level IV, prospective observational study.
Keywords: Anterior cruciate ligament, anterior cruciate ligament reconstruction, hamstring graft, preserved tibial attachment, ligamentization.
Hamstring (HS) tendons are among the most commonly used autografts for anterior cruciate ligament (ACL) reconstruction because of their reliable availability, favorable biomechanical strength, and low donor-site morbidity. Conventionally, these grafts are completely detached from their tibial insertion before implantation. Following reconstruction, detached HS grafts undergo a biological process known as ligamentization, which comprises an initial phase of graft necrosis, followed by cellular proliferation, revascularization, and subsequent remodeling toward the biological and mechanical characteristics of the native ACL [1].
The ligamentization process can be indirectly assessed using magnetic resonance imaging (MRI). During the early necrotic and proliferative phases, grafts typically demonstrate increased signal intensity on T1- and T2-weighted sequences, reflecting immature collagen organization and reduced biomechanical strength. With progressive maturation, the graft signal gradually becomes homogeneous and hypointense, resembling that of a native ACL over a period of 1–3 years [2]. An intact, well-matured ACL graft usually demonstrates low to intermediate signal intensity on sagittal and oblique T2-weighted images, coronal proton density fat-suppressed images, and short tau inversion recovery sequences. Although mild intermediate signal intensity may be observed during normal healing, the presence of fluid-equivalent signal traversing the graft is considered abnormal.
Hyperintense graft signal intensity on MRI has been associated with inferior biomechanical properties, incomplete graft maturation, and an increased risk of graft failure. Conversely, lower graft signal intensity and preserved graft volume are generally regarded as surrogate markers of improved graft maturity and structural organization [3]. Several studies have demonstrated a correlation between MRI-derived graft characteristics and mid- to long-term clinical outcomes, underscoring the role of MRI as a reliable, non-invasive tool for monitoring graft healing following ACL reconstruction.
During the early post-operative period, reconstructed ACL grafts are biomechanically vulnerable because of graft necrosis and incomplete biological incorporation. This vulnerability necessitates cautious rehabilitation protocols and generally precludes early return to sports. Consequently, multiple strategies have been explored to enhance graft healing and accelerate ligamentization, including preservation of the native ACL remnant, periosteal graft augmentation, and biological adjuvants such as platelet-rich plasma and amniotic products. However, despite promising experimental data, consistent clinical evidence supporting the routine use of these techniques remains limited, and none have been universally adopted into standard clinical practice [4].
Experimental animal studies have demonstrated that preservation of the tibial insertion of HS grafts may maintain intrinsic graft vascularity and neural elements, thereby improving graft viability and potentially bypassing the avascular necrotic phase of ligamentization [5,6]. Clinically, tibial attachment – preserving HS graft techniques have been shown to yield satisfactory functional outcomes without increasing surgical complexity or donor-site morbidity [7]. A prospective randomized comparative study evaluating attachment-preserving versus free HS grafts reported comparable clinical outcomes in both groups, while demonstrating superior intra-articular graft morphology on MRI at 6 months in the attachment-preserving cohort [8]. Similarly, a longitudinal MRI-based study reported relatively uniform graft signal intensity in attachment-sparing HS grafts at 12 weeks, 6 months, and 2 years postoperatively, suggesting a more stable and consistent maturation profile [9].
Despite these encouraging observations, there remains a paucity of data specifically evaluating graft behavior during the critical early phases of ligamentization – namely the necrotic (0–4 weeks) and proliferative (4–12 weeks) phases – when the graft is believed to be most biologically and mechanically vulnerable. In particular, early post-operative MRI assessment during this window has received limited attention as a means of indirectly evaluating graft viability and maturation.
Therefore, the present study aimed to evaluate early graft morphology using MRI at 8 weeks following ACL reconstruction with tibial attachment – preserving HS grafts. This time point was selected to coincide with the transition between the necrotic and proliferative phases of ligamentization. By assessing graft signal characteristics during this early post-operative period, the study sought to determine whether preservation of the tibial insertion may allow bypassing of the necrotic and proliferative phases. Maintenance of graft viability during this critical period may have important implications for post-operative rehabilitation strategies and timing of return to functional activity, although such considerations warrant further investigation.
Study design and patient selection
This prospective observational study included 30 consecutive patients who underwent anatomic single-bundle ACL reconstruction using an HS tendon autograft with preservation of the tibial attachment. The study was conducted at a tertiary care orthopedic center after obtaining approval from the institutional ethics committee, with ethical clearance number 116/ETH/GMC dated March 30, 2017, and written informed consent was obtained from all participants before enrollment.
All patients had symptomatic ACL insufficiency confirmed by clinical examination and MRI. Pre-operative clinical assessment included the Lachman and pivot shift tests. Functional status was evaluated preoperatively using validated outcome measures, namely the Lysholm knee score and Tegner activity level [10].
Surgical technique
All surgical procedures were performed by the senior author under spinal anesthesia with tourniquet control, following a standardized operative protocol. HS graft harvest was performed through a 3–4 cm longitudinal incision over the pes anserinus, located approximately 4 cm distal to the joint line and 3 cm medial to the tibial tuberosity. The sartorial fascia was incised to expose the semitendinosus and gracilis tendons. After careful identification, fibrous expansions to the gastrocnemius and semimembranosus muscles were released. Both tendons were harvested using an open-loop tendon stripper, with deliberate preservation of their tibial insertion.
The free ends of the tendons were whip-stitched using non-absorbable Ethibond sutures for a length of 10–15 mm. The tendons were then folded to create a quadrupled graft, which was passed through cylindrical sizers to determine the appropriate tunnel diameter. The final graft diameter ranged from 7 to 9 mm and consisted of four strands of semitendinosus and gracilis tendons (Figs. 1 and 2).
An accessory anteromedial portal was used in addition to standard anterolateral and anteromedial portals to facilitate anatomic femoral tunnel placement [11]. The femoral footprint was identified using established osseous landmarks, including the lateral intercondylar ridge and the lateral bifurcate ridge. Tibial tunnel placement in the anteroposterior plane was guided by extending a line from the inner margin of the anterior horn of the lateral meniscus, approximately 7 mm anterior to the posterior cruciate ligament. Mediolateral positioning was centered within the native ACL footprint, corresponding to the depression medial to the medial tibial spine.
Femoral and tibial tunnels were prepared in a standard fashion. After confirming appropriate tunnel length and graft sizing, the graft was passed through the tunnels. Femoral fixation was achieved using a cortical suspensory device (fixed loop EndoButton). The knee was cycled through approximately 25 repetitions from 0° to 120° of flexion to precondition the graft and achieve uniform tension. Tibial fixation was performed at 20° of knee flexion using a U-staple on the free limb of graft.
Post-operative rehabilitation
All patients followed a standardized accelerated rehabilitation protocol as described by Shelbourne and Nitz [12]. Weight-bearing and range-of-motion exercises were initiated early and progressed as tolerated. Advancement to higher-level activities was guided by clinical stability and functional recovery.
MRI evaluation
MRI evaluation was performed at 8 weeks postoperatively to assess early graft morphology during the anticipated necrotic–proliferative phase of ligamentization. MRI analysis focused on graft integrity, anatomical positioning, and signal intensity characteristics of the intra-articular portion of the graft (Fig. 3). Particular emphasis was placed on graft signal intensity as an indirect marker of graft viability and maturation.
Graft morphology was objectively assessed using the Figueroa scoring system, which evaluates graft signal intensity relative to reference structures such as the posterior cruciate ligament and adjacent musculature [13,14]. The total integration score was not calculated, as complete graft incorporation is not expected during this early post-operative period.
Clinical follow-up and outcome measures
Patients were followed for a minimum duration of 6 months. Final clinical evaluation included assessment of knee stability using manual Lachman and pivot shift tests. Functional outcomes were reassessed using the Lysholm knee score and Tegner activity level to determine post-operative functional recovery.
Statistical analysis
Statistical analysis was performed using standard statistical software. Continuous variables were expressed as means and ranges, while categorical variables were reported as frequencies and percentages. Pre-operative and post-operative Lysholm knee scores and Tegner activity levels were compared using a paired t-test for normally distributed data. A P < 0.05 was considered statistically significant. Given the descriptive nature of MRI assessment and the absence of a comparative control group, MRI findings were analyzed descriptively without inferential statistical testing.
The study cohort comprised 30 patients, including 26 males (87%) and 4 females (13%), with a mean age of 28.7 years (range, 18–45 years). The right knee was involved in 17 patients (57%) and the left knee in 13 patients (43%). The interval between injury and surgery ranged from 6 weeks to 5 years, with a mean duration of 12 months. The mechanism of injury was sports related in 16 patients (53%), falls in 8 patients (27%), and road traffic accidents in 6 patients (20%).
The mean duration of surgery was 94.66 min (range, 80–120 min). Associated intra-articular injuries were identified in 10 patients (33%), including medial meniscal tears in 7 patients (23%), lateral meniscal tears in 2 patients (7%), and combined medial and lateral meniscal tears in 1 patient (3%).
At final follow-up, the pivot shift test was negative in all patients. Lachman test grading demonstrated grade 0 laxity in 29 patients (96.66%) and grade 2 laxity in 1 patient (3.33%). No patient demonstrated grade 3 laxity (Table 1).
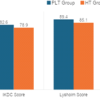
Functional outcomes showed statistically and clinically significant improvement following surgery. The mean Lysholm knee score improved from a pre-operative mean of 54.43 to a final mean score of 92.4. Preoperatively, Lysholm scores were categorized as poor (<65) in 18 patients (60%) and fair (65–83) in 12 patients (40%). At final follow-up, scores improved to excellent (>95) in 23 patients (76.67%) and good (84–94) in 7 patients (23.33%), with no patient demonstrating a poor or fair outcome. The mean Tegner activity level improved from a pre-operative value of 3 (range, 2–4) to a post-operative value of 5.27 (range, 3–8).
Post-operative complications were minimal. Three patients (10%) reported numbness or paresthesia in the saphenous nerve distribution, and two patients (6.66%) experienced localized tenderness at the tibial fixation site. No major complications such as infection, graft failure, or revision surgery were observed.
MRI evaluation performed at 8 weeks postoperatively demonstrated anatomic graft placement in all patients. Graft positioning was within accepted anatomical limits based on assessment of graft inclination angle, relationship to Blumensaat’s line, femoral tunnel clock-face position, femoral graft angle, and tibial tunnel center expressed as a percentage of maximum anteroposterior and mediolateral tibial dimensions (Table 2).
Assessment of graft morphology using the Figueroa scoring system revealed hypointense (signal intensity comparable to the posterior cruciate ligament) or isointense (signal comparable to adjacent musculature) graft signal in 29 patients. One patient demonstrated a hyperintense graft signal. The total Figueroa score was not calculated, as complete graft integration is not expected during this early stage of ligamentization. Overall, MRI findings indicated preservation of graft morphology without evidence of overt graft necrosis during the early post-operative period (Figs. 4 and 5).
In the present study, clinical outcomes assessed using Lachman test grading, Lysholm knee score, and Tegner activity level at final follow-up were comparable to those reported in previously published series utilizing tibial attachment – preserving HS grafts (Table 3) [7,8,15,16]. These findings indicate that preservation of the tibial insertion does not compromise short-term clinical stability or functional recovery following ACL reconstruction. To our knowledge, this study represents one of the few clinical investigations specifically evaluating graft morphology during the early necrotic – proliferative phase of ligamentization following tibial attachment – preserving HS ACL reconstruction.
The primary objective of this study was to evaluate early graft behavior during the ligamentization process, based on the hypothesis that preservation of the tibial insertion may maintain intrinsic graft vascularity and thereby mitigate or bypass the avascular necrotic phase of ligamentization. Given the impracticality of direct histological assessment in clinical settings, graft viability was indirectly assessed using MRI signal characteristics as a surrogate marker. At 8 weeks postoperatively, grafts were visualized in all patients, with hypointense or isointense signal characteristics observed in 29 of 30 patients. These findings suggest preservation of graft morphology and absence of overt graft necrosis during this biologically vulnerable early post-operative period.
Preservation of graft morphology during early healing may be clinically relevant, as the reconstructed ACL is known to be biomechanically vulnerable during the necrotic and early proliferative phases of ligamentization. Maintenance of graft viability during this period may reduce susceptibility to early graft elongation or reinjury and supports the concept of individualized, criterion-based rehabilitation protocols rather than uniform post-operative restrictions.
The findings of the present study are consistent with experimental and clinical observations reported by Zaffagnini et al. [17] and Fang et al., [18] who demonstrated that preservation of the tibial insertion maintains neurovascular structures supplying the graft, potentially avoiding the mechanical impairment associated with graft necrosis. Reduced early graft necrosis may contribute to improved biological incorporation and enhanced structural integrity of the reconstructed ligament, although the clinical implications of this effect require further validation.
Several clinical studies have reported similar observations. Ruffilli et al. [8] found no difference in final ligamentization between attachment-preserving and detached HS grafts at 6 months; however, superior graft incorporation was noted in the attachment-preserving group. Liu et al. [9] demonstrated that insertion-preserved HS grafts exhibited lower and more stable MRI signal intensity at 12 weeks, 6 months, and 2 years compared with detached grafts, which showed progressive signal changes over time. More recently, Zhang et al. [19] reported earlier functional recovery and greater long-term stability over a 60-month follow-up period in patients undergoing insertion-preserved HS graft reconstruction. Similarly, Grassi et al. [20] observed improved MRI features – including reduced tunnel enlargement, lower signal-to-noise quotient, and decreased intra-articular fluid – in grafts with preserved tibial insertion.
The present study adds to the existing body of literature by specifically focusing on the early post-operative window corresponding to the necrotic and proliferative phases of ligamentization. The demonstration of predominantly low or intermediate graft signal intensity at 8 weeks suggests that preservation of the tibial insertion may favor early graft viability. In contrast, conventional ACL reconstruction using detached HS grafts is known to undergo an initial phase of graft necrosis, accompanied by a temporary reduction in mechanical properties during early healing.
Nevertheless, it remains uncertain whether favorable early graft morphology on MRI directly translates into superior biomechanical strength, accelerated rehabilitation, or earlier return to sports. Addressing these questions will require well-designed prospective comparative studies incorporating quantitative MRI parameters, biomechanical assessment, and longer-term functional outcomes.
Limitations
The present study has certain limitations. The sample size was relatively small and the study lacked a comparative control group using conventionally detached HS grafts, which limits definitive conclusions regarding the superiority of the tibial attachment – preserving technique. Assessment of graft viability was indirect and based on MRI signal characteristics rather than histological evaluation or advanced quantitative imaging, which may not fully reflect the biological process of ligamentization. Furthermore, MRI assessment was performed at a single early post-operative time point, and longer-term radiological follow-up would be required to correlate early graft signal characteristics with sustained clinical outcomes. Objective instrumented laxity measurements were not used, and stability assessment relied on clinical examination, which may introduce observer-related variability. Finally, as this was a single-center study, the findings may not be generalizable to all patient populations or surgical practices.
Future multicentric randomized or comparative studies with larger sample sizes, longer clinical and radiological follow-up, and objective assessment of graft maturation are required to validate the potential biological and functional advantages of tibial attachment – preserving HS grafts in ACL reconstruction. Further studies incorporating quantitative MRI parameters, comparative cohorts, and return-to-sport metrics may better define the clinical relevance of early graft signal characteristics.
ACL reconstruction using a tibial attachment – preserving HS autograft resulted in satisfactory early clinical stability and significant functional improvement. Early post-operative MRI evaluation demonstrated predominantly hypo- to isointense graft signal characteristics at 8 weeks, suggesting preservation of graft morphology during the biologically vulnerable early phase of ligamentization. While these findings support the potential biological advantage of tibial attachment preservation, larger comparative studies with longer follow-up are required to determine whether these early MRI features translate into superior long-term clinical outcomes.
Tibial attachment–preserving HS graft in ACL reconstruction is a safe and effective technique that provides good early clinical stability and functional outcomes, while potentially enhancing early graft viability and ligamentization – thus supporting its use as a biologically advantageous alternative to conventional graft harvesting.